Describe the process involved in the hydrolysis reaction for carbohydrates, and name the organ that is responsible for the release of insulin and glucagon.
Describe the process involved in the hydrolysis reaction for carbohydrates, and name the organ that is responsible for the release of insulin and glucagon.
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter6: Metabolism
Section: Chapter Questions
Problem 4RQ: Energy is stored long-term in the bonds of and used short-term to perform work from a(n) ___________...
Related questions
Question
100%
Describe the process involved in the hydrolysis reaction for carbohydrates, and name the organ that is responsible for the release of insulin and glucagon.
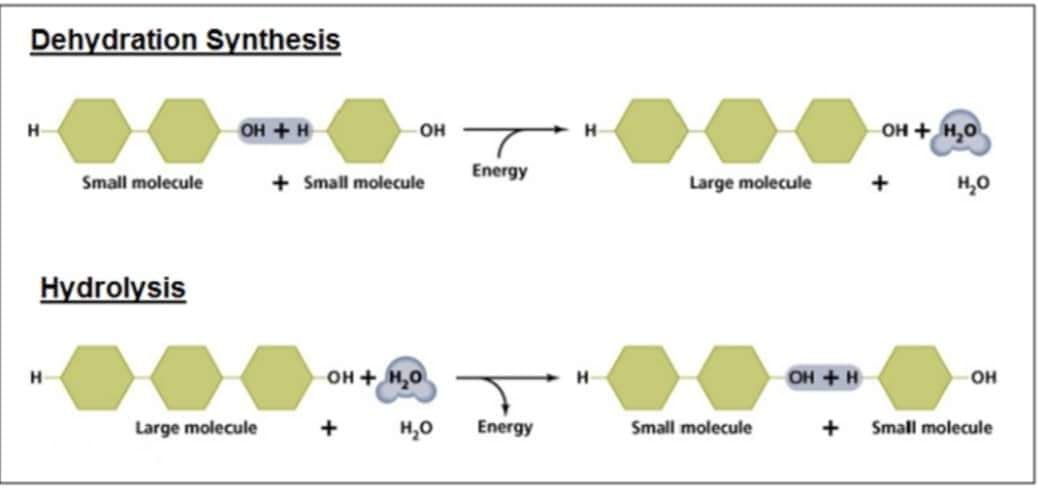
Transcribed Image Text:Dehydration Synthesis
H
OH + H
Small molecule
Hydrolysis
Large molecule
OH
+ Small molecule
OH + H₂O
+
H₂O
T
Energy
Energy
H
H
Large molecule
Small molecule
OH + H
OH + H₂O
+
но
OH
+ Small molecule
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning