a) What is the equilibrium that influences the colour change of the Eriochrome Black T indicator? b) Consider how ETDA coordinates to metal ions and then consider this ligand below. Could this ligand be substituted for EDTA in a similar experiment? Why? Identify all possible donor atoms in the structure c) What is the function of the buffer solution for this experiment? Consider the form of EDTA (H4Y) compared to the form in the reaction equation below: The reaction of ETDA with metal cations, e.g. Mn+, may be written as: Mn+ + H2Y2– = MYn–4 + 2 H+
a) What is the equilibrium that influences the colour change of the Eriochrome Black T indicator? b) Consider how ETDA coordinates to metal ions and then consider this ligand below. Could this ligand be substituted for EDTA in a similar experiment? Why? Identify all possible donor atoms in the structure c) What is the function of the buffer solution for this experiment? Consider the form of EDTA (H4Y) compared to the form in the reaction equation below: The reaction of ETDA with metal cations, e.g. Mn+, may be written as: Mn+ + H2Y2– = MYn–4 + 2 H+
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter2: Atoms, Molecules, And Ions
Section: Chapter Questions
Problem 81QAP
Related questions
Question
a) What is the equilibrium that influences the colour change of the Eriochrome Black T indicator?
b) Consider how ETDA coordinates to metal ions and then consider this ligand below. Could this ligand be substituted for EDTA in a similar experiment? Why? Identify all possible donor atoms in the structure
c)
What is the function of the buffer solution for this experiment? Consider the form of EDTA (H4Y) compared to the form in the reaction equation below:
The reaction of ETDA with metal cations, e.g. Mn+, may be written as:
Mn+ + H2Y2– = MYn–4 + 2 H+
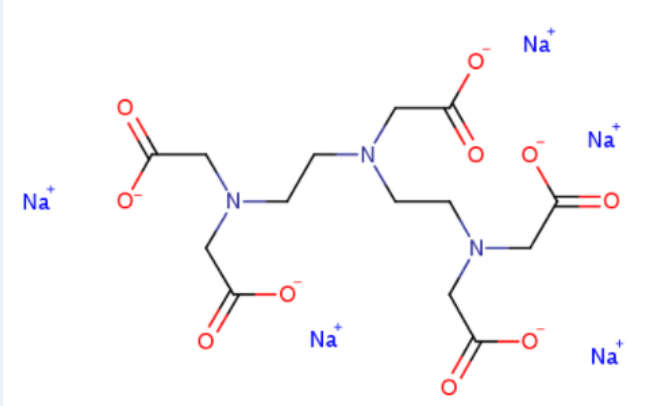
Transcribed Image Text:Na
-N
Na
Na*
N-
Na
Na
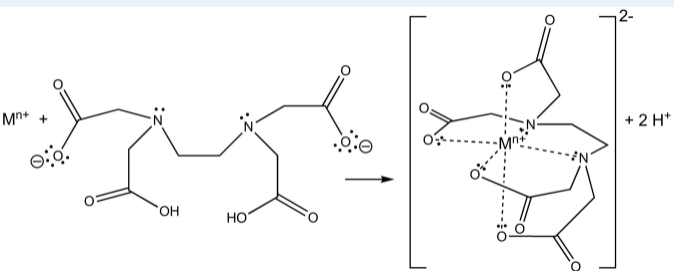
Transcribed Image Text:2-
Mn+
+ 2 H*
HO,
HO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning