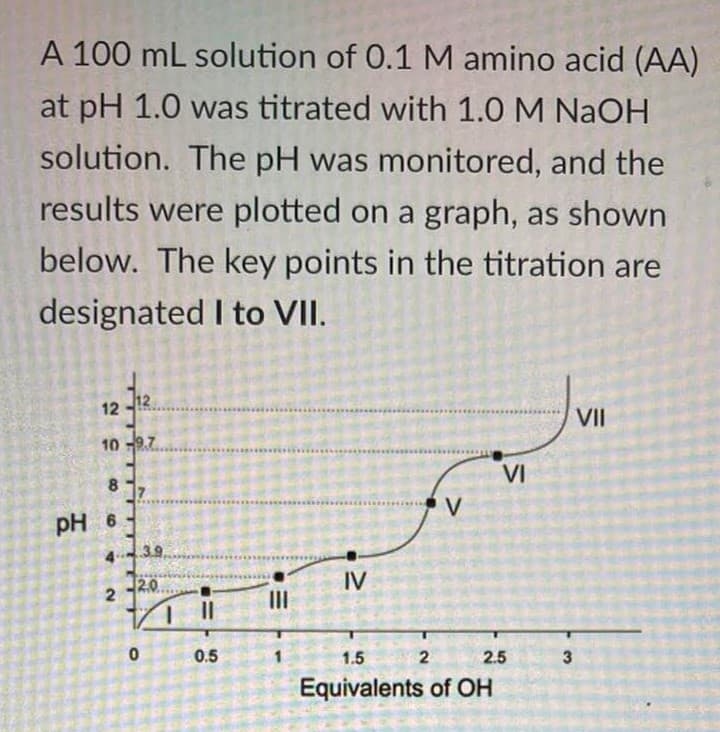
Based on the given figure, a. where is the region or point where AA is predominantly present as a (-2) charged species b. where is the effective buffering range for the amino acid on the acidic region
Q: Suggest a reason why amino acids are usually more soluble at pH extremes than they are at neutral…
A: Introduction: Amino acids are a group of organic compounds containing two functional groups- amino…
Q: In a hospital laboratory, a 10.0 mL sample of gastric juice, obtained several hours after a meal,…
A: Introduction: pH is an indicator that is used to check the acidity or basicity of the solution. It…
Q: The Bohr effect refers to: a. the decrease in affinity of Hb for O2 when the pH goes down b.…
A: The hemoglobin has an affinity towards oxygen which makes it capable to carry oxygen throughout the…
Q: Calculate the salt to acid ratio ([Aˉ]/[HA]) of a solution of an amino acid at pH 2 and pKa of…
A: The buffer solution is any solution that is a mixture of a weak acid and conjugate base or…
Q: Explain why the negative charges incorporated in the filtering membrane are able to reject negative…
A: Introduce yourself: Protein is essential to the construction and function of the organism.
Q: Based on the pka values of the amino acids, is there any amino acid that could serve as a buffer at…
A: Amino acids are chemical molecules with amino and carboxyl functional groups as well as a side chain…
Q: The graph given below shows the results of an experiment in which the UV absorbance (A280) of a…
A: Proteins present in a solution absorb ultraviolet light having an absorbance maxima of 280 and 200…
Q: If several like-charged proteins are bound simultaneously to an ion exchange column, they can be…
A: Ion-exchange chromatography is a protein purification technique that can separate proteins based on…
Q: V-A. Which of the following amino acids will elute first in a cation-exchange column using a buffer…
A: Ion Chromatography Separates ions and polar molecules on the basics of their affinity to the ion…
Q: The pH 6.0 buffer solution had resulted in a higher logD value than the pH 7.0 buffer solution and…
A: The pH 6.0 buffer solution showed higher logD value than buffer solution having pH 7.0 and pH 8.0…
Q: The simple form of |Hoff equation is: Il = [B]RT In this equation the [B] is the molar concentration…
A: Given Values: The final equation of the Hoff equation is changed to this form which is as follows:…
Q: V-A. Which of the following amino acids will elute first in a cation-exchange column using a buffer…
A: Cation exchange column separate iond and molecules based on their net overall surface charge.
Q: What determines when an amino acid ionized? i.e. Why are both Lysine (pKa 10.53) and Argenine (pKa…
A: Amino acid are the building blocks of the protein. Each amino acid has a common basic structure with…
Q: You can elute the bound proteins from the Diethylaminoethyl (DEAE) sepharose column by A. increasing…
A: Diethylaminoethyl (DEAE) sepharose is a positively charged resin used in ion-exchange…
Q: What generates the peak at 410 nm? a. The heme group of cytochrome c b. The heme group of bovine…
A: Absorbance is the optical density i.e. the quantity of light absorbed by the solution which is…
Q: The Bohr effect refers to the decrease in affinity of Hb for 02 when the pH goes up. OA. the…
A: Bohr's effect shows when there is an increase in partial pressure of CO2 (PCO2), it shifts the O2…
Q: when preparing a buffer solution for a biological experiment, one should choose a) a weak acid and…
A: Buffer solutions often are weak acid/strong acid and conjugate base. buffers resist the change…
Q: Based on the Hill Plot below, this protein-ligand interaction displays log Y 1-Y 2.5 0 -2.5 -2.5…
A: Hill equation developed by Archibald Hill in 1910 quantitatively describing cooperative ligand…
Q: A mixture containing glutamic acid, arginine, phenylalanine and valine was subjected to anion…
A: Amino acids differ from each other on the basis of their side-chain properties. There are twenty…
Q: The commercial hydrogenation of vegetable oil often leads to a trans acid. Explain this statement.
A: Fat hydrogenation is the process of combining fat typically vegetable oils with hydrogen in order to…
Q: Given a tripeptide Cys-His-Lys, Cys: Pk: = 1.71; Pk2= 10.78; Pke= 8.33 His: Pk = 1.82; Pkz= 9.17;…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: Consider a negatively charged protein adsorbed on anion-exchange gel at pH 8. (a) How will a…
A: Proteins are essential nutrients for the human body. They are one f the building blocks of body…
Q: Propionic acid with a pk of 4.88 is a weak acid. Which situation is likely to occur in a propionic…
A: Introduction: The ionization constant (pKa) is a value that tells us the tendency of compounds or…
Q: Suggest a reason why amino acids are usually more soluble at pH extremes than they are at neutral…
A: There are about twenty essential and nonessential amino acids are often present in proteins. Amino…
Q: The order of elution of amino acids X, Y, VW and Z in a cation exchanger eluted with a buffer of…
A: Ion exchange chromatography is used to separate molecules based on the charge. In the ion exchange…
Q: Consider a HCI solution with a pH of 4.70. a) Calculate the concentration of HCl. b) Identify the…
A: pH is the scale which is used to specify the acidity or basicity of an aqueous solution. Acidic…
Q: A conjugate acid-base paira. acts as a buffer.b. can combine with H+ in a solution.c. can release H+…
A: A buffer solution is an aqueous solution consisting of a mixture of a weak acid and its conjugate…
Q: What can be deduced from the following information? % N- C- Protein Nonpolar polar acidic basic term…
A: Polar aminoacids are more. Hence hydrophilicity is more. Globular proteins are folded like…
Q: QUESTION: What are the buffer regions/effective pH range?
A: Buffer solution is a solution which pH does not varies on dilution or addition of small amount of…
Q: The salt of diclofenac, a nonsteroidal anti-inflammatory drug (NSAID), has a chemical formula…
A: Diclofenac is a non-steroidal anti-inflammatory, an analgesic drug used to relieve pain and swelling…
Q: Sketch a plot that shows a typical binding curve for the interactions between a ligand, L (in terms…
A: The interactions that lie between a protein and a ligand is classified as reversible as well as…
Q: B) An aqueous solution (pH ~7) containing mixture of three amino acids; glycine (G), arginine (R)…
A: At pH=7, Glycine is uncharged. Arginine is positively charged. Glutamic acid is negatively…
Q: Given a tripeptide Cys-His-Lys, Cys: Pk1 = 1.5; Pk2 = 10.8; PkR = 8.5 His: Pk1 = 1.6; Pk2 = 9.0; PkR…
A: Hi. Thank you for the question, As per the honor code, we are allowed to answer three sub-parts at a…
Q: n case of the same amino acid as in the picture, in which ionization state would this amino acid be…
A: Amino acid given in picture is tyrosine. It is aromatic amino acid having uncharged polar…
Q: Which factors will cause protein denaturation? Group of answer choices a. aqueous phosphate buffer…
A: Proteins are synthesized by linking amino acids via amide or peptide bonds. After the synthesis of…
Q: Enter an estimate for the charge on the peptide KPIVRFDEESC at pH 8.0.
A: The polypeptide is considered as the long chain of amino acid, which are joined by the peptide bond.…
Q: When the pH is acidic, the concentration of H+ is high and functional group(s) [ Select ] will be…
A: Amino acids is Organic compound they have both acidic and basic functional groups that is why this…
Q: Based on how the water molecules are surrounding the two ions (labeled A&B), you can tell that…
A: Water Water is considered to be a polar molecule. Water is liquid at condition of standard…
Q: or the following amino acids: Isoleucine and Tyrosine a. Draw its complete protonic equilibria.…
A: Hi, thank you for the question. As per the honor code, we are allowed to answer three sub-parts at a…
Q: An import relationship that allows us to determine the pKa of a weak acid using a pH titration curve…
A: The Henderson-Hasselbalch equation gives the relation between pH and pKa. pH defines the…
Q: Diethylaminoethyl cellulose is a positively charged resin used in ion-exchange chromatography with a…
A: Diethylaminoethyl cellulose (DEAE-C) in chromatography is a positively charged resin. It is used in…
Q: Draw the structural form of valine that predominates in solution at each of the following ph values:…
A: Amino acids are biomolecules that form the protein. They also act as precursor molecules in the…
Q: V-A. Which of the following amino acids will elute first in a cation-exchange column using a buffer…
A: Ion exchange chromatography is a technique which is used to separate the protein on the basis of…
Q: What are the assumptions of the Hill equation? A. The total number of binding sites will be filled.…
A: The answer is (e) none of the above .
Q: Based on our pka's table, pKa1 of -COOH group of Cys = 1.71 pKa2 of -NH2 group of Cys = 10.78…
A: pKa is defined as the number which indicates the acidity of the particular molecule. It shows the…
Q: Which of these peptides contain an amino acid with a side chain IMIDAZOLE? A. Oxytocin B.…
A: The amino acid that contain IMIDAZOLE side chain is HISTIDINE. Neither oxytocin nor vasopressin…
Q: Assume that pKa(1) = 2.0 and that pKa(2) = 9.0. Suppose that the total concentration of the amino…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: To analyze a sample by mass spectrometry, the sample must consist of gas phase ions. a) Explain why…
A: Introduction Proteins are complex macromolecules that are active in an aqueous solution as water…
Based on the given figure,
a. where is the region or point where AA is predominantly present as a (-2) charged species
b. where is the effective buffering range for the amino acid on the acidic region
