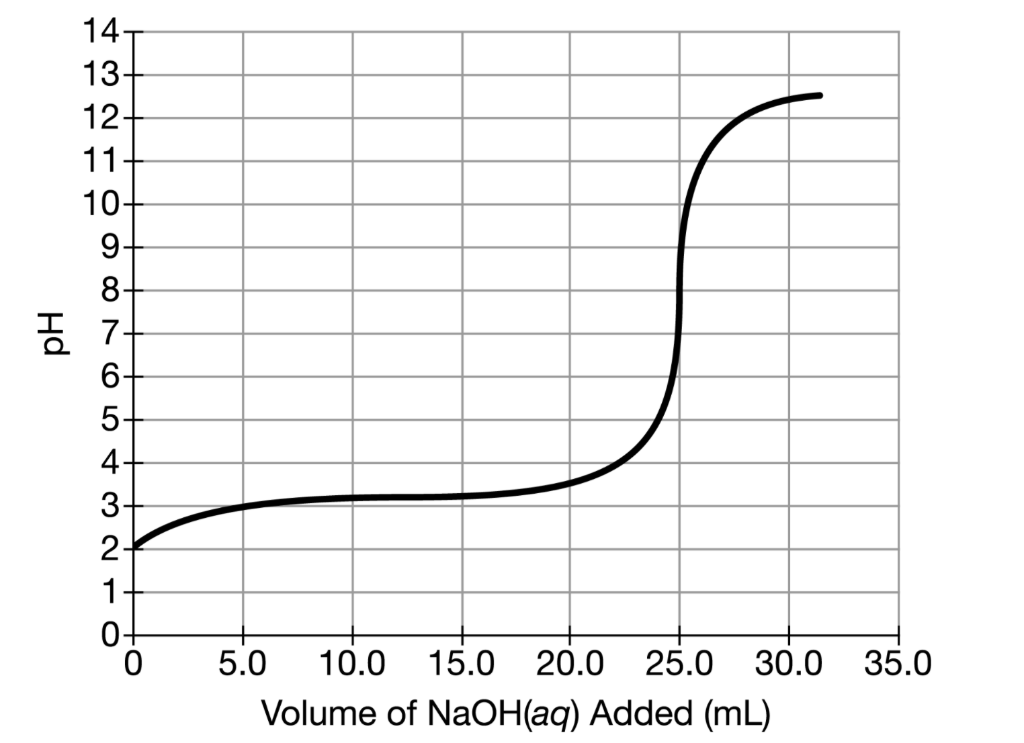
A student titrated 50.0mL of a 0.10M solution of a certain weak acid with NaOH(aq). The results are given in the graph above. What is a pH value between 2.5- 7.5 where the concentration of the weak acid being titrated is less than the concentration of its conjugate base?
A student titrated 50.0mL of a 0.10M solution of a certain weak acid with NaOH(aq). The results are given in the graph above. What is a pH value between 2.5- 7.5 where the concentration of the weak acid being titrated is less than the concentration of its conjugate base?
Chapter9: Energy For Today
Section: Chapter Questions
Problem 9E
Related questions
Question
100%
A student titrated 50.0mL of a 0.10M solution of a certain weak acid with NaOH(aq). The results are given in the graph above.
What is a pH value between 2.5- 7.5 where the concentration of the weak acid being titrated is less than the concentration of its conjugate base?
Transcribed Image Text:14-
13-
12-
11-
10-
9-
8-
5-
3-
2-
1
5.0
10.0
15.0
20.0
25.0
30.0 35.0
Volume of NaOH(aq) Added (mL)
N O 5 ¢32
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning