band marked with * scored positive for this activity. pH 9 TH pH 3 Decreasing pH )) If you perform the ion exchange chromatography at pH = 7.5, would you want to use an anion or cation exchanger? anion exchanger cation exchanger
band marked with * scored positive for this activity. pH 9 TH pH 3 Decreasing pH )) If you perform the ion exchange chromatography at pH = 7.5, would you want to use an anion or cation exchanger? anion exchanger cation exchanger
Biochemistry
9th Edition
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Chapter1: Biochemistry: An Evolving Science
Section: Chapter Questions
Problem 1P
Related questions
Question
ch2
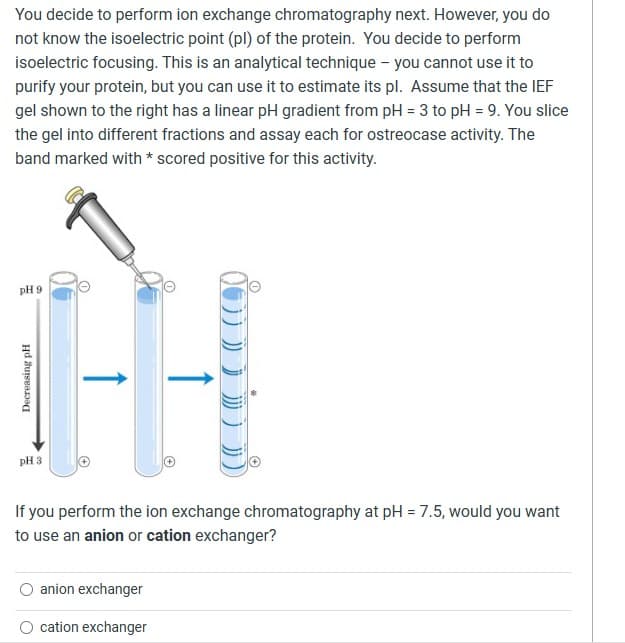
Transcribed Image Text:You decide to perform ion exchange chromatography next. However, you do
not know the isoelectric point (pl) of the protein. You decide to perform
isoelectric focusing. This is an analytical technique - you cannot use it to
purify your protein, but you can use it to estimate its pl. Assume that the IEF
gel shown to the right has a linear pH gradient from pH = 3 to pH = 9. You slice
the gel into different fractions and assay each for ostreocase activity. The
band marked with * scored positive for this activity.
pH 9
Decreasing pH
pH 3
If you perform the ion exchange chromatography at pH = 7.5, would you want
to use an anion or cation exchanger?
anion exchanger
cation exchanger
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Fundamentals of General, Organic, and Biological …
Biochemistry
ISBN:
9780134015187
Author:
John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:
PEARSON