Q: How would you convert the following compounds into butanoic acid using reagents from the table? Use…
A: i) Ch3-CH2 - CH2 - Br →Mgldry etherCH3CH2CH2 -MgBr→2H3O+1Co2CH3CH2CH2 - O||C - OH Butanoic acid. 1…
Q: 3. Let the compound below serve as your starting material, compound A: LOH
A: Compounds can be classified as primary, secondary or tertiary based on number of carbon atoms they…
Q: (a) Account for the following :(i) Cl – CH2COOH is a stronger acid than CH3COOH.(ii) Carboxylic…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: 3. Arrange the following in order of increasing reactivity towards a hydrogen halide. Explain. a.…
A: The order of increasing reactivity towards Hydrogen halide :
Q: QUESTION 2 Consider the following alkyl halide compounds. CH3CH2CH(CI)CH2CH3 CH3C(CH3)(CI)CH2CH3…
A: E2 elimination means Elimination bimolecular reaction. In this reaction rate of reaction depends…
Q: 3. Propose a synthesis of the molecule below using A., B., and C. as your carbon atom sources. You…
A: The above organic transformation can be carried out as follows
Q: Develop a separation scheme for the following compounds. Make sure that you end with each compound…
A: The question involves the experimental technique of separation the compounds. we have been given an…
Q: Heating compound X with aqueous formaldehyde forms Y (C17H23NO), which has been converted to a…
A: Y has to be identified and how it is formed has to be explained
Q: 3.1 Show with the aid of reaction mechanisms how the compound given below reacts under: (i)…
A:
Q: (CH3),CHCI AlCl3 CH2 CH3
A: Given,
Q: a. Write the keto-enol tautomerization equation for the following compound OH LOH LOH (a) (b) b.…
A:
Q: a) Para, ortho and meta nitrophenol are constitutional isomers of one another that all vary in the…
A: Nitro phenols are the derivative of benzene having formula OHC6H5-X(NO2)x. There are 3 types of…
Q: Explain each of the following true statements briefly: 1. Hexane (C6H14, a hydrocarbon) is not a…
A: Nucleophile is a rich electron species that contains a lone pair of electrons and can donate to a…
Q: 3) The alcohol Compaund grven below is used perfume moling. Demonstrate benzene the synthesis of t8s…
A: Given: Reactants given are bromobenzene and butene. To find: To synthesize a compound which is an…
Q: 2. Each of the circled hydrogens in the compound shown below could potentially be removed by…
A: Here we have to predict which of the following bases will deprotonate the following indicated acidic…
Q: How many carbonyl groups are generated upon treatment of the molecule below with ozone, followed by…
A: We have given the organic compound and find out How many carbonyl groups are generated upon…
Q: Give the major product: NABH4 3 H20, H+ Cl2, OH- phenylhydrazine 5 2 moles CH3CH2OH H+ OH- -H20 7 H.…
A:
Q: 2 of 15 Describe the synthesis to make the following product from (bromomethyl)cyclopentane. You can…
A:
Q: Chlorine is a(n) (CIRCLE one) ortho-para/meta director. Explain your answer.
A: Please find your solution below : Chlorine has dual nature when it is attached to benzene ring. It…
Q: Chemistry Question 2. Compound A is heated with acrolein and base to gives a product as shown below.…
A:
Q: - Which organic product is most likely to be present following the reaction between excess chlorine…
A: Reaction will be- CH4+Cl2→CH3Cl+HCl But in presence of excess of chlorine then reaction will not…
Q: excess CH3B then NaOH, A N.
A: This is an excessive alkylation then Hofmann elimination reaction in which Hofmann alkene product is…
Q: a) Sketch all the steps required for the following synthesis. Compound H b) Write the IUPAC name for…
A: GIVEN:-
Q: Reagents A. Cu(SO4)2, NaHCO3, Na-citrate B. concentrated HCI, orcinol, FeCl3 C. Alkyl chloride,…
A: Here we have to differentiate the following given two carbohydrates by chemical test.
Q: 2. Provide mechanistic explanations for the following observations (a) HCI CI (b) CI HCI
A:
Q: Suggest a synthesis for the molecule shown below. Only starting materials containing six (6) carbons…
A: The key reactions involved in the synthesis of given target material are: 1) Friedel-Crafts…
Q: CH3 F CH3 B 0 10 ОН
A:
Q: Can you please help me figure out this problem #5. I believe it is asking to use a reagent of…
A:
Q: 7. Based on the reaction scheme above: (a) Draw the structural formula for compound A to H.
A: benzene is aromatic compound . Benzene involve in electrophilic substitution reactions .(benzene is…
Q: In the following three compounds(1,2,3) arra
A: Benzene is having pi-electron excess, hence it undergo electrophilic substitution reactions ,…
Q: Be sure to answer all parts. An allylic alcohol contains an OH group on a carbon atom adjacent to a…
A: We know primary carbocation is less stable than secondary carbocation. So if a primary carbocation…
Q: [1] LDA [2] CH3I
A:
Q: 2. Consider the two cyclic molecules below A. В. a. The CH2 hydrogens of molecule A. and B. show…
A:
Q: CH3 НО KMNO4 OH- H+ TOULENE BENZOIC ACID
A:
Q: To practice working through the early parts of a multistep synthesis, devise syntheses of…
A: The reaction of Grignard reagent with aldehyde followed by hydrolysis form alcohol. The reaction…
Q: Provide an arrow pushing mechanism for the following reaction. Include protons and each step clearly…
A:
Q: Draw the structure of the compound you would expect from Sy2 reaction of the molecule below with…
A: Ans
Q: A dilute solution of 6-phenylhexanoylchloride in CS, (carbon disulfide, a solvent) was added slowly…
A: Friedel Craft reaction is an electrophilic substitution reaction. It may have alkyl halide (in…
Q: A retrosynthetic analysis of 1 was undertaken suggesting a forward synthesis starting from compounds…
A: We have to synthesize organic compound 1 :
Q: 10. Provide unambiguous structural formulas for reaction products A, B, and C.
A: Given,
Q: Chemistry 3. Propose a synthesis of the molecule below using A., B., and C. as your carbon atom…
A: Here we are required to synthesize the given compound.
Q: Provide the reagents necessary to carry out the following conversion OH OH A 1) HCN; 2) PCC B 1)…
A:
Q: 2. Consider the following scheme. NH2 NH2 Reagent A Но ÓH Compound T HO Compound S KOH, NH,NH2…
A: a) The IUPAC name of the given compound T is, 1-amino-1-(4-hydroxy phenyl) propan-2-one b)…
Q: Question attached
A: Synthesis forward reaction and listing reagents and steps for the given reaction,
Q: Br 1. Pd°Ln, EtN, MECN, Me Me 2. MCPBA, CH2Cl2 3. HCIO, EIOH provide step by step analysis for each…
A: 1. a. Heck coupling. Epoxidation. Acid catalyzed epoxide opening with ethanol. (epoxide is opened at…
Q: 3. Propose a synthesis of the molecule below using A., B., and C. as your carbon atom sources. You…
A:
Q: 1. Draw 3 isomers of C7H12O2 then state where you will dispose it (the kind of waste disposal should…
A:
Q: 1. The following diagram shows the partial synthesis of an important antihistamine drug currently…
A: To write the oxidation state of *C in all the three structures.
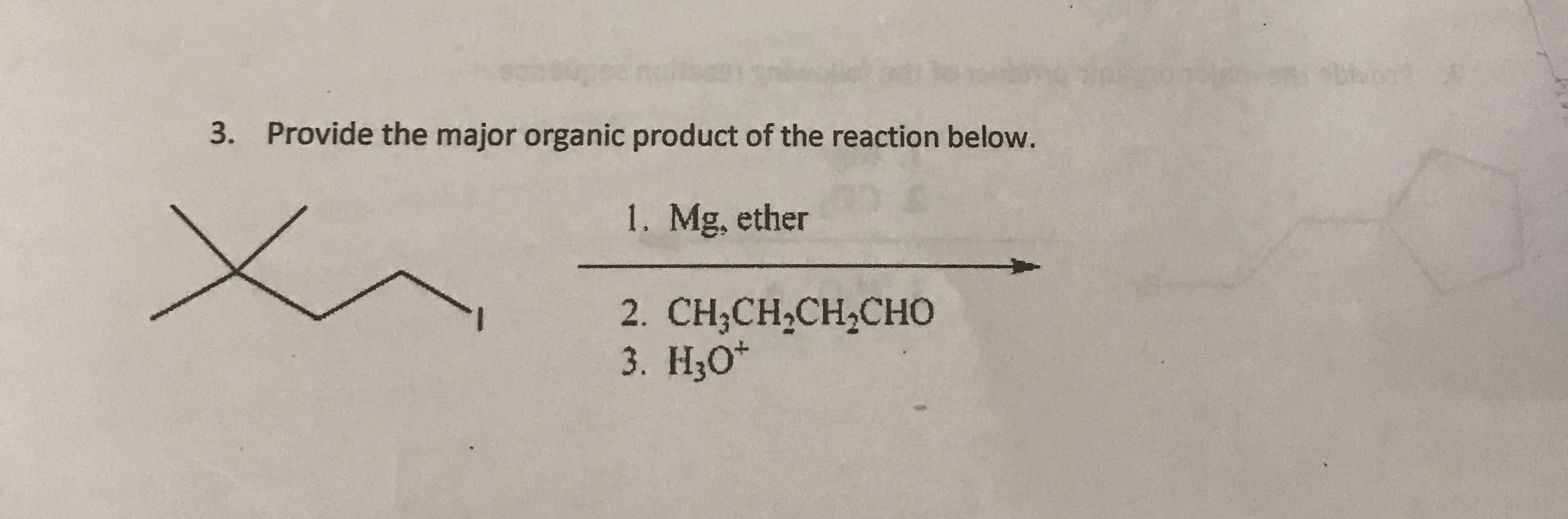
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

- determine the limiting reaction in the following sn2 reaction given 2.00mmol of sodium saccharin and 0.16mL of iodoethane. yielding product of 0.151g N-ethylsaccharinFor 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is usedPlease help with the following Organic Chemistry reaction problem. Thank you.
- an alkene when acting with HCl shows a weight increase of 52.14% .What is the molecular formula of the alkene?Calculate the % yield-show work in a clear organized matter-be aware of significant figures. Which is the limiting reagent? A Freidel – Crafts Alkylation Name_______________ Data: Alkyl chloride used________tert-butyl chloride___________________________________ Mass alkyl chloride____15.7549 g_______________________________________ Mass 1,4-dimethoxybenzene_____5.0035 g_______________________________ Mass product isolated____2.7834 g______________________________________ Appearance of product__________dry snow white solid________________________________ m.p. rextal product__101 deg Celsius ____________________________________________ Published m.p. product____102-104 o C_____________ source____Chemspider________________ Name of product__1,4-di-tert-butyl-2,5-dimethoxybenzene._______________________________________________4) Why might a student obtain a percent yield more than 100% for this preparation?
- 1pentanol + NaBr/H2SO4 -> 1-bromopentane Limiting reagent: 1-Pentanol Percent Yield: 64.5% The typical yield for this reaction is about 80%. Compare this to the yield attained..What issues might account for the yield being lower than expected?Assuming a 56.8% yield, how kany ml of alkene is required to produce 22.5 mL of 2,3-dimethyl-2-butanol? MW of alkene: 84.16, d: 0.653 MW of 2,3-dimethy-2-butanol: 102.17, d:0.823Cromolyn sodium, developed in the 1960s, has been used to prevent allergic reactions primarily affecting the lungs, as, for example, exercise-induced emphysema. It is thought to block the release of histamine, which prevents the sequence of events leading to swelling, itching, and constriction of bronchial tubes. Cromolyn sodium is synthesized in the following series of steps. Treatment of one mole of epichlorohydrin with two moles of 2,6-dihydroxyacetophenone in the presence of base gives I. Treatment of I with two moles of diethyl oxalate in the presence of sodium ethoxide gives a diester II. Saponification of the diester with aqueous NaOH gives cromolyn sodium. Q.Propose a mechanism for the formation of compound I.
- Cromolyn sodium, developed in the 1960s, has been used to prevent allergic reactions primarily affecting the lungs, as, for example, exercise-induced emphysema. It is thought to block the release of histamine, which prevents the sequence of events leading to swelling, itching, and constriction of bronchial tubes. Cromolyn sodium is synthesized in the following series of steps. Treatment of one mole of epichlorohydrin with two moles of 2,6-dihydroxyacetophenone in the presence of base gives I. Treatment of I with two moles of diethyl oxalate in the presence of sodium ethoxide gives a diester II. Saponification of the diester with aqueous NaOH gives cromolyn sodium. Q.Propose a structural formula for compound II and a mechanism for its formation.Cromolyn sodium, developed in the 1960s, has been used to prevent allergic reactions primarily affecting the lungs, as, for example, exercise-induced emphysema. It is thought to block the release of histamine, which prevents the sequence of events leading to swelling, itching, and constriction of bronchial tubes. Cromolyn sodium is synthesized in the following series of steps. Treatment of one mole of epichlorohydrin with two moles of 2,6-dihydroxyacetophenone in the presence of base gives I. Treatment of I with two moles of diethyl oxalate in the presence of sodium ethoxide gives a diester II. Saponification of the diester with aqueous NaOH gives cromolyn sodium. Q. Is cromolyn sodium chiral? If so, which of the possible stereoisomers are formed in this synthesis?What is the limiting reagent in the reaction below?



