a. Outline and describe the reaction scheme of the production of glucose from maltodextrin by using glucoamylase.
Q: Instructions: Type the one letter code using CAPITAL LETTERS ONLY. Do not include spaces between the…
A: Amino acids are the building blocks of protein and they continue the alpha-amino group, carboxyl…
Q: Which of the following is the characteristic that is common to all lipids?* A. They are made up of…
A: The simplest form of lipid is fatty acids which are a long chain of hydrocarbons. A biological…
Q: 2. What amino acids might be found in the binding pocket for this substrate? Please give an example…
A: Binding of a substrate to an enzyme catalytic site brings about conformational changes in the enzyme…
Q: Video resource: Biochemistry of carbohydrates by Armando Hasudungan
A: Carbohydrates are organic molecules arranged in form of aldehyde or ketones with multiple…
Q: 1. In Gel filtration chromatography, when will you stop collecting eluents if sample is not colored?…
A: As you have posted more than one question, we will solve first question for you. I request you to…
Q: 4. the first two reactions in glycolysis associated with unfavorable AG° values, i.e., AGº > 0, both…
A: Glycolysis is a process where glucose is broken down to pyruvate to yield energy. The energy formed…
Q: Which of the following is incorrect about chymotrypsin, trypsin, and elastase? a. They catalyze the…
A: Introduction: Chymotrypsin is a digestive enzyme present in our bodies which is secreted by the…
Q: A glucose 6-phosphate dehydrogenase (G6PD) knockout mouse was created but none of the pups survived.…
A: Glucose-6-phoshate dehydrogenase deficiency is a genetic disorder in which hemolysis of red blood…
Q: VII. Analysis of a peptide antibiotic purified from a strain of Bacillus brevii resulted in the…
A: Differential hydrolysis involve fragmentation in the peptide bond backbone using different…
Q: How is RNA silencing inherited or passed down through generations? What is the nature of RNA…
A: RNA silencing is a gene regulation mechanism that occurs at the transcription level. It can be:…
Q: A chemical reaction is more likely to occur spontaneously if A. the products of the reaction are…
A: For a reaction to occur spontaneously, the free energy change i.e delta G should be negative meaning…
Q: Substituting antisense oligonucleotide non-bridging oxygen atoms with a sulpha atom: Select
A: Ans. 1. Option d is correct The antisense nucleotide non bridging oxygen atom replace by the sulphur…
Q: The Effect of Various Factors on the Rate of an Enzyme Reaction. These experiments will use the…
A: Enzymes are proteins which act as biocatalysts. The activity of enzymes is influenced by factors…
Q: Which of the following is incorrect about the enzyme chymotrypsin? a. It catalyzes the addition of…
A: Chymotrypsin is a serine protease that catalyses the cleavage of peptides on the Carboxy side of the…
Q: How many bypassed reactions does gluconeogenesis have? 10 2 3 6 1
A: The synthesis of glucose from the non-carbohydrate precursors is called gluconeogenesis. Major…
Q: create a flow chart or diagram to illustrate the digestion and absorption of carbohydrates. The…
A: Carbohydrates are composed of carbon, oxygen, and hydrogen which are connected by the glycosidic…
Q: how lactic acid reach the damage muscle.
A: In humans due to intense exercise, the muscle cells lack oxygen for aerobic respiration and hence it…
Q: Give me handwritten answer?
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: Using appropriate diseases as examples, describe three molecular techniques that can be used in…
A: Three molecular techniques involved in disease diagnosis are: 1. Various amplification techniques…
Q: A 100 ml solution of 0.1 M amino acid (AA) at ph 1.0 was titrated with NaOH solution. The pH was…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: How can intact DNA be obtained from a solution of DNA and RNA?
A: The first step towards isolating any nucleic acid is to extract all the nucleic acid, that is both…
Q: Which of the following statements concerning fatty acids is correct? a. Used to make phospholipids…
A: Introduction: Fatty acids consist of a long hydrocarbon chain and a carboxylate group at the end.…
Q: Which properties of a fatty acyl chain cause the Tm to be lower? 1. shorter hydrocarbon chain length…
A: Fatty acids are the simplest form of lipids, it can either be saturated or unsaturated and can be…
Q: Put these concentrations in decreasing order of magnitude (that is the largest concentration first…
A: Given Values: Acrylamide stock conc. = 30% SDS stock = 10% Final concentration in the gel = 12%…
Q: Insulin does not enter the cell that it affects. Instead, it starts a signal cascade within the…
A: Cell signaling is the way of communication of cells. It involves cascades of chemical reactions in…
Q: In gluconeogenesis, what are the precursors of glucose? Select all that apply. lactate amino…
A: Gluconeogenesis is a process in which glucose is synthesized from non-carbohydrate precursors. This…
Q: Protein A interacts with biomolecule B and forms a complex AB, with a dissociation constant KD = 1…
A: Consider the following reaction: P + L ⇌k2k1 PL where P is the protein, L is the ligand and k1 and…
Q: Discuss some of the strategies which have been developed for using nucleic acids as therapeutic…
A: Elucidation of metabolic pathways that are related to a disease combined with the unravelling of the…
Q: Consider the following reaction. CH₂-CH-COO-CH₂-C-Coo- он b Which group of enzymes catalyzes this…
A: The six functional classes of enzymes are hydrolases, oxidoreductases, lyases, transferases, ligases…
Q: A (-) charge in the transition state can be stabilized by a catalyst, which is usually a(n): a.…
A: A negative charge in the transition state can be stabilized by a BASE. Incorrect options- a. anion…
Q: 3. Below is a polypeptide with an unknown number of amino acids. (Standard 4) One the diagram below:…
A: A peptide is polymer of amino acids linked by a peptide bond. A peptide bond is formed due to…
Q: hoose the conditions when PDH is activated. a) acetyl CoA/CoA ratio is high b) NAD+/NADH ratio is…
A: PDH is a regulatory enzyme situated in inner mitrochondrial membrane which is comprise of three…
Q: What mutation can be prevented when we exercise?
A: As our body grows, damages, and repairs with its progression so does our genetic material. The DNA…
Q: Which of the following is incorrect about an enzyme-catalyzed reaction? a. Its progress can be…
A: Because reactant concentrations drop when reactants are transformed into products, reaction speeds…
Q: (b) Why is selectivity of photodynamic very important? How can it be treatment achieved?
A: Photodynamic treatment involves use of molecules called photosensitizers which gets activated in the…
Q: Glycolysis has 3 metabolically irreversible steps that serve as regulatory points. They are ____:…
A: Glycolysis is a process of breakdown of glucose into pyruvate with net release of 2 ATP and 2 NADH.…
Q: You assay 0.5 ml of 1/2000 diluted egg white for protein, as described in the practical schedule,…
A: Standard calibration curves are plotted by taking concentration of the solution in x axis and…
Q: omplex II of electron transport chain is involved in the KREB'S Cycle during the conversion of…
A: Tricarboxylic acid (TCA) cycle or the citric acid cycle is the principle source of energy for cells…
Q: Answer for the number 6 question, Thank you. No need for long explanation.
A: If energy is released in a reaction, these reactions are called exergonic. If energy is consumed in…
Q: Macronutrients provide us the energy to live, move, and function. Evaluate the sentence above,…
A: Bioenergetics refers to the study of living system and it's energy transformations within and among…
Q: Give an example of a typical imaging process for a particular condition and state what radioisotope…
A: Imaging processes involve the use of radioisotopes and can be used for diagnostic purposes. Some of…
Q: 4. How would you summarize the effect of enzyme concentration on enzyme activity?
A: Enzymes are biological catalysts that increase the rate of reactions taking place in the living…
Q: The patient complains about excess sweating, tiredness, bone pain. Her vitamin D levels were…
A: When the body does not get enough nutrients, such as vitamins and minerals, this is referred to as a…
Q: 4 This is a three-part question about a tripeptide. Answer all three sub-questions. The pKR of side…
A: As per the general rule of writing amino acid sequences, the N terminal amino acid is written on the…
Q: Jak Jak 1. Receptors bind cytokines, dimerize, and bind Jaks 2. Jaks phosphorylate each other and…
A: Since you have posted multiple questions with multiple sub-parts, we will solve first three…
Q: In the Lysozyme purification, if the specific activity in the elate was 108 units/mg and the…
A: Egg white is generally rich in lysozyme and hence it is used for its purification. Lysozyme provides…
Q: How much index [18] embalming fluid would you need to mix with 15oz of an index [10] embalming fluid…
A: Embalming fluids are a mixture of formaldehyde, glutaraldehyde, methanol and other solvents. It is…
Q: Consider the structure of the amino acid L-alanine. Which functional groups are common to all amino…
A: All amino acids consist of four moieties - one hydrogen atom (H), one carboxyl group (COOH), one…
Q: For the organism that live there, only the size of a habitat matters, not it’s shape. true or…
A: The composition and arrangement of physical matter at a location is defined as habitat structure. A…
Q: Question Completion Status: 2,0 1,6- 1,2 0,8 0,4 2000- CH₂ 31 CH-COO 260 300 340 400 nm If you…
A: Isocitrate dehydrogenase is an enzyme involved in the TCA cycle. The TCA cycle occurs in the…

Step by step
Solved in 3 steps with 2 images

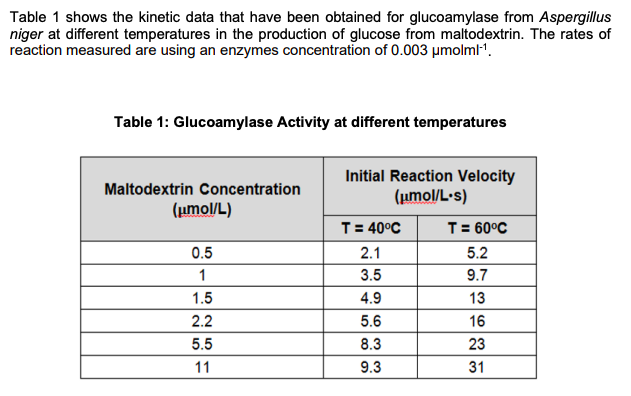
- Table 1 shows the kinetic data that have been obtained for glucoamylase from Aspergillus niger at different temperatures in the production of glucose from maltodextrin. The rates of reaction measured are using an enzymes concentration of 0.003 μmolml-1. Outline and describe the reaction scheme of the production of glucose from maltodextrin by using glucoamylase.Calculate the mass of invertase (in mg) and concentration of invertase (in mM) contained in a 25.0mL sample of yeast extract that has 3,000 total units of activity, assuming that pure invertase has a specific activity of 1,000 units/mg with a mass of 270kD.For the following aspartase reaction in the presence of the inhibitor hydroxymethylaspartate, determine Km and whether the inhibition is competitive or noncompetitive. You have to plot thegraph on the graph paper and also by using excel.[S] V, No Inhibitor V, Inhibitor Present(molarity) (arbitrary units) (same arbitrary units) 1 x 10-4 0.026 0.0105 x 10-4 0.092 0.0401.5 x 10-3 0.136 0.0862.5 x 10-3 0.150 0.1205 x 10-3 0.165 0.142
- Conversion of F1,6BP to GA3P and DHAP by aldolase is striking in that it is strongly unfavorable when all species are at 1 M (∆G°’ = +22.8 kJ/mol) but favorable when the species are at their physiological conditions (∆G = -5.9 kJ/mol). Based on ∆G°’ and ∆G of aldolase and TIM, and using a reasonable physiological value of 15 mM for the concentration of F1,6BP, calculate the concentrations of GA3P and DHAP under physiological conditions (use T = 37°C). Why is the reaction so much more favorable under these conditions than when all species are at 1 M?The standard free energy change for the reaction catalyzed by phosphoglucomutase is -7.1kJ/mol, (a) calculate ΔG at 37°C when the concentration of glucose-1-phosphate is 1-mM and the concentration of glucose-6-phosphate is 25-mM, (b) Is the reaction spontaneous under these conditions?The KM for the reaction of chymotrypsin with N-acetylvaline ethyl ester is 8.8 × 10−2 M, and the KM for the reaction of chymotrypsin with N-acetyltyrosine ethyl ester is 6.6 × 10−4 M. (a) Which substrate has the higher apparent affi nity for the enzyme? (b) Which substrate is likely to give a higher value for Vmax?
- Calculate the standard free-energy change of the reaction catalyzed by theenzyme phosphoglucomutase, given that, starting with 20 mM glucose 1-phosphate and no glucose 6- phosphate, the final equilibrium mixture at 25 °C and pH 7.0 contains 1.0 mM glucose 1-phosphate and 19 mM glucose 6-phosphate. Does the reaction in the direction of glucose 6-phosphate formation proceed with a loss or a gain of free energy?The following initial-rate data were obtained on the rate of binding of glucose w ith the enzyme hexokinase (obtained from yeast) present at a concentration of 1.34 mmol dm-3. What is (a) the order of reaction with respect to glucose. (b) the rate constant?[C6H12O6]/(mmol dm-3) 1.00 1.54 3.12 4.02vo/(mol dm-3 s-1) 5.0 7.6 15.5 20.0The standard reduction potential for ubiquione (A or coenzyme Q) is .045 V, and the standard reduciton potential (E) for FAD is -0.219 V. Using these values, show that the oxidation for FADH2 by ubiquinone theoretically liberates enough energy to drive the synthesis of ATP. Faraday constant =96.48KJ/Vol delta G' standard for ATP Synthesis is +30.5 KJ/mol R=8.314 J/mol K=1.987 cal/mol K
- The enzyme β-methylaspartase catalyzes the deamination of β-methylaspartate. For this aspartate reaction in the presence of the inhibitor hydroxymethylaspartate (3.8 M), determine KM and whether the inhibition is competitive or noncompetitive (KI = 1.0 M). [S], M V w/o inhibitor, M/s V w/ inhibitor, M/s 1x10-4 0.0259 0.0098 5x10-4 0.0917 0.040 1.5x10-3 0.136 0.086 2.5x10-3 0.150 0.120 5x10-3 0.165 0.142 In the ABSENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= __________ (1[S])(1[S]) + __________, and the KM is __________ M. In the PRESENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= ____________ (1[S])(1[S]) + ___________, and the KM is ___________ M. The type of inhibition is ____________. Round-off all answers to two (2) significant figures.The Keq (25C) of the reaction below is 635.67. Fructose 1,6-biphosphate <-->fructose -6-phosphate + Pi. a) What is the standard Gibbs free energy change for this reaction? b) if the concentrationof fructose 1,6 biphosphate is adjusted to 0.85 M and that of fructose 6 phosphate and phosphate adjusted to 0.055 M, what is the actual free energy changeThe enzyme, fumarate, has the following kinetic constants: k 1 k 2 k -1 where k 1 = 10 9 M -1 s -1 k -1 =4.4 x 10 4 s -1 k 2 = 10 3 s -1 a. What is the value of the Michaelis constant for this enzyme? b. At an enzyme concentration of 10 -6 M, what will be the initial rate of product"

