A student prepared a series of starch standards using the iodine method and measured the absorbance at 590 nm. She obtained the following data.
A student prepared a series of starch standards using the iodine method and measured the absorbance at 590 nm. She obtained the following data.
Chapter26: Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 26.1QAP
Related questions
Question
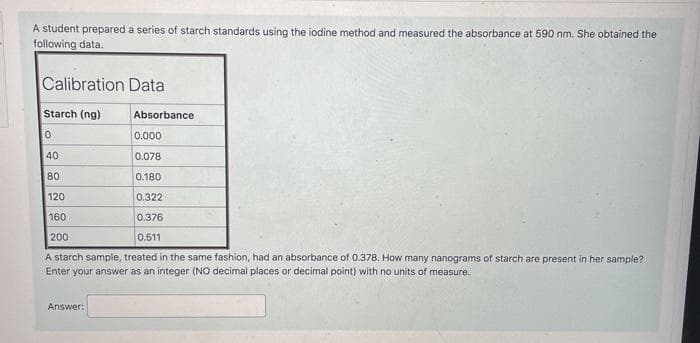
Transcribed Image Text:A student prepared a series of starch standards using the iodine method and measured the absorbance at 590 nm. She obtained the
following data.
Calibration Data
Starch (ng)
Absorbance
0.000
40
0.078
80
0.180
120
0.322
160
0.376
200
0.511
A starch sample, treated in the same fashion, had an absorbance of 0.378. How many nanograms of starch are present in her sample?
Enter your answer as an integer (NO decimal places or decimal point) with no units of measure.
Answer:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

