A biochemist carefully measures the molarity of ammonium chloride in 75.3 mL of photobacterium cell growth medium to be 44.0 µM. Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume the cell growth medium falls to 5.13 mL. Calculate the new molarity of ammonium chloride in the photobacterium cell growth medium. Be sure your answer has the correct number of significant digits.
A biochemist carefully measures the molarity of ammonium chloride in 75.3 mL of photobacterium cell growth medium to be 44.0 µM. Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume the cell growth medium falls to 5.13 mL. Calculate the new molarity of ammonium chloride in the photobacterium cell growth medium. Be sure your answer has the correct number of significant digits.
Chapter7: Solutions And Colloids
Section: Chapter Questions
Problem 7.86E
Related questions
Question
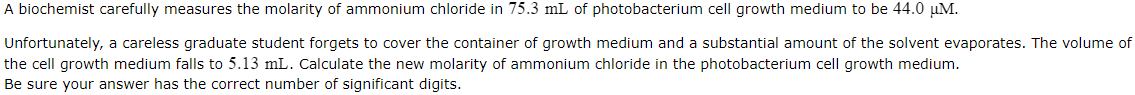
Transcribed Image Text:A biochemist carefully measures the molarity of ammonium chloride in 75.3 mL of photobacterium cell growth medium to be 44.0 µM.
Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume of
the cell growth medium falls to 5.13 mL. Calculate the new molarity of ammonium chloride in the photobacterium cell growth medium.
Be sure your answer has the correct number of significant digits.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning