A person makes a quantity of iced tea by mixing 690 g of hot tea (essentially water) with an equal mass of ice at its melting point. Assume the mixture has negligible energy exchanges with its environment. (a) If the tea's initial temperature is T; = 96°C, when thermal equilibrium is reached what are the mixture's temperature Tand (b) the remaining mass m; of ice? If T; = 68°C, when thermal equilibrium is reached what are (c) T; and (d) m? The specific heat of water is 4186 J/kg-K. The latent heat of fusion is 333 kJ/kg.
A person makes a quantity of iced tea by mixing 690 g of hot tea (essentially water) with an equal mass of ice at its melting point. Assume the mixture has negligible energy exchanges with its environment. (a) If the tea's initial temperature is T; = 96°C, when thermal equilibrium is reached what are the mixture's temperature Tand (b) the remaining mass m; of ice? If T; = 68°C, when thermal equilibrium is reached what are (c) T; and (d) m? The specific heat of water is 4186 J/kg-K. The latent heat of fusion is 333 kJ/kg.
Chapter1: Temperature And Heat
Section: Chapter Questions
Problem 80P: How many grams of coffee must evaporate from 350 g of coffee in a 100-g glass cup to cool the coffee...
Related questions
Question
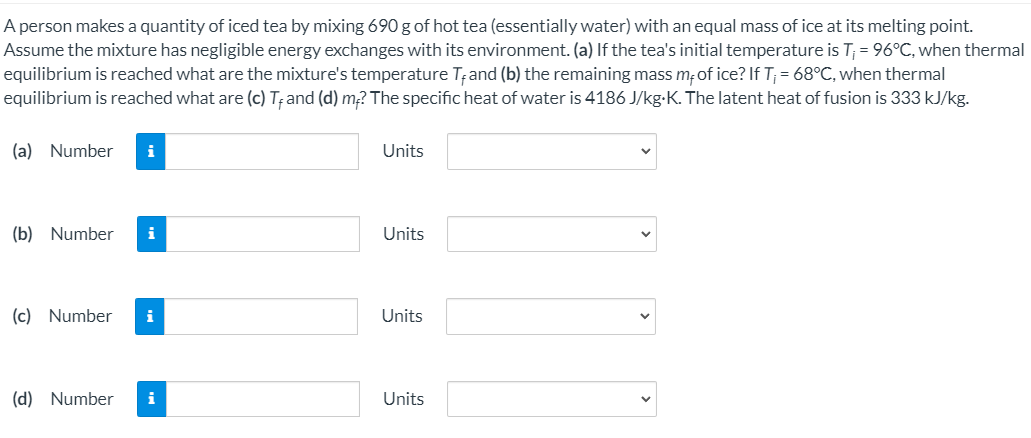
Transcribed Image Text:A person makes a quantity of iced tea by mixing 690 g of hot tea (essentially water) with an equal mass of ice at its melting point.
Assume the mixture has negligible energy exchanges with its environment. (a) If the tea's initial temperature is T; = 96°C, when thermal
equilibrium is reached what are the mixture's temperature T;and (b) the remaining mass m;of ice? If T; = 68°C, when thermal
equilibrium is reached what are (c) T; and (d) m? The specific heat of water is 4186 J/kg-K. The latent heat of fusion is 333 kJ/kg.
(a) Number
i
Units
(b) Number
i
Units
(c) Number
i
Units
(d) Number
i
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning