Consider the following two temperature vs composition phase diagrams of two ideal binary mixtures - one consisting of liquids A and B and the other consisting of liquids Cand D- with TA, TB, Te, and Tp being the boiling point temperatures of A, B, C, and D respectively at constant pressure. The curves in red are the vaporization curves while the curves in blue are the condensation curves. PA, Pe, P, and Pp* are the vapour pressures of A, B, C, and D respectively. Tg - TA is greater than Tp-Tc: The number of vaporization-condensation cycles required to achieve greater than 99 % purity in the first solution is NAg and the number of vaporization- condensation cycles required to achieve greater than 99 % purity in the second solution is NcD Which of the following stat aments is true?
Consider the following two temperature vs composition phase diagrams of two ideal binary mixtures - one consisting of liquids A and B and the other consisting of liquids Cand D- with TA, TB, Te, and Tp being the boiling point temperatures of A, B, C, and D respectively at constant pressure. The curves in red are the vaporization curves while the curves in blue are the condensation curves. PA, Pe, P, and Pp* are the vapour pressures of A, B, C, and D respectively. Tg - TA is greater than Tp-Tc: The number of vaporization-condensation cycles required to achieve greater than 99 % purity in the first solution is NAg and the number of vaporization- condensation cycles required to achieve greater than 99 % purity in the second solution is NcD Which of the following stat aments is true?
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter17: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 128IP: Some nonelectrolyte solute (molar mass = 142 g/mol) was dissolved in 150. mL of a solvent (density =...
Related questions
Question
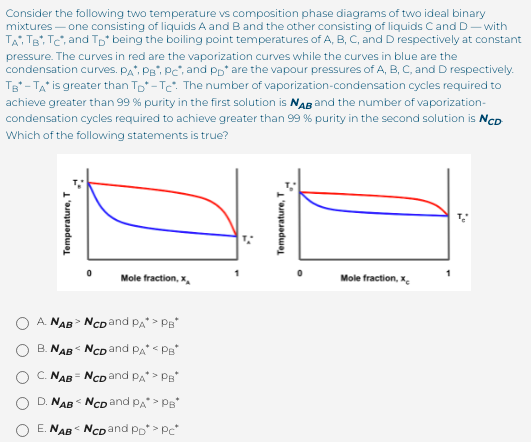
Transcribed Image Text:Consider the following two temperature vs composition phase diagrams of two ideal binary
mixtures – one consisting of liquids A and Band the other consisting of liquids Cand D- with
TAi TB, Tc, and Tp being the boiling point temperatures of A, B, C, and D respectively at constant
pressure. The curves in red are the vaporization curves while the curves in blue are the
condensation curves. PA, PB, Pc, and Pp* are the vapour pressures of A, B, C, and D respectively.
Tg - TA is greater than Tp* - Te. The number of vaporization-condensation cycles required to
achieve greater than 99 % purity in the first solution is NAB and the number of vaporization-
condensation cycles required to achieve greater than 99 % purity in the second solution is NcD
Which of the following statements is true?
Mole fraction, x
Mole fraction, x.
O A. NAB NCD and pa* > PB*
B. NAB NCDand Pa* < PB*
C. NAB = NCD and Pa* > PB*
D. NAB NCD and PA> PB*
E. NAB NCDand pD* > Pc
O O
Temperature, T
Temperature, T
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,