Drag and drop orbitals and electron counts to complete the electron configuration of in Be sure to arrange the subshells in order of energy, with the lowest-energy subshell at highest-energy subshell at the right. Drag the appropriate labels to their respective targets. • View Available Hint(s) Reset| 1 2 4 5 6. 7 8 9. 10 1s 2s 3s 4. 5s 2p 3p 4p 5p 3d 4d G1 G1 G1 G1 G1 G1 G1 G2 G2 G2 G2 G2 G2 G2
Drag and drop orbitals and electron counts to complete the electron configuration of in Be sure to arrange the subshells in order of energy, with the lowest-energy subshell at highest-energy subshell at the right. Drag the appropriate labels to their respective targets. • View Available Hint(s) Reset| 1 2 4 5 6. 7 8 9. 10 1s 2s 3s 4. 5s 2p 3p 4p 5p 3d 4d G1 G1 G1 G1 G1 G1 G1 G2 G2 G2 G2 G2 G2 G2
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter11: Modern Atomic Theory
Section: Chapter Questions
Problem 115AP
Related questions
Question
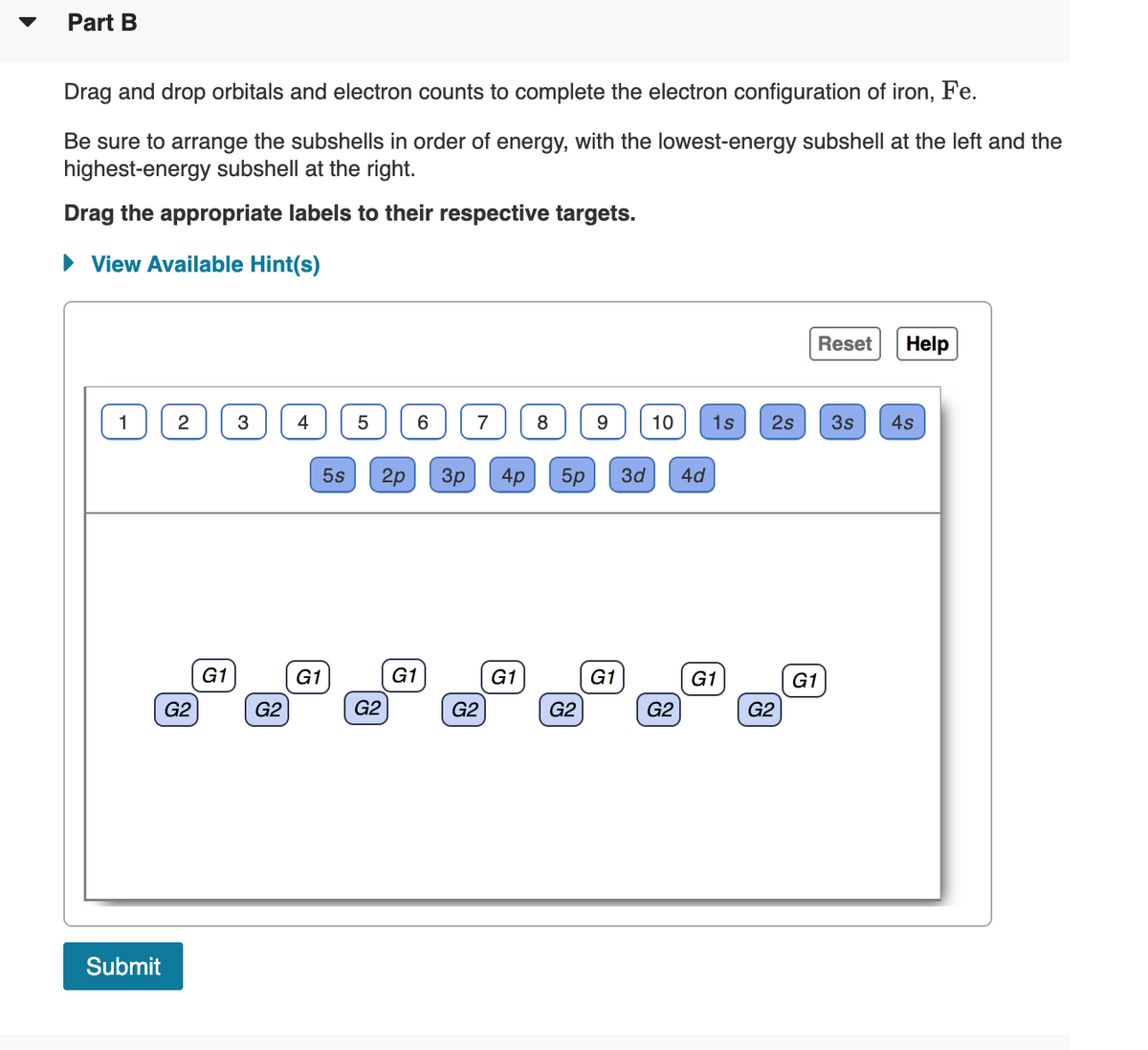
Transcribed Image Text:Part B
Drag and drop orbitals and electron counts to complete the electron configuration of iron, Fe.
Be sure to arrange the subshells in order of energy, with the lowest-energy subshell at the left and the
highest-energy subshell at the right.
Drag the appropriate labels to their respective targets.
• View Available Hint(s)
Reset
Help
4
6.
8
10
1s
2s
3s
4s
5s
2p
Зр
4p
5p
3d
4d
G1
G1
G1
G1
G1
G1
G1
G2
G2
G2
G2
G2
G2
G2
Submit
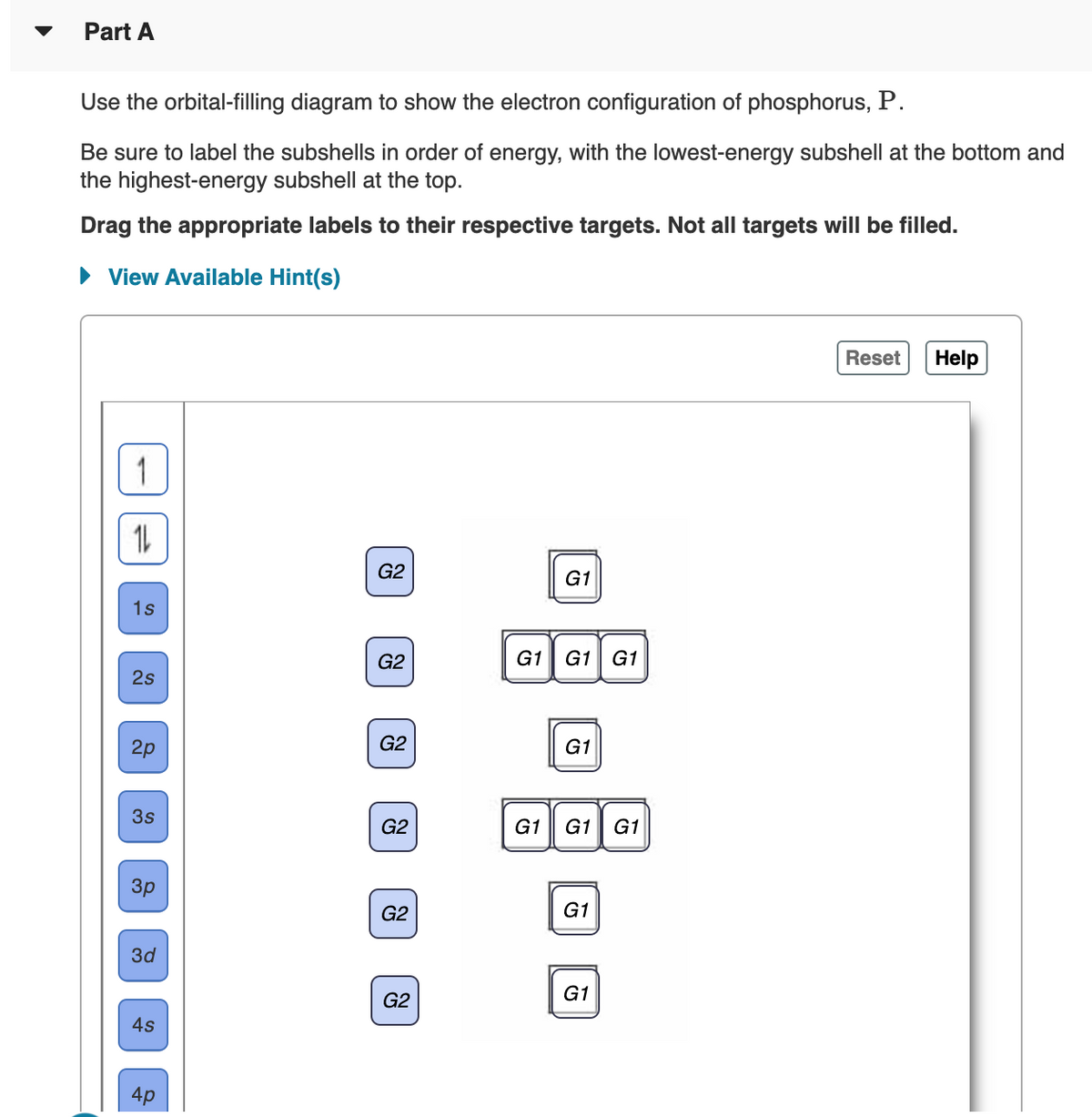
Transcribed Image Text:Part A
Use the orbital-filling diagram to show the electron configuration of phosphorus, P.
Be sure to label the subshells in order of energy, with the lowest-energy subshell at the bottom and
the highest-energy subshell at the top.
Drag the appropriate labels to their respective targets. Not all targets will be filled.
• View Available Hint(s)
Reset
Help
| 1
G2
G1
1s
G2
G1
G1 G1
2s
2p
G2
G1
3s
G2
G1
G1
G1
Зр
G2
G1
3d
G2
G1
4s
4p
1L
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
Transcribed Image Text:W History Bookmarks Profiles Tab Window
sity and Sig x
eringchemistry.com/myct/itemView?assignment
late them to electron
cupation of orbitals by electrons
has two electrons in the 1s
ation of He is 1s².
writing the electron configuration
minimize the energy of
orbitals fill before higher-
following order:
5p 6s 4f 5d 6p 7s 5f 6d.
principle, orbitals can hold
and when two electrons
st have opposing spins.
-bitals of identical energy
Died singly with parallel
E
MasteringChemistry: Module 8 X
O
80
C
2
R
Q
F
V
%
5
T
Enter your payment details | ba X
ProblemID=201341795
G
11
2s
2p
3s
3p
3d
4s
Help
Use the orbital-filling diagram to show the electron configuration of phosphorus, P.
Be sure to label the subshells in order of energy, with the lowest-energy subshell at the bottom and the highest-energy subshell at the top.
Drag the appropriate labels to their respective targets. Not all targets will be filled.
View Available Hint(s)
4p
6
B
F6
G1
Y
G2
H
G2
G2
MacBook Air
G2
G2
&
N
G1
G1 G1 || G1
F7
+
G1
G1 G1 G1
G1
14EEBBBBBB
7
8
U
G1
J
DII
F8
I
M
(
9
Reset
K
Help
O
)
(Co
O
L
7
EX
F10
Ơ
Tue Mar 14 6:08 PM
P
* 0
Review I Constants I Periodic Table
+
Update:
6 of 19
{
[
F12
>
}
:
1
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning