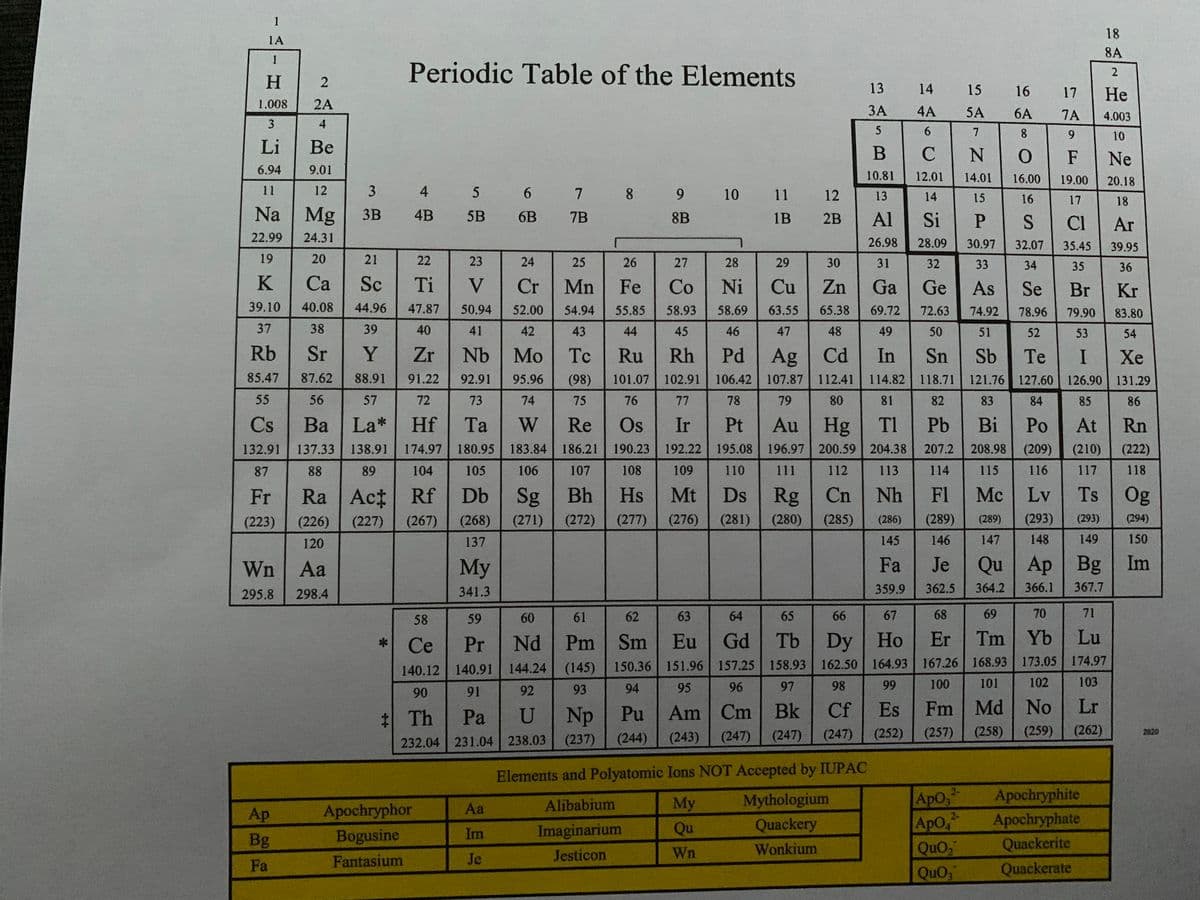
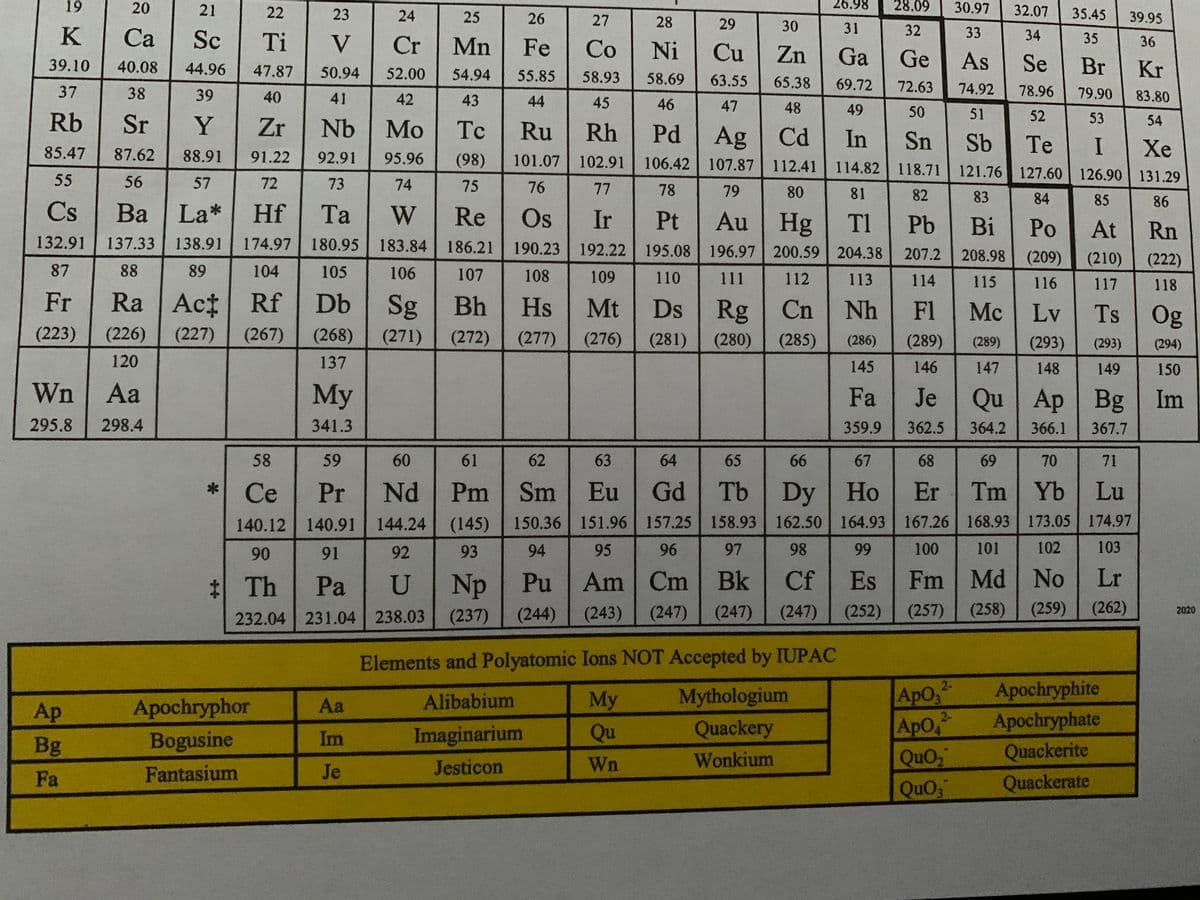
a) Water is a polar solvent and hexane (C6H14) is a nonpolar solvent. In which solvent is WnQuO2 (ionic compound) more likely to be soluble? b) Indicate the compartment that will increase in volume for the following pairs of solutions separated by a semipermeable membrane: 2 M sucrose or 3 M sucrose? c) Calculate the mass of solute needed to prepare 1300 mL of a 7.0 % (m/v) Wn2ApO4 solution.
a) Water is a polar solvent and hexane (C6H14) is a nonpolar solvent. In which solvent is WnQuO2 (ionic compound) more likely to be soluble? b) Indicate the compartment that will increase in volume for the following pairs of solutions separated by a semipermeable membrane: 2 M sucrose or 3 M sucrose? c) Calculate the mass of solute needed to prepare 1300 mL of a 7.0 % (m/v) Wn2ApO4 solution.
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter22: Surfaces
Section: Chapter Questions
Problem 22.6E
Related questions
Question
(Attached in the images is a special periodic table in case you need it)
a) Water is a polar solvent and hexane (C6H14) is a nonpolar solvent. In which solvent is WnQuO2 (ionic compound) more likely to be soluble?
b) Indicate the compartment that will increase in volume for the following pairs of solutions separated by a semipermeable membrane: 2 M sucrose or 3 M sucrose?
c) Calculate the mass of solute needed to prepare 1300 mL of a 7.0 % (m/v) Wn2ApO4 solution.
Transcribed Image Text:1
18
1A
8A
1
Periodic Table of the Elements
H.
13
14
15
16
17
Не
1.008
2A
ЗА
4A
5A
6A
7A
4.003
3
4
6.
7
8.
10
Li
Be
F
Ne
6.94
9.01
10.81
12.01
14.01
16.00
19.00
20.18
11
12
3
4
6.
7
9.
10
11
12
13
14
15
16
17
18
Na Mg
3B
4B
5B
6B
7B
8B
1B
2B
Al
Si
CI
Ar
22.99
24.31
26.98
28.09
30.97
32.07
35.45
39.95
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
K
Ca
Sc
Ti
V
Cr
Mn
Fe
Co
Ni
Cu
Zn
Ga
Ge
As
Se
Br
Kr
39.10
40.08
44.96
47.87
50.94
52.00
54.94
55.85
58.93
58.69
63.55
65.38
69.72
72.63
74.92
78.96
79.90
83.80
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
Rb
Sr
Y
Zr
Nb Mo
Tc
Ru
Rh
Pd
Ag Cd
In
Sn
Sb
Те
I.
Xe
85.47
87.62
88.91
91.22
92.91
95.96
(98)
101.07 102.91
106.42 107.87 112.41
114.82 118.71
121.76 127.60 126.90 131.29
55
56
57
72
73
74
75
76
77
78
79
80
81
82
83
84
85
86
Cs
Ba
Hf
Та
W
Re
Os
Ir
Pt
Au Hg
Tl
Pb
Bi
Ро
At
Rn
132.91
137.33
138.91
174.97
180.95 183.84 186.21
190.23 192.22 195.08 196.97 200.59 204.38
207.2
208.98 (209)
(210)
(222)
87
88
89
104
105
106
107
108
109
110
111
112
113
114
115
116
117
118
Fr
Ra Ac¢
Rf
Db
Sg
Bh
Hs
Mt
Ds
Rg
Cn
Nh
F1
Mc
Lv
Ts
Og
(223)
(226)
(227)
(267)
(268)
(271)
(272)
(277)
(276)
(281)
(280)
(285)
(286)
(289)
(289)
(293)
(293)
(294)
120
137
145
146
147
148
149
150
Wn Aa
My
Fa
Je
Qu Ap
Bg | Im
295.8
298.4
341.3
359.9
362.5
364.2
366.1
367.7
58
59
60
61
62
63
64
65
66
67
68
69
70
71
* Ce
Pr
Nd Pm Sm
Eu
Gd
Tb
Dy Ho
Er
Tm Yb
Lu
(145)
150.36 151.96 157.25 158.93
162.50 164.93 167.26 168.93 173.05 174.97
140.12 140.91
144.24
92
93
94
95
96
97
98
99
100
101
102
103
90
91
Pu
Am Cm Bk
Cf
Es
Fm Md No
Lr
Np
(244)
t Th
Pa
U
(252)
(257)
(258)
(259)
(262)
232.04 231.04 238.03
(237)
(243)
(247)
(247)
(247)
2020
Elements and Polyatomic Ions NOT Accepted by IUPAC
Apochryphite
Apochryphate
ApO,
ApO,
QuO
QuO
Mythologium
Quackery
Aa
Alibabium
My
Apochryphor
Bogusine
Ap
2-
Im
Imaginarium
Qu
Bg
Quackerite
Jesticon
Wn
Wonkium
Fa
Fantasium
Je
Quackerate
8.
Transcribed Image Text:19
20
21
22
23
24
26.98
28.09
30.97
32.07
35.45
39.95
25
26
27
28
29
30
K
Cа
31
32
33
Sc
Ti
V
Cr
34
35
Mn
Fe
Co
Ni
Cu
Zn
Ga
Ge
As
36
39.10
40.08
44.96
47.87
Se
Br
Kr
50.94
52.00
54.94
55.85
58.93
58.69
63.55
65.38
37
38
39
69.72
72.63
74.92
78.96
79.90
40
41
42
43
44
45
46
47
83.80
48
49
50
51
52
Rb
Y
Zr
Nb
Mo
Tc
Ru
Rh
Ag Cd
53
54
In
Sb
Te
Хе
121.76 127.60 126.90 131.29
85.47
87.62
88.91
91.22
92.91
95.96
(98)
101.07
102.91
106.42 107.87 112.41
114.82 118.71
55
56
57
72
73
74
75
76
77
78
79
80
81
82
83
84
85
86
Cs
Ba | La*
Hf
Та
W
Re
Ir
Pt
Au Hg
TI
Pb
Po
At
Rn
132.91
137.33
138.91
180.95 183.84 186.21
190.23
192.22 195.08
196.97 | 200.59 204.38
207.2
208.98
(209)
(210)
(222)
87
88
89
104
105
106
107
108
109
110
111
112
113
114
115
116
117
118
Fr
Ra Act Rf
Db
Bh
Hs
Mt
Ds
Rg
Cn
Lv
Ts
Og
(223)
(226)
(227)
(267)
(268)
(271)
(272)
(277)
(276)
(281)
(280)
(285)
(289)
(286)
(289)
(293)
(293)
(294)
120
137
145
146
147
148
149
150
Wn
Aa
My
Fa
Je
Qu Ap
Bg Im
295.8
298.4
341.3
359.9
362.5
364.2
366.1
367.7
58
59
60
61
62
63
64
65
66
67
68
69
70
71
Dy Ho
150.36 151.96 157.25 158.93 162.50 164.93 167.26 168.93 173.05 174.97
Ce
Pr
Nd
Pm Sm
Eu
Gd
Tb
Er
Tm Yb
Lu
140.12 140.91
144.24
(145)
90
91
92
93
94
95
96
97
98
99
100
101
102
103
# Th
U.
Np
Pu
Am Cm Bk
Cf
Es
Fm Md No
Lr
(243)
(247)
(247)
(247)
(252)
(257)
(258)
(259)
(262)
2020
232.04 231.04 238.03
(237)
(244)
Elements and Polyatomic Ions NOT Accepted by IUPAC
2-
Apochryphite
ApO3
ApO
Ap
Apochryphor
Aa
Alibabium
My
Mythologium
2-
Imaginarium
Qu
Quackery
Apochryphate
Bg
Bogusine
Im
Quackerite
QuO
QuO
Jesticon
Wn
Wonkium
Fa
Fantasium
Je
Quackerate
*-
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning
