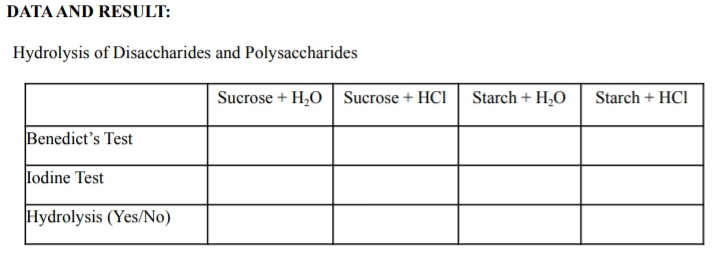
DATA AND RESULT: Hydrolysis of Disaccharides and Polysaccharides Sucrose + H,O Sucrose + HCI Starch + H,O Starch + HCI Benedict's Test lodine Test Hydrolysis (Yes/No)
Q: Listed below are five amino acids. Use the genetic code to determine the exact codon for each amino ...
A: Transition refers to a point mutation that changes a purine nucleotide to another purine or a pyrimi...
Q: Using the data provided in the table, the estimated p50 for myoglobin is mmHg and the fraction satur...
A: Myoglobin (Mb) is an iron and oxygen binding protein that is present usually in the cardiac and ske...
Q: What is the m/v % concentration of a solution of LiCl with a density of 2.07 g/mL in 40.0 mL of deio...
A: Given Values: Mass or weight of LiCl = 2.07 grams The volume of the solution = 40 ml The density of ...
Q: What group is added to modify methionine on the initiator tRNA in bacteria O phenyl O formyl O methy...
A: Initiator tRNAs (tRNAi) transport methionine (or its product, formyl-methionine) to ribosomes, where...
Q: What are the advantages of the presence of organelles in eukaryotic cells?
A: The eukaryotic cell has several organelles and is compartmentalized.
Q: 3. Tumor derived growth factor B (TGFB) is a secreted protein that has a diverse range of blological...
A: TGF-β (Transforming Growth Factor-β) employs the Smad proteins as the intracellular mediator of sign...
Q: Upon the addition of diazonium salts, ketoacids like glutamate and pyruvate forms in the presence of...
A: 1. The correct answer of question is: a) both false 2. The correct answer of question is: d) incr...
Q: For item 2 choose only one correct answer. No need for explanations.
A: Photosynthesis is a food-making mechanism used by green plants and some other lower-level organisms....
Q: Name the three classes of lipids found in the body and in foods. What are some of their functions in...
A: Lipids are biomolecules which are non-polar in nature. Lipids are not considered as true polymers be...
Q: Based on our class discussion of the currently accepted mechanism for ribosome catalysis, draw a gra...
A: By transferring the nascent peptide chain from P-site peptidyl-tRNA to A-site aminoacyl-tRNA (aa-tRN...
Q: What is the difference between symptom and sign. Give examples of symptoms and signs
A: Signs are something that physicians and other people notice it. Symptoms are something that patients...
Q: Help with #4 thanks
A: Enzymes are proteins that aid in the speeding up of metabolic reactions. Certain chemicals can slow ...
Q: Distinguish between prokaryotic and eukaryotic microorganisms and compare them usingthe characterist...
A: All living organisms are made up of cells, that is cells are the building blocks of life. Organisms ...
Q: Which of the following has the highest iodine content? Which of the following has the lowest meltin...
A: 1. Linoleic acid has the highest iodine content. 2. Unsaturated fatty acids have lower melting poin...
Q: For the following, calculate pka, and pka, of 1 Glycine, determine the 2 buffering regions, and calc...
A: Glycine is an amino acid with H-atom as the side chain. The isoelectric point of a amino acid (pI) i...
Q: Determine the values of Km and Vmax for the decarboxylation of β-keto acid given the following data....
A: Those classes of proteins that mainly act as a catalyst in the living body by increasing the rate of...
Q: What are examples of polyiodides? Include their structure
A: Polyiodides are a class of polyatomic halide anions that consists entirely of iodine atoms. It is pr...
Q: Draw the product of the reaction below. (Upload your answer here) H- но ? NaBH4 HO -H H- ČH2OH
A: Given structure :- D-GALACTOSE (monosaccharide) In presence of NaBH4 , REDUCTION reaction oc...
Q: Explain how the carbonate-bicarbonate buffer system works in balancing acid-base in the blood.
A: Buffers are solutions that have weak acid and its conjugate base. They nullify small changes in the...
Q: WHAT ARE THE ADVANTAGES AND DISADVANTAGES OF CARBOHYDRATES IN BIOCHEMISTRY? GIVE AT LEAST 3 FOR EACH...
A: Carbohydrates are the one of the biomolecules that provide energy to the human body. They are mainly...
Q: What risks are involved in genetic engineering of crop plants? How do these risks compare with other...
A: Genetic engineering is a technique where the genes are edited or newly genes are inserted using gene...
Q: In UV/Visible spectrophotometer analysis for a multicomponent system, there are only two dyes used i...
A: UV/Visible Spectrophotometry detects the absorbance of light in the UV/Visible range of the electrom...
Q: [N] NO MATCH 1. yields 2 moles of fatty acid, phosphoric acid, glycerol and choline upon hydrolysis ...
A: Since we only answer up to 3 sub-parts, we'll answer the first 3. Please resubmit the question and s...
Q: Name the experiment shown above and briefly describe how it is set up as well as the role of each co...
A: Gel electrophoresis is a method of separation of protein (SDS-PAGE, polyacrylamide gel electrophores...
Q: Between direct and indirect allosteric kinase inhibitors, which do you think requires a larger confo...
A: In Allosteric modulation of enzyme/protein function, a modulator molecule binds at a site other than...
Q: Approximate molecular weight for an unknown protein from gel-filtration experiment is 130 kDa. Thirt...
A: Given Values: Molecular weight obtained from gel-filtration experiment = 130 kDa Weight of the fluor...
Q: Does genetically modified fish is good or bad to humans?
A: With increase in human population, food demand is increasing and there is high pressure on the suppl...
Q: Equation for the esterification of glycerol and three ethanoic acids
A: Esterification reaction refers to condensation reaction between alcohols and carboxylic acids. This ...
Q: Qualitative Analysis of Proteins Tests
A: Proteins are large biological molecule composed of amino acid, amino acids contain both amino (nh2) ...
Q: How to use the sterile filter? Show the different sizes and specifications of sterile filters.
A: Sterilization is the method that is used to eliminate all forms of life and biological agents presen...
Q: The synthetic process by which monomers are covalently linked is (a) hydrolysis (b) isomerization (c...
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen but there are other element...
Q: what type of reaction is the the reverse synthesis of ceramides from sphingosine
A: Ceramides belong to the family of waxy lipids that contain sphingosine and fatty acids. It is abunda...
Q: Below is the titration of histidine. Calculate the average charge of histidine at pH 6.50. ÇOON Hist...
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer the first q...
Q: importance in familiarizing and knowing the uses of common laboratory
A: The question is all about the knowledge and familiarity of common laboratory equipments that is used...
Q: Provide a detailed narrative description of the mechanism of carbonic anhydrase.
A: Carbonic anhydrase : It is a metaloenzyme (complexed with zinc) which mainly catalyzes the inter con...
Q: If you have a protein kinase that is regulated by both small molecule inhibitors as well as by phosp...
A: Protein kinases is enzyme which catalyses the transfer of phosphate between their substrates. A prot...
Q: "VDAATFKQANDNG" is the sequence of an a helix. Which of the following interactions is probable? (. d...
A: VDAATFKQANDNG In alpha-helix : H-bonding occurs b/w ith(CO group) and i+4th(NH group) amino acids.
Q: Complete the table below using the textbook or other information sources, if necessary. How will the...
A: A functional group is a group of atoms in a molecule that gives particular characteristics and chemi...
Q: d) must be an Fe-containing protein. If she were correct, what type of protein is this most likely t...
A: Ans d) Iron-sulfur proteins, also known as nonheme iron proteins contain iron complexed to cysteine ...
Q: 1mL of the stock solution acetaminophen (1mg/mL) is used for the estimation of PC using n-octanol. A...
A: Acetaminophen is also commonly known as a Paracetamol that is a medicine used to treat the fever and...
Q: Calculate how many milligrams of biological oxygen demand (COD) if the BOD is 25 ppm.
A: Here COD in question is Chemical oxygen demand and BOD is biological oxygen demand. Normally relatio...
Q: In cells, certain hormones, such as epinephrine, have the ability to raise the concentration of cAMP...
A: The ability of a protein to bind to a DNA segment can be analyzed using the method of Electrophoreti...
Q: What's the benefit of an insecticide?
A: Chemicals used to control pests and diseases in plants and animals are known as insecticides. Insect...
Q: Based
A: Insulin binds to the receptor activates several cascade of events.
Q: How are blood glucose concentration affected by heavy alcohol consumption? Please explain with extre...
A: Alcohol plays major role on health. Alcohol can effect every organ of the body. In an average person...
Q: 1. What is hemoglobinopathy? What is sickle-cell anemia? Explain the concept relating to protein str...
A: Hemoglobinopathy is a group of inherited blood disorders which occurs due to abnormal production or ...
Q: do archaebacteria have autotroph or heterotroph nutrients
A: Nutrients are the components of food like carbohydrates, fats, vitamins, proteins, etc. These nutrie...
Q: Why did Okazaki propose that lagging strand synthesis proceeds through the synthesis of short fragme...
A: The effective copying of double-stranded chromosomal DNA is required for cellular DNA replication. T...
Q: Is the H+/sucrose cotransport system involved in passive or active transport? How do you know?
A: Secondary active transport is the transport in which the electrochemical gradient that is generated ...
Q: 2) A group of three researchers (who often tend to disagree with one another) isolated a new protein...
A: a) This must be enzyme containing a iron- sulphur cluster not a Mn- cluster.Iron- sulphur cluster co...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Color Reactions of Intact Protein: 1ml of water and 0.5 g of casein 1ml of water and 0.5 g of bean protein 1ml of water and 0.5 g of glutentopic: ISOLATION AND CHARACTERIZATION OF CARBOHYDRATES 1. Describe the reaction that takes place when starch is hydrolyzed by an enzyme. What is the product of starch hydrolysis? 2. What enzyme is present in the saliva? What conclusion can be made regarding the action of this enzyme on starch?topic: ISOLATION AND CHARACTERIZATION OF CARBOHYDRATES 1. Erythrose is an aldoterose. Describe the result when it is subjected to each of the following tests:a. Molisch’s Testb. Barfoed’s Testc. Benedict’s Testd. Seliwanoff’s Test2. Illustrate the reaction and identify the principle involved in the Nelson test.
- Answer using Person Square method. Calculate to prepare a 100 g fish feed that includes 45% crude protein (CP), using Fish meal with 65% CP (as source of Protein) and Soybean meal with 40% CP (as source of carbohydrates). Show your solution. What is the total contribution of fish meal in 100 g diet? How much soybean meal is needed to produce the fish feed?Structure activity relationship of pindololActivity 6.3.1: Complete the Table Polysaccharide Unique Feature Important Function Chondroitin Heparin Hyaluronate Dermatan Sulfate Keratan Sulfate Mucin
- As sweet as sucrose is, an equimolar mixture of its constituent monosaccharides, D-glucose and D-fructose, is sweeter. Besides enhancingsweetness, fructose has hygroscopic properties that improve the texture of foods, reducing crystallization and increasing moisture. In the food industry, hydrolyzed sucrose is called invert sugar, and the yeast enzymethat hydrolyzes it is called invertase. The hydrolysis reaction is generally monitored by measuring the specific rotation of the solution, which is positive (+66.4°) for sucrose, but becomes negative (inverts) as more D-glucose (specific rotation = +52.7°) and D-fructose (specific rotation = −92°) form. From what you know about the chemistry of the glycosidic bond, how would you hydrolyze sucrose to invert sugar nonenzymatically in a home kitchen?Assignment on KetohexosesConstruct the structure of the following enantiomers or mirror images:1. D and L Fructose2. D and L Allulose3. D and L Sorbose4. D and L TagatoseResult nad Discussion Lead Acetate Reaction: Samples: lysine, cysteine, methionine Reagents: 10% Sodium Hydroxide (NaOH) and Lead Acetate Pb(CH3COO)2 -To 1 ml of the amino acid solution taken in a test tube, add few drops of sodium hydroxide (40%) and boil the contents for 5-10 mins over a bunsen burner. Cool the contents and add few drops of 10% Lead acetate solution and observe.