Step by step
Solved in 4 steps with 2 images

- Table 2. Volume of BSA, protein content, and absorbance readings of reference solutions Solution Volume of BSA standard solution (μL) Protein content(μg/mL) Absorbance value At 595 nm 1 0 0 0 2 10 1 0.022 3 30 3 0.065 4 50 5 0.106 5 70 7 0.178 6 100 10 0.299 7 120 12 0.380 Make a graph by plotting the absorbance values versus the BSA protein content (in μg) for theseven reference solutions. When constructing the graph, be…1.2. Using DEAE-cellulose as ion exchange resin, indicate the starting and ending pH for the narrowest experimental pH range used to separate an amino acid mixture consisting of Cys, His and Leu Starting pH: _____ Ending pH: _____pH 3.5 4.5 5.5 6.5 7.5 8.5 Absorbance 0.098 0.027 0.068 0.028 0.032 0.054 Concentration in diluted supernatant (mg/ml) 0.196 0.054 0.136 0.0056 0.0064 0.0108 Concentration in undiluted supernatant (mg/ml) 0.98 0.27 0.68 0.28 0.32 0.54 Formula for the amount (g) of soluble proteins in the soy flour extract: In 15 ml of soy flour extract (with 1/50 dilution), Soluble protein (g) = (C x V x F) / 1000 C = concentration (mg/ml); F = dilution factor; V = volume of solution (ml) Calculate the % solubility of protein in the soy flour (Assume the soy flour contains 35% protein (w/w))
- pH 3.5 4.5 5.5 6.5 7.5 8.5 Absorbance 0.098 0.027 0.068 0.028 0.032 0.054 Concentration in diluted supernatant (mg/ml) 0.196 0.054 0.136 0.0056 0.0064 0.0108 Concentration in undiluted supernatant (mg/ml) 0.98 0.27 0.68 0.28 0.32 0.54 Formula for the amount (g) of soluble proteins in the soy flour extract: In 15 ml of soy flour extract (with 1/50 dilution), Soluble protein (g) = (C x V x F) / 1000 C = concentration (mg/ml); F = dilution factor; V = volume of solution (ml) Calculate the amount of soluble protein (in grams) in the soy flour extractThe pH of a 0.0082 M solution of HNO₃ isThe prescriber ordered oxacillin 500mg IM q6h. The instructions on the 2g vial state to reconstitute the powder with "11.5mL of sterile water for injection, yielding 250mg/1.5mL" Whatis the strength of the reconstituted solution? How many milliliters would you administer?
- TE buffer consists of 10 mM of Tris-Cl, pH 7.6 and 1mM of EDTA, pH 8.0. You need to prepare 3L of TE buffer and you have the following stock solutions: 500 mM Tris-Cl and 1 M EDTA. How will you prepare the 3L of TE? Write work clearlyYou are supplied with the following: NaCl (Mr = 58.443 g/mol) 2.5 M Tris-Cl, pH 8 oplossing / solution (1 Litre) EDTA, sodium salt (Mr = 380.2 g/mol) 10 % Sodium dodecyl sulfate solution Proteinase K solution (50 mg dissolved in 1 ml ddH2O)You need a digestion buffer consisting of the following: 15 mM NaCl 75 mM Tris-Cl, pH 8 16 mM EDTA, pH 8 0.8 % Sodium dodecyl sulfate 0.75 mg/ml proteinase KCalculate: How will you prepare 500 ml of the digestion buffer?3. a) An aqueous solution is 3.50% by mass potassium bromide, KBr, and has a density of 1.02 g/mL. The molality of potassium bromide in the solution is _________m. b)An aqueous solution of magnesium nitrate has a concentration of 0.474 molal. The percent by mass of magnesium nitrate in the solution is _________%. c)An aqueous solution of cobalt(II) bromide has a concentration of 0.153 molal. The percent by mass of cobalt(II) bromide in the solution is _________%.
- Assuming equal concentrations of conjugate base and acid, which one of the following mixtures is suitable for making a buffer solution with an optimum pH of 7.4–7.6? NaOCl / HOCl (K a = 3.2 × 10 –8) NH 3 / NH 4Cl (K a = 5.6 × 10 –10) NaNO 2 / HNO 2 (K a = 4.5 × 10 –4) NaCl / HCl CH 3COONa / CH 3COOH (K a = 1.8 × 10 –5)How many moles of KBr are there in 89.5 mL of 0.150 M KBr?If you added three proteins (Protein A, pI 5.0; Protein B, pI 6.0; Protein C, pI 7.0) to an anion exchange column equilibrated with bicine buffer, pH 8.5, in what order would these protein be expected to elute as the column was loaded, washed, and then increasing amounts of NaCl were added?

