Q: What is deionized water? What kind of impurities are not removed by deionization?
A: Purified water does not contain any impurity. There are various methods for purification of water…
Q: What complications would a dried-out column (solvent level is below the top of the silica) introduce…
A: Answer
Q: objecti syntnesizing, characterization and ligand exchange of arene molybdenum complexes using…
A:
Q: What are the processes of concentrate samples analysis in Uv-visible spectroscopy? Please answer…
A:
Q: In this experiment, we rely upon Infrared (IR) Spectroscopy to confirm that the expected reaction…
A: IR spectroscopy is a technique used for the identification and structural analysis of a compound.
Q: What are the advantages of a dropping-mercury electrode in polarography? Why is polarography used…
A:
Q: Which one below is not one of the reasons for adding inorganic additives to organic matrix in…
A: They combine the advantages of the inorganic material (ex: rigidity, thermal stability) and the…
Q: 1.A compound Y (150.0 g/mol) in a 1.00 cm quartz cell has an extinction coefficient (ε) of 23,150 at…
A: Answer to Question No:1 The energy of a photon can be calculated by the famous formula- E=hcλwhere,…
Q: What are flame tests used to identify?
A: When the electrons of atoms are ground state, the electrons will be present in the lowest possible…
Q: a. The structure of a standard C4 group chemically bonded to a silica surface through a silyl ether…
A: a) C 4 silica gels are used for the separation of hydrophobic substances in reversed phase…
Q: Two solutions of the same UV-absorbing molecule were analyzed by UV-vis spectroscopy on the same…
A: Given the path length of the cell, b = 1 cm Since the two solutions, A and B have the same…
Q: Which of the following statements is/are false about flame test? I. The color emitted by a compound…
A: A multiple choice question based on flame test, which is to be accomplished.
Q: One common way to determine Phosphorous in urine is to treat the sample after removing the protein,…
A: #(i): Calibration curve is plotted by taking concentration(ppm P) in X-axis and absorbance in…
Q: List two major disadvantages of taking the IR spectrum of a solid by dissolving it in carbon…
A: The two major disadvantages of taking the IR spectrum of a solid by dissolving it in carbon…
Q: What are the different tetrabasic forms of EDTA? Describe their individual reactivities.
A: The structure of EDTA: EDTA (Ethylenediaminetetraacetic acid) is an amino polycarboxylic acid. The…
Q: In general, we want to have absorptions less than or equal to 1. Given that an ethanol solution of…
A: Given that: absorbance - 1 extinction coefficient (ε) = 23,150 length of light = 1.00 cm To…
Q: Which are true for polarography? I Polarography is amperometry conducted with a dropping…
A: Please find the below text answer. Thank you
Q: b) Describe the principles of operation of graphite furnace Atomic Absorption Spectroscopy (GFAAS)…
A: According to the Q&A guidelines of Bartleby, I am allowed to answer only three sub-parts of the…
Q: Assume that there was an excess of (L)-isovaline on the Murchison meteorite. Discuss the…
A: Noorduin, et al., have demonstrated the enantiomeric enhancement upon crystallization by using amino…
Q: Explain melting point determination as a characterization technique.
A:
Q: Nickel bis-dimethylglyoximate complex was depősited with a weight of (4.869g) with the filter paper…
A: % of Ni ions = ? Mass of complex + filter paper = 4.869 g Mass of filter paper = 1.30 g
Q: Discuss the phenomenon that gives rise to band broadening in chromatography.
