Describe the amino acid composition in terms of the general characteristics and comment on what the amino acid composition tells you about likely cellular location of the protein.
Describe the amino acid composition in terms of the general characteristics and comment on what the amino acid composition tells you about likely cellular location of the protein.
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter28: Dna Metabolism: Replication, Recombination, And Repair
Section: Chapter Questions
Problem 16P: Helicase Unwinding of the E. coli Chromosome Hexameric helicases, such as DnaB, the MCM proteins,...
Related questions
Question
Describe the amino acid composition in terms of the general characteristics and comment on what the amino acid composition tells you about likely cellular location of the protein.
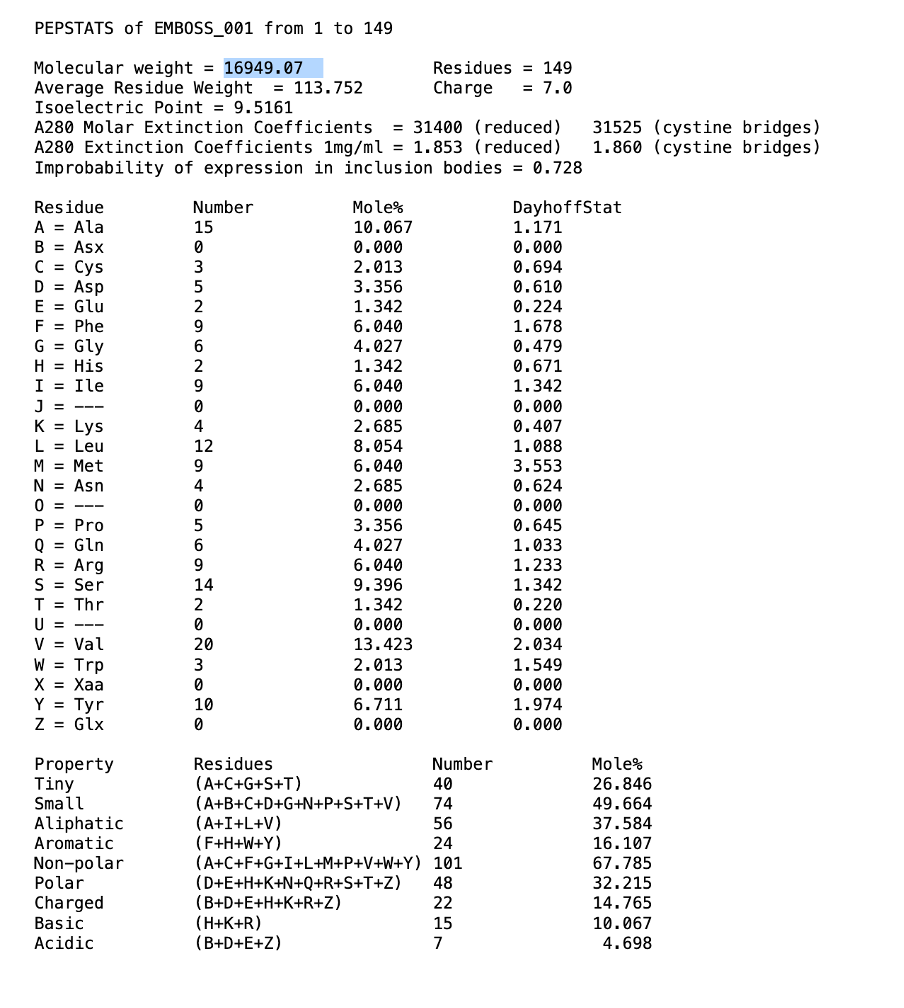
Transcribed Image Text:PEPSTATS of EMBOSS_001 from 1 to 149
Molecular weight = 16949.07
Average Residue Weight = 113.752
Isoelectric Point = 9.5161
A280 Molar Extinction Coefficients = 31400 (reduced) 31525 (cystine bridges)
A280 Extinction Coefficients 1mg/ml = 1.853 (reduced)
Improbability of expression in inclusion bodies = 0.728
1.860 (cystine bridges)
DayhoffStat
1.171
0.000
Residue
A = Ala
B = Asx
C = Cys
D = Asp
E = Glu
F = Phe
G = Gly
H = His
I = Ile
J = ---
K = Lys
L = Leu
M = Met
N = Asn
0 =
P = Pro
Q = Gln
R = Arg
S = Ser
T = Thr
U = ---
V = Val
W = Trp
X = Xaa
Y = Tyr
Z = Glx
Property
Tiny
Small
Aliphatic
Aromatic
Non-polar
Polar
Charged
Basic
Acidic
Number
15
0
3
5
2
9
6
2
9
0
4
12
9
STONONOMOGO
4
0
5
6
9
14
2
0
20
3
0
10
0
Residues
(A+C+G+S+T)
(A+I+L+V)
(F+H+W+Y)
Mole%
10.067
0.000
2.013
(B+D+E+H+K+R+Z)
3.356
1.342
6.040
4.027
1.342
6.040
0.000
2.685
Number
40
74
56
24
(A+C+F+G+I+L+M+P+V+W+Y) 101
48
22
15
7
(H+K+R)
8.054
6.040
2.685
0.000
3.356
(A+B+C+D+G+N+P+S+T+V)
(B+D+E+Z)
4.027
6.040
9.396
1.342
0.000
13.423
2.013
0.000
6.711
0.000
(D+E+H+K+N+Q+R+S+T+Z)
Residues = 149
Charge = 7.0
0.694
0.610
0.224
1.678
0.479
0.671
1.342
0.000
0.407
1.088
3.553
0.624
0.000
0.645
1.033
1.233
1.342
0.220
0.000
2.034
1.549
0.000
1.974
0.000
Mole%
26.846
49.664
37.584
16.107
67.785
32.215
14.765
10.067
4.698
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning