boiling point B 6.0 x 10-3 Gas - Triple point (T;) A 00.0098 100 374.4 Temperature (°C) B. Give the number of degrees of freedom and identify the phase(s) present in the following: 1. АВ : F- 2. ВС : F%3 3. BD : F B : P = 5. Е : F: 6. F :F = 7. G : F= I| || || || || |||| P P P PPPA ||
boiling point B 6.0 x 10-3 Gas - Triple point (T;) A 00.0098 100 374.4 Temperature (°C) B. Give the number of degrees of freedom and identify the phase(s) present in the following: 1. АВ : F- 2. ВС : F%3 3. BD : F B : P = 5. Е : F: 6. F :F = 7. G : F= I| || || || || |||| P P P PPPA ||
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.58E: Use the phase diagram of water in Figure 6.6 and count the total number of phase transitions that...
Related questions
Question
100%
Unit 4 Act. 1-4 B
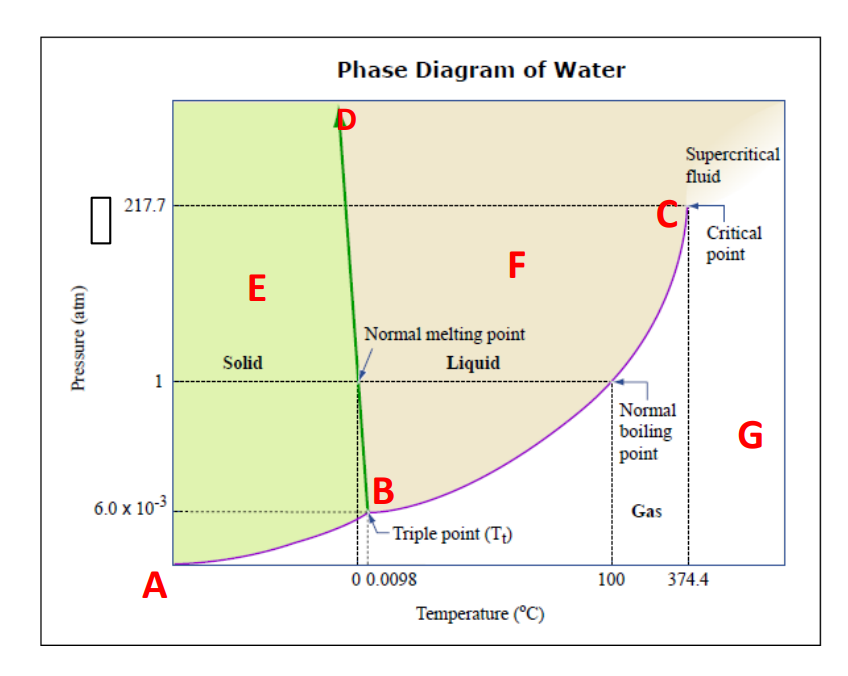
Transcribed Image Text:Phase Diagram of Water
Supercritical
fluid
217.7
Critical
point
F
E
Normal melting point
Solid
Liquid
1
Normal
boiling
point
G
B
6.0 x 10-3
Gas
- Triple point (T;)
A
0 0.0098
100
374.4
Temperature (°C)
Pressure (atm)

Transcribed Image Text:boiling
point
B
6.0 x 10-3
Gas
Triple point (T;)
A
0 0.0098
100
374.4
Temperature (°C)
B. Give the number of degrees of freedom and identify the phase(s) present in the
following:
1. AB : F=
P :
2. BC : F =
3. BD : F =
P =
P
4. В
: F:
5. E
: F
P
6. F
: F
P :
7. G : F=
P =
I| || || | || || ||
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax