Consider the Haber-Bosch process for the synthesis of ammonia from its elements. Calculate the theoretical yield in moles NH: from the complete reaction of 59.8 grams N2 in the presence of excess H2 gas according to the following balanced chemical equation: N:(g) + 3 H:(g) - 2 NH:(g) 1 metN. 2 mol NH, 59.8 4.27 mol NH: STARTING AMOUNT 28.02 3 metNE 1 mol N2 2 mol NH, 59.8 g N2 x = 4.27 mol NH. 28.02 g N2 3 mol N2 ADD FACTOR DELETE ANSWER RESET *( ) 7.04 17.04 3 2.13 3.52 1.01 59.8 14.01 28.02 4.27 g/mol N2 g/mol NH. g NH, g N2 mol NH. mol N2
Consider the Haber-Bosch process for the synthesis of ammonia from its elements. Calculate the theoretical yield in moles NH: from the complete reaction of 59.8 grams N2 in the presence of excess H2 gas according to the following balanced chemical equation: N:(g) + 3 H:(g) - 2 NH:(g) 1 metN. 2 mol NH, 59.8 4.27 mol NH: STARTING AMOUNT 28.02 3 metNE 1 mol N2 2 mol NH, 59.8 g N2 x = 4.27 mol NH. 28.02 g N2 3 mol N2 ADD FACTOR DELETE ANSWER RESET *( ) 7.04 17.04 3 2.13 3.52 1.01 59.8 14.01 28.02 4.27 g/mol N2 g/mol NH. g NH, g N2 mol NH. mol N2
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter6: Chemical Reactions: An Introduction
Section: Chapter Questions
Problem 50AP: The Hall process is an important method by which pure aluminum is prepared from its oxide (alumina,...
Related questions
Question
Having trouble solving this
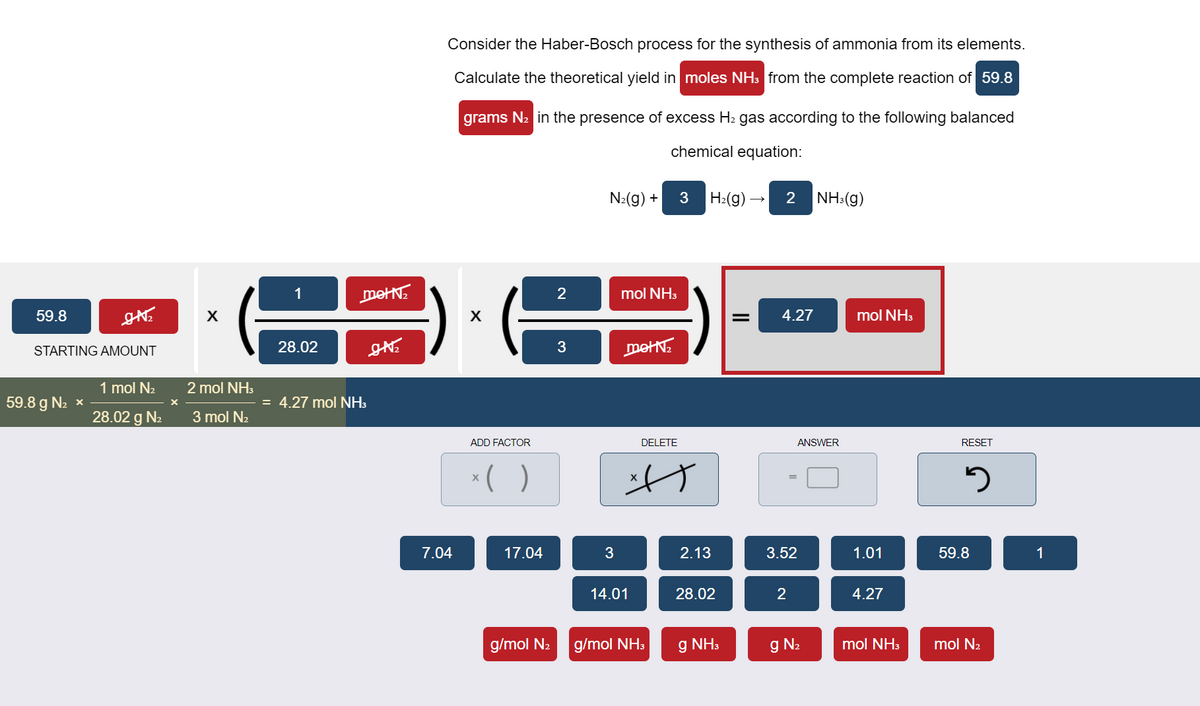
Transcribed Image Text:Consider the Haber-Bosch process for the synthesis of ammonia from its elements.
Calculate the theoretical yield in moles NH: from the complete reaction of 59.8
grams N2 in the presence of excess H2 gas according to the following balanced
chemical equation:
N:(g) +
H:(g) –
2
NH:(g)
1
metN:
mol NH3
59.8
4.27
mol NH3
STARTING AMOUNT
28.02
3
metN.
1 mol N2
2 mol NH3
59.8 g N2 x
= 4.27 mol NH3
28.02 g N2
3 mol N2
ADD FACTOR
DELETE
ANSWER
RESET
*( )
7.04
17.04
3
2.13
3.52
1.01
59.8
1
14.01
28.02
4.27
g/mol N2
g/mol NH3
g NH3
g N2
mol NH3
mol N2
2.
2.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning