Q: elect the best answer. 1.) C- CO,Et b.) 1.) NaOEt, ELOH, A c.) 1.) H2SO4, H20, A 1.) NAOH, H2O, A…
A: In this question, we want to select best Reagent. I will explain you, how we can select best…
Q: assign the acid its appropriate pKa
A: The strength of the acid is measured by the stability of the conjugate base. Besides the pKa value…
Q: Lalaulate the eH of a 3 lox1D M la/OH),s Selution.CafoH)is. isa strory buse.
A:
Q: Nitromethane is a weak base. CH3NO2(aq) + H2O(1) = CH3NO2H+(aq) + OH¯(aq) A solution with a…
A: Concentration of nitromethane = [CH3NO2] = 0.0500 mol/L pH of solution = 11.5 We need to determine…
Q: using the IUPAC rules name every (all four) molecule.
A: The given structure is, From the structure it is clear that, The longest carbon chain has 7 carbon…
Q: 5. CH3COOH + CH;CH2OCOCH3 + H2O -COCH3 6. (6a) _(6b)__ - + H2O -СООН 7. (7а). + 2 NaOH _(7b) +…
A:
Q: Q 20 Complete the following reaction: NC. NaOCH3 ELOH Ph NC Ph Ph CN CN Ph Ph NC
A:
Q: Calculate the pH of a buffer solution prepared by mixing 75 mL of 1.0 M lactic acid (see Table 2.6)…
A: A buffer solution is one which resists any change in its pH even upon the addition of small amounts…
Q: но H H. H. он + 3H20 -C H. H H H. H. 1. H,0' H-C- H-C -C- -C- -C H 2. H H но †N\CH»¾Ñ-Č(CH»),T+ 2n…
A: Polymerization are classified in chain polymerization, step growth polymerization. Now chain…
Q: 1. н-СЕС-Н Br Na* H (1 equiv.) in THF (solvent) 2. HØSO4/H,SO/H,O 3. LDA THF -78 °C 4. OEt
A: H2SO4/HgSO4 converts terminal alkyne to ketone LDA abstracts proton from the less hindered site
Q: e basic anions?
A: This is a multiple questions. According to guidelines in multiple questions, we can solve only the…
Q: `Me 1) excess Me-I N. Me 2) 60% NaOH; acidic work-up Ме NH2
A:
Q: Draw the reaction mechanism.
A: Given reaction,
Q: Given that the Ka of acetic (CH3COOH) is 1.70*10-5 at 5C. Calculate the Ka of the acetic acid at…
A: Solution: We have dissociation constant Ka1 for acetic acid at temperature T1, 5 degree is 1.70 *…
Q: uestion 5 Which one of the following is NOT a good way to ma O CH3COOH, CH31 and K2CO3 O CH3OH,…
A:
Q: Be sure to answer all parts. Calculate the following values. (a) What is the pK, of NO₂? (K₂ of…
A:
Q: And Cl+CH3OH+ eve+ + CH3O 4o CI (methoxide ion)
A: Since methoxide ion is acting as a base Hence it will extract a H+ from the compound.
Q: 1. Which is the hydrolytic reaction for the basic compound ethylamine (CH:CH:NH:)? CH;CH,NH-(ae) +…
A: A hydrolytic (or hydrolysis) reaction is the cleavage of a chemical bond by the addition of H2O(l)…
Q: 13. p-Aminobenzoic acid ( M.Wt=137 g/mol), 0.1 mol is converted ethyl-p-aminobenzoate (ethyl…
A:
Q: h) ? (name, str, stereochem) -----cold dilute KMNO4---- (±) erythro pentane 2,3 diol H- HHOH CHCH i)…
A: h) cis compund + cis addition results erythro pentane-2,3-diol. i) 3E -hex-3-ene reaction with…
Q: NaOH но- Br
A: The given reaction is reaction of phenol with NaOH and then with allyl bromide.
Q: OH HH HHHH HHHH HHE I I II IILI Н-о—с-с -С-с-с-с-с-с-С-С-с-с-с- HHH I | | L -с-с-с-—С-н H H HHH H H…
A: Given carboxylic acid is saturated fatty acid
Q: Oxymercuration- Demercuration Reaction OAc mercurinium ion Hg(OAc) Hg* Oxymercuration-Demercuration…
A:
Q: mCH3 व्कnशम्परिवककाट ९वmm-hio १Tmphithihe ) athic hydorophilic hx৭00१h०bic IC
A: There are 14 carbons in the long chain and this is an alkene . Its name is tetradecene. In this…
Q: -OH- CI
A: Protonation of O-atom of epoxide . Attack to nucleophile to the most substituted side.
Q: When the Following ronction Using 19 starting material 500g Propanate, A-C-C-C-0-C-4 fi Methyl…
A:
Q: reactanrdcts of the reaction the products should have the same overall charge but the charge will be…
A: Complete detail solution is given on pages as below
Q: pH of unboiled H2O: 5.90 [H+] for unboiled H2O [OH−] for unboiled H2O
A: In our daily life, importance of pH is there. The Importance of pH is everywhere in food and…
Q: a. m-chloroethylbenzene I.CH.CCI AICI, ZaHg) H,NNH, or HCL, A HO, A CH CH,CH, CH, 2 HO or H Pd/C CI…
A: We have find out mechanism for both reactions of a and b.
Q: 2. Salicylic acid reacts with two equivalents of ICI to give one of the products below. Which one is…
A: ICl breaks down to form I+ and Cl- ions because chlorine is more electronegative than iodine due to…
Q: (b) Calculate the pH of 0.0005 mol dm³ ethanoic acid when its pK₁ = 4.75 CH₂COOH(aq) à CH₂COO (aq) +…
A: Given: 0.000 mol dm-3 ethanoic acid with pKa=4.75 To find: the pH.
Q: *Lo00 wt-2, NaoH : 0.1N
A: Normality : It is number of gram mole of solutes present or dissolved in 1 litre of solution.
Q: (b) Calculate the pH of 0.0005 mol dm³ ethanoic acid when its pK 4.75 CH₂COOH(aq) à CH₂COO (aq) + H*…
A:
Q: NaN, compound a compound b + compound c Reagents m. Na / NH, n. H,SO, H9SO. o. (sia),BH then H,O,…
A: In the given reaction the alcohol is converted into alcohol and the alcohol is converted into alkyl…
Q: 16. Acetylsalic ylic acid (ASA), C9 H804(s), is used as
A: 1. Molecular mass of ASA (C9H8O4) : = 9 (12) + 8 (1) + 4 (16) g/mol = 108 + 8 + 64 g/mol…
Q: Vhat is the product? H. 1. PhMgBr. Et,O 2 H,0*
A:
Q: Please show arrows for mech. thank you f. он Cu(CH;CN);OTf, TEMPO, air, NMI, bpy Ph OH Ph он РОС,…
A:
Q: Benzoic acid (C6H5C02H) has Ka =6.3 x 10-5 a) What is the value of Kb for the benzoate ion…
A: We have given benzoic acid ( C6H5COOH ). The equation of the reaction becomes :C6H5COOH + H2O →…
Q: H. HO" CH, CH,0" CH,Of H,C OMe 1. EIONa CH OE 2. mild acidie workup
A: The reaction of the aldehyde with the cyclohexandione in the presence of base leads to the…
Q: What is the corrеct Нaworth projеction for B-D-alloругanose? CH,CH CH,OH CH-OH CH;OH CH,OH он он он…
A:
Q: L J K & & co COOEt OH Ethanol & H₂SO4 COOEt COOEt 1. NaOEt, then CH3CH₂l 2. NaOH then acidify 3.…
A: Detail mechanistic pathway is given below to find out the major product
Q: 1) CO2 (s) g) Ph(CH2)MgBr 2) 11 (aq) H'(aq/A h) 6-Bromohexanoic Acid + NACN - 1) NaOEt/E:OH i)…
A: Solution: g) Grignard reagent react with co2 in presence of acidic condition corresponding…
Q: 16) 100. 80. 8 Rel. Abundance Rel. Abundance 40. 100. 80.- 60.- 40. 15 (5.4%) A) 2-propenal propanal…
A:
Q: 2- Complete the following reactions. NaOH NaCI OH C,H15- C,H15 NaOH C,H15
A: Carboxylic acids reacts with strong base NaOH and undergoes acid base reaction producing salt and…
Q: S-OH TSOH HO, acetic acid p-toluenesulfonic acid pKa = 4.8 pK = -7
A:
Q: What would the concentration of CH3C00 be at pH 5.3 if 0.1M CH3CO0H was adjusted to that pH. 1) pH =…
A: Henderson-Hasselbach equation. this equation shows the relationship between pH of a solution, the…
Q: Cro., aqueous ammoniacal AGNO. Brz in concentrated aqueous snoənbe H.SO, CH:Cl HCl with ZnCla FeCl.…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: RIGEROUS OX CH3-CH,- OH A) MILD OX B) H*/140°C 2 CH — CH—он C) H20 H* D) H,0 + A) CH3-CH2-OH
A:
Q: 2014 male luler Yomul nach paoden Hnanle , A= 271 (s) Comprunds ond Y yhoore he Compound X has 8-…
A: This Question is based upon NMR Spectroscopy.
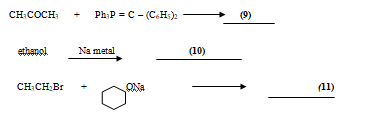
Fill in the blanks, write the name of the reactant and the product.
Step by step
Solved in 2 steps with 2 images

- 1) A polyester made from sebacic acid (CAS# 111-20-6) and 1,6-hexanediol (CAS# 629-11-8) has the molecular weight fractionation listed below. Calculate an appropriate average molecular weight for this polyester. Source: Batzer, H., Macromolecular Chemistry and Physics 5A 5.00-mL aliquot of a solution that contains 3.05 ppm Ni2+ is treated with an appropriate excess of 2,3-quinoxalinedithiol and diluted to 50.0 mL. The molar absorptivity of a Ni2+- 2,3-quinoxalinedithiol solution at 510 nm is 5520 L mol-1 cm-1. What is the absorbance of the above diluted Ni2+- 2,3-quinoxalinedithiol solution at 510 nm in a 1.00-cm cell?Draw the structure of the predominant form of CF3CH2OH (pK a = 12.4) at pH = 6.
- Rank the following groups in order of decreasing priority. −F, −NH2, −CH3, −OHWILL HEXANE DISSOLVE fenethylline IN GC-MS?Calculate the concentration of atmospheric carbon compounds in ppbv using the data in the table below: Compound Concentration (µg/m3) Toluene 980 m,p-Xylene 910 o-Xylene 510 Benzene 370 Ethylbenzene 310 1,3,5-Trimethylbenzene 230 1-Ethyl-4-methylbenzene 200 Hexane 150 Heptane 130 1-Erthyl-2-methylbenzene 120
- 4. o-dichlorobenzene and p-dichlorobenzene can be separated by TLC. Which compound is more polar? Which compound travels faster on the TLC sheet when hexane is used as the solvent? Explain. (hint- consider molecular polarity) 5. p-dihydroxybenzene and o-dihydroxybenzene can be separated by TLC using a solvent of medium polarity. The ortho isomer has a significantly higher Rf value than the para isomer. Suggest a reason for this difference in Rf values. (hint- consider the structure of these two molecules) 6. Explain how TLC and column chromatography can be used together to ensure the perfect separation of two organic compoundsmmol of 1,3-dibenzoylpropane used to 4 sig. fig Used: 405 mg molecular weight: 252.31 g/molCompounds Mass 3-nitrophthalic acid used 200 mg = 0.2g 8% aqueous hydrazine used 0.4mL 3-nitrophthalhydrazide obtained 130 mg = 0.13 g sodium hydrosulfite dihydrate 0.6 g luminol obtained 70 mg = 0.07 g compute yield for nitrophthalhydrazide in the first step (assume nitrophthalic acid is limiting reagent) compute yield for luminol in the second step (using nitrophthalhydrazide as limiting reagent) compute yield for the overall reaction

