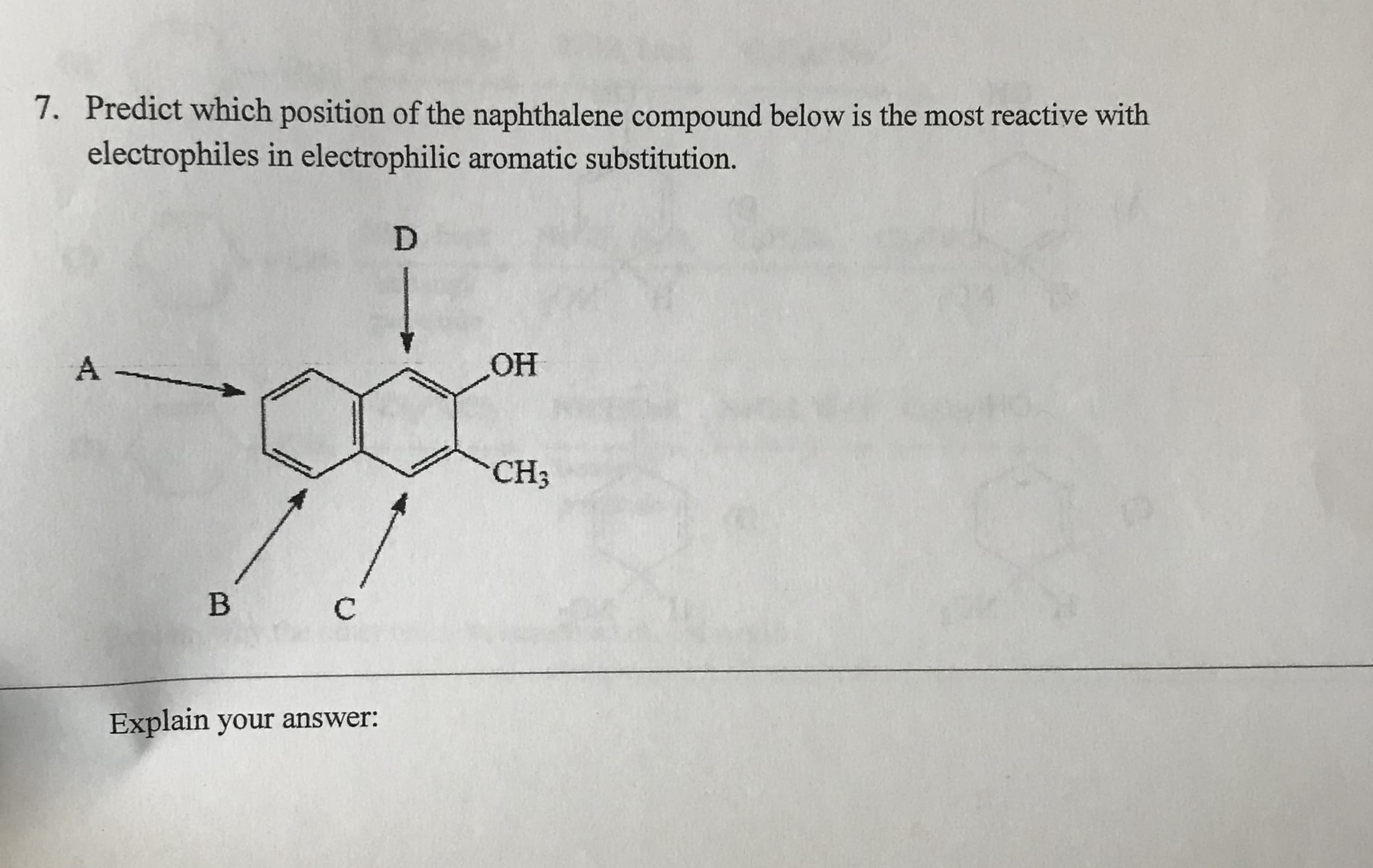
7. Predict which position of the naphthalene compound below is the most reactive with electrophiles in electrophilic aromatic substitution. D OH A CH3 B C Explain your answer:
Q: 5. How do you separate acetanilide from sand?
A: Acetanilide is a water soluble substance whereas sand is insoluble in water. Add some hot hot water...
Q: How to determine the number of moles of the C8H10N4O2, C,H,N,O in 0.1119mg of caffeine C8H10N4O2? (N...
A: The moles of compound is calculated by the following formula,
Q: CaCO3(s) + 2H+(aq) Ca2+(aq) + H2O(l) + CO2(g) this reaction occurs spontaneously as written when all...
A:
Q: A mixture is made up of 4 molecules of CH3CH2 CH2OH and 5 formula units of Pb3(PO4)4 * 6H2O. Why ar...
A: A molecule is composed of two or more elements that are covalently bonded.It is the smallest particl...
Q: Identify each as a strong electrolyte, weak electrolyte, or nonelectrolyte: a. Al2(CO3)3 ...
A: Electrolytes are the substances which ionize in water to give component ions. The ions are positive ...
Q: Indicate the answer choice that best completes the statement or answers the question 1. What is the ...
A: To explain the given statement, consider HCl molecule.
Q: 3. Which of the following is the Lewis dot structure for the fluoride ion? a. b. c.
A: The fluorine atom forms fluoride, which has 8 valence electrons, and is isoelectronic with a Noble G...
Q: What number of mole and mass of silver oxide, Ag2O, is required to produce 17.85 g of silver sulfadi...
A: The given reaction shows that the reacting ratio of Ag2O to AgC10H9N4SO2 is 1:2.Find the number of m...
Q: Questions 1. a) Write a detailed mechanism for the formation of dibenzalacetone. b) Give the IUPAC n...
A: (a)A detailed mechanism for the formation of dibenzalacetone is given by, Step 1: Deprotonation.Step...
Q: If the RDA for vitamin C is 60. mg per day, and there are 70. mg of vitamin C per 100. g of oranges,...
A: The relation between oz. and g is as follows:1 oz. = 28.3495 gThus,3 oz. = 85.0485 gThe relation bet...
Q: 2 Please wnte the Mechanism Par the reduchion of the acid Chloride Shawn belas to theCorcespalkn alc...
A: LiAlH4 is a reducing agent. It increases the concentration of hydride ions in the reaction which fav...
Q: Does a RAD measure accuracy or precision?
A: The Relative Average Deviation of a data set is a percentage that tells you how much, on average, ea...
Q: sodium chloride is 39.34% sodium . what mass of sodium chloride will contain 2.00 g of chloride .
A: Given that the percentage of sodium in sodium chloride is 39.34 %. Then the percentage of chlorine i...
Q: 4. Provide the structure of the intramolecular aldol condensation/dehydration product that results w...
A: Aldol condensation is a condensation reaction in which aldehyde having alpha-hydrogen reacts with en...
Q: X X Experimer 0 Mail Dirk X Experiment 10 B Homepag G sodium + + Yuzu: Safe X https://www.the colleg...
A: Solution for sodium chloride solute :First we have to calculate the mass of sample.
Q: In the molecule depicted by the structure shown below, Br 1 5 2 4 3 identify the carbon atoms which ...
A: The structue of the given molecule is shown as follows:
Q: 5. A balloon used for atmospheric research has a volume of 1.0 x 106 L. Assume that the balloon is f...
A: The combined gas law relates "before and after" atmospheric conditions of a gas. The volume of heli...
Q: Atomic weight of an element with two naturally occurring isotopes. Isotope Isotopic Mass Function...
A: For an element, the identifying features are its atomic number and atomic massAtomic number gives th...
Q: Which of the following bonds would be the most polar? a. C-Br b. C-I c. Cl-Cl d. C-F
A: The difference in electronegativity between the two atoms of the bond determines the polarity of the...
Q: Consider the below reaction mechanism:Step 1: S(s) + O2(g) SO2(g) fast; k1 = rate constant for forwa...
A: Step 1-A reaction mechanism is given as shown-where k1 = rate constant for forward reaction for step...
Q: Grignard Reactions Provide the structure of the major organic product in the reaction shown below. 1...
A: The Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or...
Q: How would I set up this problem. A graduated cylinder contains 19.0mL of water. What is the new wate...
A: The density tells you how many grams are equal to 1mlDensity = ρ = mass/volume The density of silver...
Q: 4. Which of the following compounds would be expected to have the lowest melting point? a. AIF3 b. R...
A: Ionic compounds with higher charges have higher melting point due to strong intermolecular forces an...
Q: Balance the equations Element/compund Element/compund Product LiCl Na S Al I2 Ba ...
A: Adjusts the coefficients of elements to write the balanced equation.For the formation of LiCl,
Q: A metal sample weighing 51.0 g and at a temperature of 100.0 oC was placed in 38.8 g of water at 25....
A: Hey, since there are subpart questions posted, we will answer first three questions. If you want any...
Q: How many grams of Sc are there in a sample of Sc that contains 4.91×1023 atoms?
A: The atomic weight of scandium is 45 gram per mole. The number of atoms present in one mole of any su...
Q: < E session.masteringchemistry.com MasteringChemistry: HW 8 <HW 8 Applications for Dilution Calculat...
A: From the given data in the question, sayThe final concentration of the diluted solution = cVolume o...
Q: What is the value of delta G at 120.00 C for a reaction in which delta H = 5.0kj/mole and delta S = ...
A: The change in Gibbs free energy is calculated as,
Q: The flask shown here contains 10.0 mL of HCl and a few drops of phenolphthalein indicator. The buret...
A: In this problem, the volume of sodium hydroxide can be calculated only when the initial concentratio...
Q: Consider the reversible reaction at equilibrium at 392oC:2A(g) + B(g) C(g)The partial pressures are ...
A: The equilibrium constant for given reversible reaction is calculated as,
Q: when acid is added to the blood buffer sytem what reacts with what to neutralize it
A: The blood buffer system contains dissolved carbon dioxide in the plasma.
Q: What is the daughter nucleus (nuclide) produced when 213Bi undergoes alpha decay?
A: Alpha decay leads to formation of a product that contains atomic number two less and mass number fou...
Q: 4. In the preparation of Acetanilide, when aniline is dissolved in HCl, what is the role of sodium a...
A: Aniline is water insoluble.
Q: 5. An atom of which of the following elements has the smallest atomic radius? a. Cl b. Rb c. Mg d. S...
A: Atomic radius is the distance between the center of nucleus and valence electron in the outermost sh...
Q: Please the follawa Componds Synthesize tram benzene Notc A Friedel -Crafts alky la tro wont wer rear...
A: The synthesis of 4-chloro toluene involves two reaction, first Friedel-Crafts alkylation and then ha...
Q: A metal "X" is used as a “sacrificial” anode on hulls of salt water vessels made of iron. The effect...
A: The voltage of a reaction in the electrochemical cell is expressed in terms of the electrode potenti...
Q: Hello, I am trying to balance a tricky (skeleton) reaction using the half reaction method: CrI3 (...
A: Step 1 : write the unbalanced chemical equation:Step2: identify and write all redox couple in react...
Q: Calculate the pH of 1M solution of acedic acid (pKa = 4.76)?
A: The pH of the acetic acid solution can be calculated using the formula shown in belowpKa = -log(10)[...
Q: Why is carbon dioxide CO2 able to be stored more effectively in ocean water vs. fresh water a. T...
A: The carbon dioxide CO2 able to be stored more effectively in ocean water than fresh water. This is ...
Q: Dcc/EN ok Amme Prodechmg Group GMNCI Br Na CEN CEN hosH T 2-2
A: Product of the reaction has to be identified.(a) Protected amino acid reaction with DCC (N,N'-Dicycl...
Q: Label each ionic compound as either "soluble" or "insoluble" in water: a. BaSO4 b. Na2CrO4 c. Cu3(...
A: (a)On the basis of solubility rules, generally sulfate (SO42-) ion forms soluble salts but BaSO4 is ...
Q: How many atoms are in one molecule of Na2SO4? How many different elements are in the compound C6H12O...
A: The compound Na2SO4 contains two sodium atoms, one sulfur atom and four oxygen atoms in its chemical...
Q: How to write a chemical equation for: _______Cu (s) + _____ O2 (g) ---> ______ Could you provid...
A: The given equation is to be completed-
Q: A chemist carries out this reaction using 10 moles of acetone and 16 moles of benzaldehyde and obtai...
A: The reaction of acetone with benzaldehyde results in the formation of dibenzalacetone. In this react...
Q: Given a reaction vessel of 0.361 M C4H8(g) which obeys first order kinetics and has a half-life of 1...
A: A first order reaction is that in which the order with respect to a reactant is one.Its integrated r...
Q: For the molecular compound sulfur trioxide, disulfur decafluoride, H20 what would you multiply the m...
A: 1 mole of a compound is equal to its 6.022×1023 molecules. Therefore, 1 mole of SO3 is equal to 6.02...
Q: Please draw the ma lor Products for the olouwing r HBr/Uvlight Hz /Lindlars Ca talyst onc
A: When the given alkene reacts with HBr under UV light, it undergoes bromination through anti-Markovni...
Q: If you are going 55 mph, what is your speed in nm per second?
A: We are going at speed of 55 mph.
Q: A 140.0 g sample of water at 25.0°C is mixed with 100.0 g of a metal at 100.0°C. After thermal equil...
A: The change in energy of a system is given by,
Q: Calculate the solubility products for the following compounds: a) SrF3 7.3x10-2 g/L b) Ag3PO4 6.7x1...
A: Step 1-Solubility- It is defined as the maximum amount of solute that can be dissolved in a solvent ...
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- 1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic centerThe compound below undergoes electrophilic aromatic substitution to form a monosubstituted product. At which position will the electrophile going to attack?Illustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Draw all the oissuvke resonance forms of methoxybenzene, including the hybrid Based on the structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.
- 6. List the following aryl halides from most to least reactive (in suzuki coupling): 2-bromotoluene, 3-bromotoluene, 4,-bromotoluene, 4-bromoanisole, 4-bromoacetophenone, 3-chlorotoluene, and 4-chloroacetophenoneQ4: Propose structure for compound that fit the following descriptions:a) An alkyl halide that gives a mixture of three alkenes on E2 reaction.b) An organo halide that will not undergo nucleophile substitution.c) An alkyl halide that gives the non-Zaitsev product on E2 reaction.d) An alcohol that reacts rapidly with HCI at 00C.In 1-methoxynaphthalene, the most probable sites during electrophilic aromatic substitution are: a. 2 and 3 b. 7 and 8 c. 2 and 4 d. 5 and 8 e. none of these
- The reaction below could run through both substitution and elimination reactions. 1. Provide the correct reagent to produce the products shown 2. State which mechanism(s) was followedRank in terms of reactivity towards the nucleophile. 1=least 4=most3-Bromocyclohexene is a secondary halide, and benzyl bromide is a primary halide. Both halides undergo SN1 substitution about as fast as most tertiary halides. Use resonance structures to explain this enhanced reactivity.
- What is the missing reagent needed to perform the following transformation? Assume mildly acidic catalysis. a) CH3NH2 b) CH2=NH c) (CH3)2NH d) CH3CH2NH2Rank the following compounds in order of increasing SN2 reaction rate with a good nucleophile. Explain yourreasoning. (CH3)2CHBr (CH3)3CBr CH3CH2Br CH3CH2Irank these from least to most reactive in nucleophilic acyl substitution with a nucleophile I)CH3COOC2H5 II) CH3COO-Na+ III)CH3COCl IV) CH3CONH2