A:
Q: 4. Determine the expected diffraction angle for the first-order reflection from the (113) set of…
A:
Q: Explain the difference between absorption and adsorption in natural dyes. Use sketches to clarify…
A: Please find your solution below : Absorption is the process in which absorbed material is uniformly…
Q: with the aid of a diagram briefly but informatively discuss the principles of the photomultiplier
A:
Q: Justify and explain following statements. (a) The adsorbent should be insoluble in the solvent. (b)…
A: Adsorption is a surface phenomenon in which molecules, atoms, ions get adsorbed on the surface. This…
Q: What happens if the solvent concentration falls below that of the silica stationary phase? What is…
A: The chromatography technique consists of two phases mobile and stationary phase .Mobile phase is the…
Q: Gadolinium is used as a contrast agent in MRI. Which magnetic state of matter is characteristic of…
A: A compound or ion is said to be paramagnetic if it contains unpaired electrons in its outermost…
Q: Which of the following is/are characteristic of silica as stationary phase in chromatography? O It…
A:
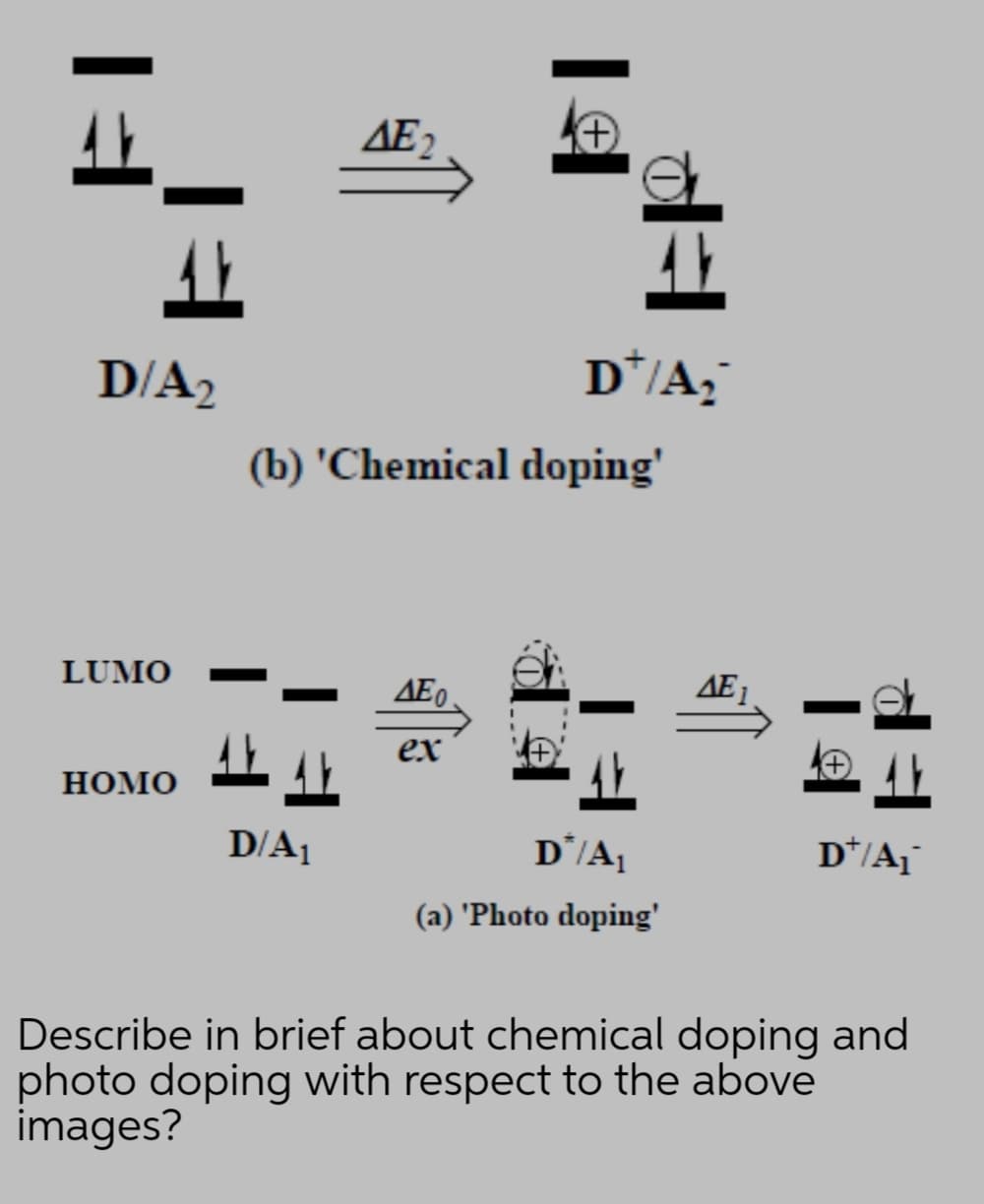
Q: What is HOMO LUMO and include reference link
A: A question based on molecules that is to be accomplished.
Q: 11. (8pts) how can silica gel Fsa TLC plates can be visualized ( give two methods? :
A: In the given question we have to visualise the F254 TLC plate, there are two method by which we can…
Q: How anodic stripping voltammetry works for analysis of chromate. How is the analytical signal…
A: A question based on electroanalytical methods that is to be accomplished.
Q: atomic absorption.
A: Atomic absorption spectroscopy is a spectroanalytical technique used for the quantitative…
Q: Discuss how the hollow cathode lamp can increase AAS selectivity.
A: Given : AAS selectivity To find : Hollow cathode lamp can increase the AAS selectivity Solution :…
Q: Explain in detail about the X-ray diffraction analytical method with suitable example.
A: X-rays are the types of electromagnetic radiation with a short wavelength which is produced by –…
Q: Make a comparative table, highlighting similarities and differences, between the materials: a.…
A: Polyamide has a much greater capacity than silica gel and cellulose and is complementary to PC in…
Q: Explain 'why glass cells are not used in, UV-spectroscopy.
A: To explain: Why glass cells are not used in UV-spectroscopy.
Q: Flame Atomic Absorption Spectroscopy (FAAS) method is selected due to its wide applicability for…
A: To discuss the safety and precautions during the FAAS experiment.
Q: In general, we want to have absorptions less than or equal to 1. Given that an ethanol solution of…
A: • According to Beer-Lambert law when a monochromatic light having intensity I is passed through the…
Q: Which of the following statements about the SF experiment is/are CORRECT? The monochromator is…
A: A multiple choice question based on analytical process that is to be accomplished.
Q: In a GC experiment, a 30.00 m long fused silica capillary column produced 150,000 theoretical…
A: Calculate the plate height for the given problem which contains number of plates, length and height…
Q: Generally high temperature is favourable for chemisorption.’ Why?
A: Given: Generally high temperature is favourable for chemisorption.
Q: The K2S208 product may be contaminated with K2SO4 or KHSO4. Suggest a method for determining the…
A: PREPARATION OF K2S2O8: The preparation reaction of potassium peroxydisulfate is takes place with the…
Q: An aqueous solution of compound X was placed in a cuvette 1 cm thick and its light absorption…
A:
Step by step
Solved in 2 steps

- Two species A and B are known to have water/hexane partition coefficient of 5.99 and 6.16. They are separated by elution on silica gel with hexane as eluent. The ratio for the packing VS/VM =0.425 Two species A and B are known to have water/hexane partition coefficient of 5.99 and 6.16. They are separated by elution on silica gel with hexane as eluent. The ratio for the packing VS/VM =0.425a. Calculate the retention factor for each soluteb. Calculate the selectivity factorc. How many plates are needed to provide a resolution of 1.5?d. What column length should be used to provide a resolution of 1.5 if the plate height ofthe packing is 1.5 ×10-3 cm?e. If the flow rate is 6.75 cm min-1, how long will it take to elute the two species?A sample of steel (0.506 g) was digested in acid and the solution made up to 100 cm3 with deionised water. Aliquots (25.0 cm3) of this solution were transferred into two volumetric flasks (100 cm3) labelled A and B. Flask A was made up to volume with deionised water. To flask B was added 10.0 cm3 of a 260 ppm standard solution of Mn2+ which was then also made up to volume with deionised water. The following absorbance results were obtained: Solution A = 0.356, Solution B = 0.578. Use these results to calculate the percentage (% w/w) of manganese in the steel sample.The molar absorptivity of Allura Red is 2.59 x 104 M-1 cm-1, account for the accuracy of the modified set-up. What could be the factors that contribute to the inaccuracy or the modified spectrophotometric set-up?
- The following volumes of a solution containing 1.10 ppm Zn2+ were pipetted into separatory funnels each containing 5.00 mL of an unknown zinc solution: 0.00, 1.00, 4.00, 8.00 and 12.00. Each was extracted with three 5 mL aliquots of CCl4 containing an excess of 8-hydroxyquinoline. The extracts were then diluted to 25.0 mL and their fluorescence measured with a fluorometer. The results for the standard addition calibration were as follows: Volume Std. Zn2+ / mL [Zn2+] / ppm Fluorometer Reading 0.00 6.12 1.00 7.41 4.00 11.16 8.00 15.68 12 20.64 a) Determine the concentration on Zn2+ ions in each of the standard solutions coming from the coming from the 1.10 ppm Zn2+ solutions, Please show your working1. Discuss briefly the specifications and test parameters for system suitability for HPLC analysis.In the food dye experiment, the molar absorptivity of Allura Red is 2.59 x 104 M-1cm-1, account for factors that may affect the accuracy of the modified spectrophotometric set up?
- A sample of body serum is to be analysed for sodium by flame emission spectroscopy. 1.00cm' aliquot of serum was pipetted into each of two 50.0cm' volumetric flasks. The first flask was diluted to volume with deionised water. The absorbance of this first solution was0.350. To the second flask 10.0 cm' of a 25.0 ppm sodium standard was added and the flask made up to volume with deionised water. The absorbance of this second solution was 0.720.(i) Calculate the concentration of the sodium in the body serum in mg dm-3.Using the absorbance of [FeSCN]+2 at the wavelength of maximum absorption which you obtained in part B, and the diameter of the tube, obtained in part A,calculate the molar extinction coefficient. given info: Concentration of Fe3+ stock solution, C1 = 0.001 M Volume of stock Fe3+ solution pipetted, V1 = 25mL Concentration of diluted stock solution, C2 = 0.0004 M Volume of Fe2+ solution and KSCN (upon mixing) = 10mL Weight of KSCN = 1g The reaction taking place is given as: The reaction taking place is given as: F e 3+ (aq) + SC N − (aq) ⇔Fe [SCN] 2+ (aq) the concentration of [FeSCN]2+ after the mixing of Fe3+ and SCN- is 2.5 x 10-3 M. Moles of [FeSCN]2+ = 2.5 x 10-5 moles Diameter:1.2cm The wavelength at which maximum absorption takes place is = 480 nmTwo species A and B are known to have water/hexane partition coefficient of 5.99 and 6.16. They are separated by elution on silica gel with hexane as eluent. The ratio for the packing VS/VM =0.425a) What column length should be used to provide a resolution of 1.5 if the plate height ofthe packing is 1.5 ×10-3 cm? b) If the flow rate is 6.75 cm min-1, how long will it take to elute the two species?
- Calculate the amount of phycocyanin in Sample 1 in mg where A620 = 0.193 and A650 = 0.095, taking into account the dilution factor of 100 ul, and the total volume of extract 45ml. Note your answer to 2 decimal places.state the three different modes of GC/MS, and explain more for each.. If the enthalpy change of an enzymatic reaction associated with the conversion of 2000 g of a target analyte substrate to 1 mol of the product is -100 kj/mol, the heat capacity of the system is 1 kj/K.Kg, Seebeck coefficient is 0.01 V/K, then estimate the output potential difference (ΔV) of a thermopile, with one pair number of the thermocouple.





