C. Standardization Weight of vial and KHPH 59.09 Weight after 1st transfer 58.2 Weight after 2nd transfer 56-5 Weight after 3rd transfer 55.6 Weight of KHPh in flask I (show calculations) 50-9-58-2 0.89 Weight of KHPh in flask II (show calculations) 1-79 53.2-56.5 Weight of KHPh in flask III (show calculations) 0.99 56- 5-55-6 I II III Initial volume reading of NaOH O-00 ml 27.00 51-00 Final volume reading of NaOH as. 00 ml SI-00 80-00 25-00 ml 24.00 ml 29.00ml Volume of NaOH used Molarity of NaOH 0.156.M 0.347M 0 152M Trial 3: KHPh in flosk III = 0-4 Show calculations for all Trials Below: Voume afNeOH used: 29 ml=0C
C. Standardization Weight of vial and KHPH 59.09 Weight after 1st transfer 58.2 Weight after 2nd transfer 56-5 Weight after 3rd transfer 55.6 Weight of KHPh in flask I (show calculations) 50-9-58-2 0.89 Weight of KHPh in flask II (show calculations) 1-79 53.2-56.5 Weight of KHPh in flask III (show calculations) 0.99 56- 5-55-6 I II III Initial volume reading of NaOH O-00 ml 27.00 51-00 Final volume reading of NaOH as. 00 ml SI-00 80-00 25-00 ml 24.00 ml 29.00ml Volume of NaOH used Molarity of NaOH 0.156.M 0.347M 0 152M Trial 3: KHPh in flosk III = 0-4 Show calculations for all Trials Below: Voume afNeOH used: 29 ml=0C
Chapter1: Excel Basics
Section: Chapter Questions
Problem 2P
Related questions
Question
Can someone please check my work
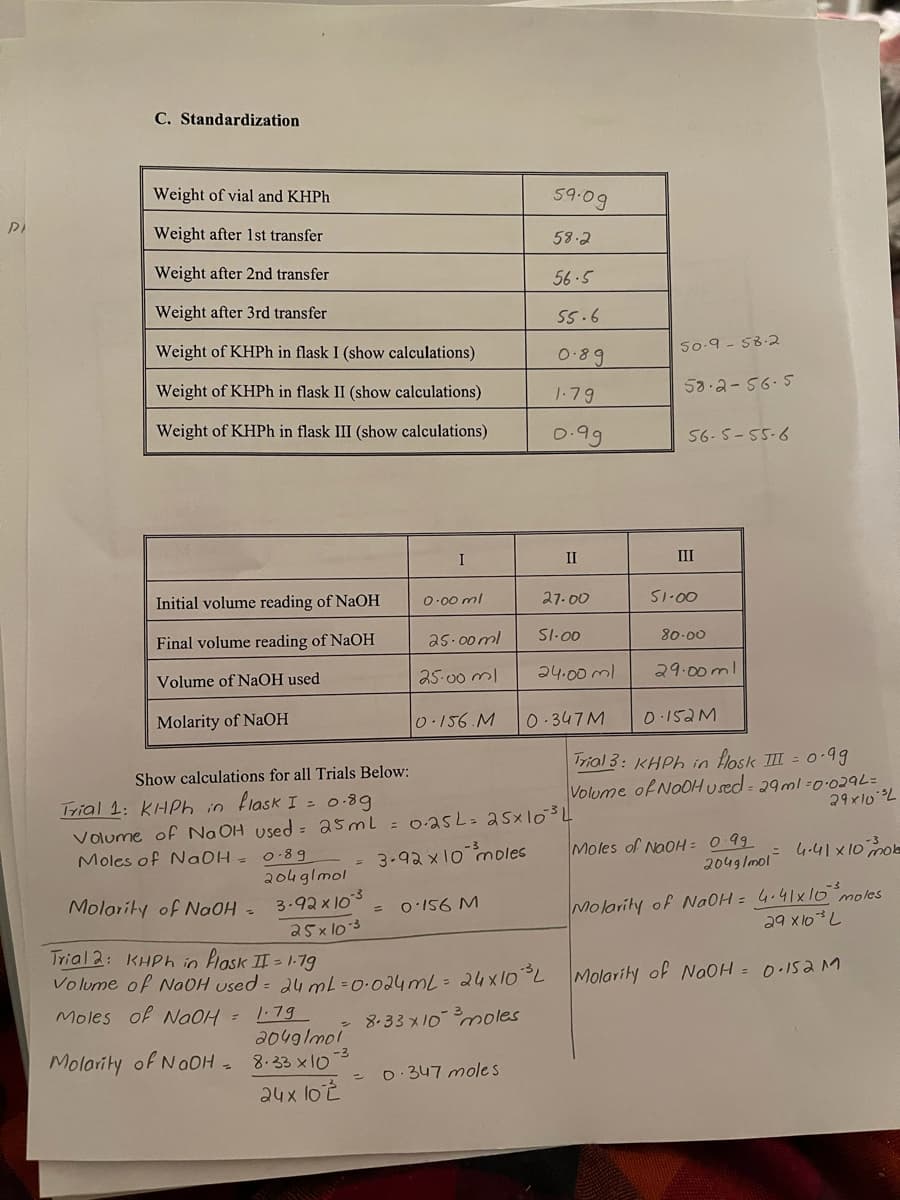
Transcribed Image Text:C. Standardization
Weight of vial and KHPH
59.09
Weight after 1st transfer
58.2
Weight after 2nd transfer
56.5
Weight after 3rd transfer
55.6
Weight of KHPh in flask I (show calculations)
0.89
50-9 - 58.2
Weight of KHPh in flask II (show calculations)
53.2-56.5
1.79
Weight of KHPH in flask III (show calculations)
0.99
56- 5-55-6
I
II
III
Initial volume reading of NaOH
O-00 ml
27.00
S1-00
Final volume reading of NaOH
as.00 ml
SI-00
80-00
Volume of NaOH used
25-00 ml
24.00 ml
29.00ml
Molarity of NaOH
0.156.M
0 347M
0 152M
Trial 3: KHPH in flosk II = o.99
Show calculations for all Trials Below:
Trial 1: KHPh in flask I = 0-89
Volume ofNoOH used 29ml =0.029L=
29x10 L
Volume of Na OH Used = 25mL = 0.2asL= a5x103
3-92x10 moles
Moles of Na0H = 0-89
204glmol
3.92x103
Moles of NaoH = 0 99
2049 lmol 4-41x10o
Molority of NaOH =
o'156 M
Molarity of NaOH = 4.41x 10 moles
%3D
a5x lo3
701X be
Trial 2: KHPh in Hask II = 1-79
Volume of NaOH used = 2u mL=0:024mL= 24X10°L
Molarity of NoOH = 0.15aM
Moles of a0H = 179
a049lmol
Molarity of NaOH 8.33 x10-3
24x lo t
- 8.33 x10 oles
0:347 mole s
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning