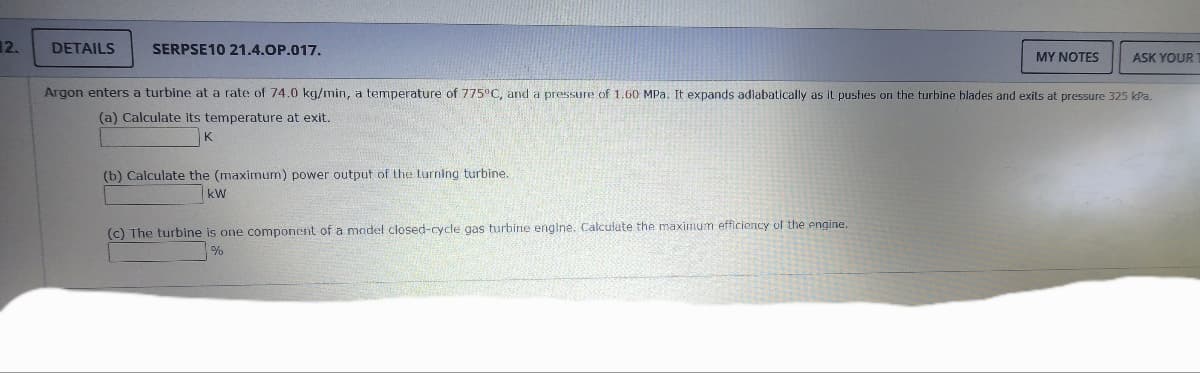
Argon enters a turbine at a rate of 74.0 kg/min, a temperature of 775°C, and a pressure of 1.60 MPa. It expands adiabatically as it pushes on the turbine blades and exits at pressure 325 kPa. (a) Calculate its temperature at exit. (b) Calculate the (maximum) power output of the turning turbine. kW (c) The turbine is one component of a model closed-cycle gas turbine engine. Calculate the maximum efficiency of the engine. %
Argon enters a turbine at a rate of 74.0 kg/min, a temperature of 775°C, and a pressure of 1.60 MPa. It expands adiabatically as it pushes on the turbine blades and exits at pressure 325 kPa. (a) Calculate its temperature at exit. (b) Calculate the (maximum) power output of the turning turbine. kW (c) The turbine is one component of a model closed-cycle gas turbine engine. Calculate the maximum efficiency of the engine. %
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter2: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 2.32E: Many compressed gases come in large,heavy metal cylindersthat are so heavy that they need a special...
Related questions
Question
12.
Transcribed Image Text:12.
DETAILS
SERPSE10 21.4.OP.017.
(b) Calculate the (maximum) power output of the turning turbine.
kW
MY NOTES
Argon enters a turbine at a rate of 74.0 kg/min, a temperature of 775°C, and a pressure of 1.60 MPa. It expands adiabatically as it pushes on the turbine blades and exits at pressure 325 kPa.
(a) Calculate its temperature at exit.
(c) The turbine is one component of a model closed-cycle gas turbine engine. Calculate the maximum efficiency of the engine.
%
ASK YOUR
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,