3. The overall result of glycolysis can be summarized by the equation on the right in which the glucose origins of the carbon atoms in pyruvate are color coded. Decide which gly- colytic reactions are responsible for the indicated distribution and
3. The overall result of glycolysis can be summarized by the equation on the right in which the glucose origins of the carbon atoms in pyruvate are color coded. Decide which gly- colytic reactions are responsible for the indicated distribution and
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter27: Metabolic Integration And Organ Specialization
Section: Chapter Questions
Problem 15P
Related questions
Question
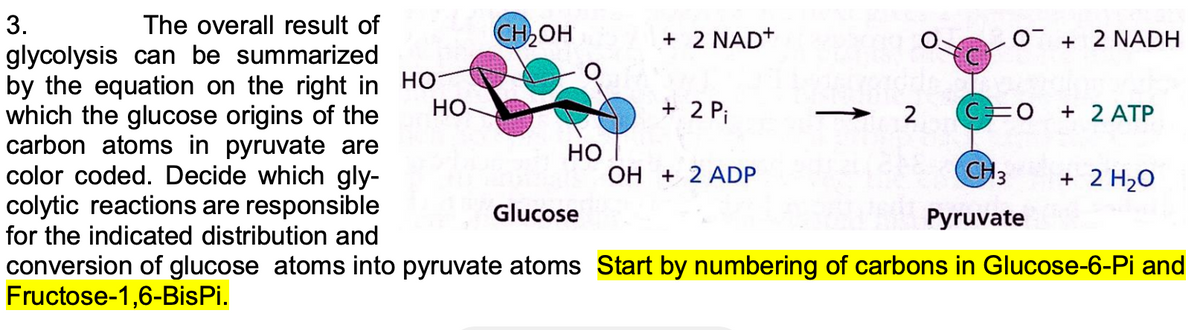
Transcribed Image Text:3.
The overall result of
CH OH
+ 2 NAD+
O + 2 NADH
glycolysis can be summarized
by the equation on the right in HO-
which the glucose origins of the
carbon atoms in pyruvate are
color coded. Decide which gly-
colytic reactions are responsible
for the indicated distribution and
HO-
+ 2 Pi
2
+ 2 ATP
Но
OH + 2 ADP
CH3
+ 2 H2O
Glucose
Pyruvate
conversion of glucose atoms into pyruvate atoms Start by numbering of carbons in Glucose-6-Pi and
Fructose-1,6-BisPi.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning