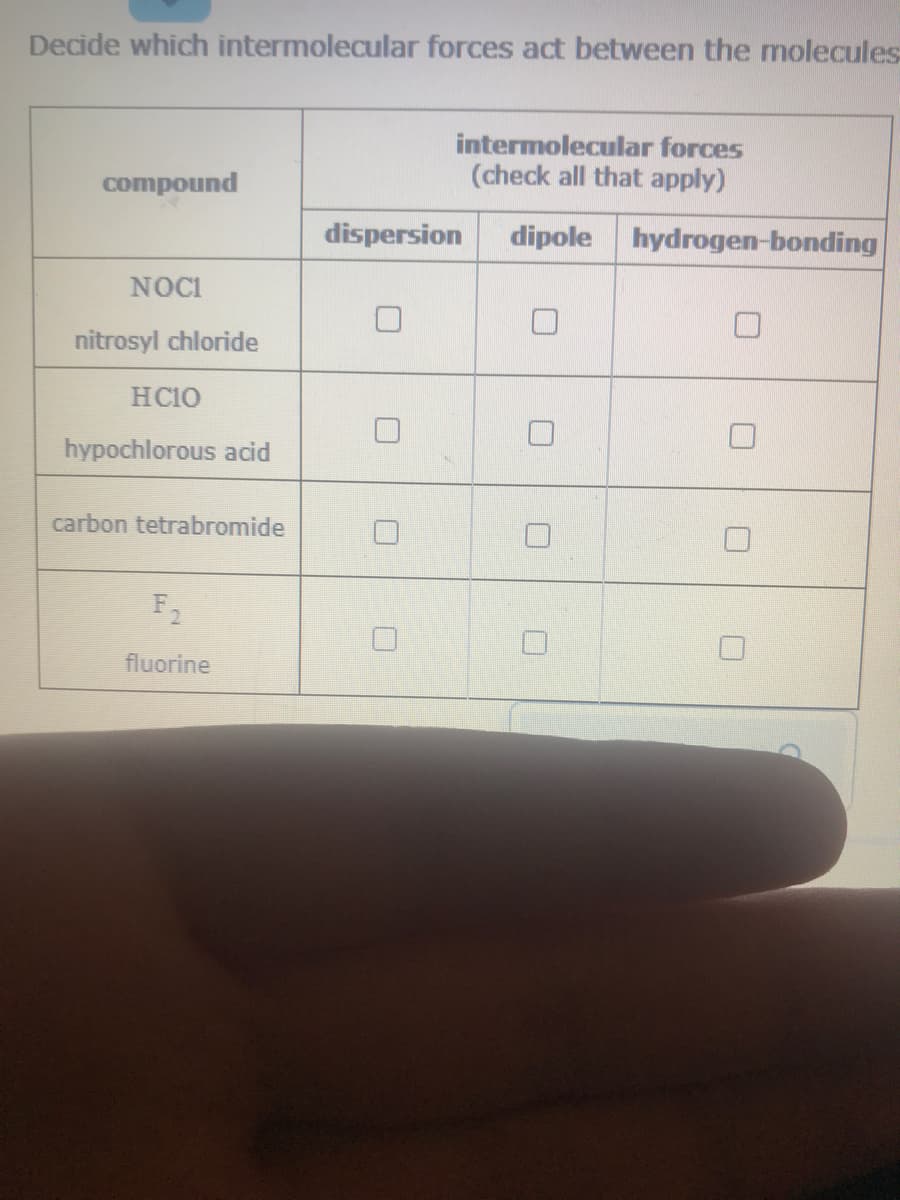
Decide which intermolecular forces act between the molecule- intermolecular forces (check all that apply) compound dispersion dipole hydrogen-bonding NOCI nitrosyl chloride HC10 hypochlorous acid carbon tetrabromide F2 fluorine
Q: A chemist mixes 8.0 g of carbon tetrachloride with 25. g of thiophene and 85. g of…
A: Introduction: Mass percent is the amount of elements with respect to their mass present in the given…
Q: No Are ions Yes present? Are molecules with permanent dipoles present? No Are polar No molecules…
A: Intermolecular forces are the attractive or repulsive forces present between the molecules. These…
Q: Choose the best answer for following question. An unequal sharing of electrons is a characteristic…
A: The attraction between atoms, ions or molecules forms chemical bonds. The chemical bond holds the…
Q: B-pt and M-pt of various organic compounds depends on intermolecular forces of attraction which…
A: Organic molecules are primarily made up of carbon, hydrogen, nitrogen, and oxygen atoms. The organic…
Q: Questions 5-7: Classify the following structures as isomers a, B, L or D (3 points). CH2OH (5) (6)…
A: Anomers are the cyclic monosaccharides or glycosides that are epimers differing from each other in…
Q: Identify the chirality center (sometimes called chiral atom) in each molecule. If the molecule does…
A: A chiral center is defined as an atom in a molecule that is bonded to four different chemical…
Q: How many stereoisomers are possible for the molecule formed by the reaction described in the…
A: Carbohydrates are an important biomolecule, all types of cells require carbohydrates for survival.…
Q: Choose the best answer for following question. Which of the following reactions combines two…
A: Answer is a.) dehydration.
Q: how many sigma bonds found in the thiocyanate ion 5 3 4 1 2
A: Covalent bonds are classified into sigma and pi bonds depending on the overlapping of atomic…
Q: Intramolecular ______ are responsible for formation of the most common three-dimensional shape in…
A: Proteins are unbranched polymers constructed from 22 standard a-amino acids. They have four levels…
Q: The formula for sugar is __________. The cation is __________, and the anion is __________.
A: Sugars are the soluble carbohydrates and used in food. Sugars are sweet in taste.
Q: The most unstable conformer in cyclohexane? Boat half chair Chair Twist boat
A: Cyclohexane is a non-polar molecule and it does not have any ring strain, which is the abnormality…
Q: hat is the pH of 0.6M acetic acid?
A: Acetic acid is a weak acid, to calculate the pH we need acid dissociation constant (ka) , for this…
Q: O=S=0 CI -NH OH H3C H3C Q1: How many functional groups present in the structure? Q2: Name the…
A: The answer is given below
Q: State whether the bonding in each compound is likely to be covalent and short reason to prove it i)…
A: An atom is the basic and fundamental unit of matter. An atom consists of electron and the nucleus in…
Q: Which hydrogen atom(s) in the following molecule is acidic? (That is, which hydrogen atom(s) will…
A: Answer - option - 3only
Q: Glycine hydrochloride (Cl− H3N+CH2COOH) is a diprotic acid that contains a carboxylic acid group and…
A: Titrations are often recorded on the graphs called titration curve, which generally contain the…
Q: 25.Which intermolecular force is responsible for the high surface tension of water? See figure for…
A: Water is a simple molecule made up of one oxygen atom bonded to two separate hydrogen atoms. Due to…
Q: Calculate the [H+], [OH-], and pH of the substance with pOH value of 4.82. Determine whether this…
A: A substance that donates hydrogen ions and accepts a pair of electrons during a chemical reaction is…
Q: Choose the best answer for following question. Hydrogen bonds are formed as a result of which of the…
A: Hydrogen bonding is an essential part of biomolecules including three main macromolecules in…
Q: Define the following terms:a. polarb. hydrogen bondc. electrostatic interactiond. salt bridgee.…
A: Intermolecular forces help in the stabilization of the molecules. All the biological molecules are…
Q: Calculate acetic acid when it requires 44.82mL of a 0.145M NaOH solution to titrate 34.95 mL of our…
A: M1VI=M2V2
Q: In X-ray crystąllography, the wavelength of an X-ray is - 1.5 Å. This is useful in determining a…
A: X-ray crystallography is a technique used in the determination of three-dimensional atomic…
Q: Calculate the molar mass of each of the following compounds:(a) Calcium carbonate, CaCO3 (b) Urea,…
A: Molar mass is defined as the mass of a substance in grams of 1 mole. It is calculated by summing all…
Q: Answer the following questions correctly choose the correct answer. 1._________dictates the maximum…
A: 1. Limiting reagent is the reactant that gets utilized first during a chemical reaction. It limits…
Q: Describe how you would prepare a 500 ml of 0.35M Glycine pH2.2 from a 5M Glycine solution of pH 7.…
A: Ans- Given Molarity = 5M Desired Molarity= 0.35 M Desired Volume (V2) = 500ml…
Q: Hydrogen and oxygen atoms within one molecule of water are held together by (a) bonds, while…
A: The answers for the fill in the blanks are given in step 2:
Q: Given the active site diagram below, which circled component coordinates the cofactor? 1 05 02 5 NH…
A: Introduction: A large number of enzymes require an additional component that is non-protein in…
Q: could you please let me know what "reactions occur" in each part of the structure?
A: Plants are the primary producers in the ecosystem. They harvest solar energy and convert it into…
Q: Describe the nature and relative strength of covalent bonds, ionic interactions, and van der Waals…
A: Bonds are formed between two atoms, ions, or molecules.
Q: Match the molecule with the correct geometry. Unit A page- 18 - Chemistry 20 38. tetrahedral 1…
A: Molecular geometry, also known as the molecular structure, is the three-dimensional structure or…
Q: Which statement about functional groups is true? Group of answer choices Functional groups may…
A: Functional groups contain specific atoms/elements bonded together in a compound by covalent bonds…
Q: What is the type of leaf outline in these figures?
A: Leaves are the vegetative organs of a plant that are specialised to perform photosynthesis mainly as…
Q: What is the pH of a grapefruit that contains 0.007 M citric acid solution (CHgO7)? C6H307(aq) +…
A: Acids are substances that are capable of donating a proton to the aqueous solution. Weak acids…
Q: correctly identifies a phosphate ion? Chemical symbol Cation Anion PO 3+ A 4- В PO, PO РОЗ- PO 3- 4.
A: Biomolecules are those that are produced or found in living organisms. They are of different types,…
Q: Which of the following structure/structures will go through a combustion reaction (is flammable).…
A: The reaction involving the release of energy (in light, flame, or heat form) when a substance…
Q: What is the Electron Geometry of a AB2C molecule? tetrahedral trigonal planar trigonal pyramidal…
A: As no compound is mentioned specifically so Electron geometry and molecular geometry is same , which…
Q: Indicate the COOH-pKa value of the HAY tripeptide on the titration curve. Titration curve of…
A: The titration curve of peptides indicates the buffering zones and the pKa value of each ionic…
Q: Identify the functional groups in the following molecule as pointed by arrow A and B, then C and D
A: An atom or group of atom in the organic molecule which determines its characteristics chemical…
Q: The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by…
A: At pH=1 (pH<7), there is an excess of H+ ions in the aqueous solutions. So, the COO-n group…
Q: For the following reaction at equilibrium NH3 + H20 e NH4+1 + OH-1 NH3 is the It is not a hydroxide…
A: In the reaction, it involves the formation of ammonium ions from ammonia by accepting the proton…
Q: Two students plan and carry out an investigation to determine if ionic or covalent bonds have…
A: Chemical compounds are often classified majorly into two categories-ionic compounds and covalent…
Q: Which of the following is a hydroxide ion? H3O + H + H2O - OH -
A: Hydrogen is defined as the chemical element, having the symbol H. It has the atomic number 1. The…
Q: Identify the predominant intermolecular forces in each of the given substances. Electrostatic…
A: The state of matter of a substance is how each molecule interacts with the other molecules around…
Q: Polymers of macromolecules are disassembled to monomers by: O a. Van der Waals interaction O b.…
A: A polymer is a large molecule that is made from many small repeating chemicals called monomers.
Q: The following amine acids are given. SH CH3 *HạN-CH- ČH2 *H,N-CH- or Cysteine Alanine Draw…
A: Introduction Fatty acids These are long-chain hydrocarbons with carboxylic group end. Three fatty…
Q: What are the predominant Intermolecular forces (Dipole-dipole, London dispersion, Hydrogen bonding…
A: An atom is the smallest particle that exists in nature. It has three sub-atomic particles known as…
Q: Tests Principle behind functional group that will give evidence of a References the test a positive…
A: Generally carbohydrates are defined as polyhydroxy aldehydes or ketones . carbohydrates also are…
Q: What ion assists in stabilization of complexes of nucleic acids? Mg2+ Fe2+ O K* Na* Ca2+
A: DNA and RNA are nucleic acids which are actually salts of metal ions. So it is impossible to…
Q: C. The dissociation constant, (Kdisso) for the ES complex
A: Option c is the answer
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- what other intermolecular forces exist between these if any? i ask because dipole-dipole are always accompanied by dispersion forcesCalculate acetic acid when it requires 44.82mL of a 0.145M NaOH solution to titrate 34.95 mL of our unknown acetic acidDoctor ordered Haloperidol 3mg po bid. You have scored tablets containing Haloperidol 2mg. How many tab will you give?
- Order: labetalol 40 mg IVP STAT now Using the following drug label, how many milliliters of labetalol will you give? __ mL Round your answer to the nearest whole number.After her latest dialysis treatment, Michelle experienced vertigo and nausea. Michelle's doctor orders 0.044 g of chlorpromazine, which is used to treat nausea. If the stock solution is 3.4 % (m/v), how many milliliters are administered?Order: Dilantin 100 mg IV dailyAvailable: Dilantin 250 mg per 5 mL How many milliliters would you give? __ mL Round your answer to the nearest whole number.
- A chemist prepares a solution of barium chlorate BaClO32 by measuring out 19.μmol of barium chlorate into a 300.mL volumetric flask and filling the flask to the mark with water. Calculate the concentration in /μmolL of the chemist's barium chlorate solution. Round your answer to 2 significant digits.Which among the bonds indicated here in a polypeptide structure is rotation impossible? (kindly show explanation or illustration. thank you!) Choices: A. 2B. information is not enough to answer the questionC. 3D. 1cis, cis-9,12-OCTAdecadiENoic acid Enumerate the number of carbons and unsaturations (double bonds) in this molecule
- i) Write down the equation derived from your Excel generated standard curve , figure legend and describe its components; ii) provide the values of the absorbance data of the unknown sample (do NOT show the absorbance data of the glycine standards). Show all details of the working out of your calculation. Indicate all units! Provide the answer with two decimals precisionDefine the following terms:a. polarb. hydrogen bondc. electrostatic interactiond. salt bridgee. dipoleThe molecular weight of ethanol (CH3CH2OH) is46 and its density is 0.789 g/cm3.A. What is the molarity of ethanol in beer that is 5%ethanol by volume? [Alcohol content of beer varies fromabout 4% (lite beer) to 8% (stout beer).]B. The legal limit for a driver’s blood alcohol contentvaries, but 80 mg of ethanol per 100 mL of blood (usuallyreferred to as a blood alcohol level of 0.08) is typical. Whatis the molarity of ethanol in a person at this legal limit?C. How many 12-oz (355-mL) bottles of 5% beer coulda 70-kg person drink and remain under the legal limit? A70-kg person contains about 40 liters of water. Ignore themetabolism of ethanol, and assume that the water contentof the person remains constant.

