
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
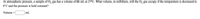
Transcribed Image Text:At atmospheric pressure, a sample of O2 gas has a volume of 61 mL at 27°C. What volume, in milliliters, will the O2 gas occupy if the temperature is decreased to
1°C and the pressure is held constant?
Volume
mL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A sample of Nitrogen gas will occupy a volume of 79.8L at 50.0°C. If the temperature is changed to 135°C, what volume will the gas occupy?arrow_forwardA sample of neon gas occupies a volume of 7.00L at 68.0 degree celcius and 393 torr. If the volume of the gas sample is increased to 9.76 L,while its temperature is increased to 123.0 degree celcius,the resulting gas pressure will be ____torr.arrow_forwardIf a 20.0 L cylinder at 19°C is charged with 5.00 g each of sulfur dioxide (SO2) and oxygen gas, what is the partial pressure of each gas? What is the total pressure?arrow_forward
- 11aarrow_forwardA sample of ammonia (NH3), a colorless gas with a pungent odor, occupies a volume of 8.00 L at a pressure of 655 mm Hg and a temperature of 25°C. What volume, in liters, will this NH3 sample occupy at the same temperature if the pressure is increased to 725 mm Hg? Volumearrow_forwarda gas occupies 15.2 liters at 0.521 atm. At what pressure would the volume be 2.50×103 mL?arrow_forward
- Hydrogen gas is the least dense of all gases. A sample of gas is found to occupy a volume of 1.23 L at 755 mm Hg and 0 degrees celcius. What volume, in liters, will this same gas occupy at 735 mm Hg pressure and a temperature of 5 degrees celcius?arrow_forwardYour laboratory professor opens a bottle of H₂S and a bottle of ether, C4H10O at the same time. Both are gases at the same temperature and pressure. Which smelly gas should you detect first? Molar Masses: C = 12.01, H = 1.01, S = 32.07, O = 16.00. I OH₂S OC₂H₁00 Oneither, since they are more dense than air. Oboth at the same timearrow_forwardA gas has a volume of 15 L at 5 degrees Celsius. What is the final temperature of the gas if its volume increases to 42 Larrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY