Based upon the number of spherical and angular nodes, label the orbitals below as 1s, 2p, etc. Specify the value of n and I for each: This is a orbital This is a orbital This is a orbital n = and I= and I= n 3= and I = In = Submit Answer 5 question attempts remaining
Based upon the number of spherical and angular nodes, label the orbitals below as 1s, 2p, etc. Specify the value of n and I for each: This is a orbital This is a orbital This is a orbital n = and I= and I= n 3= and I = In = Submit Answer 5 question attempts remaining
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter15: Introduction To Electronic Spectroscopy And Structure
Section: Chapter Questions
Problem 15.9E: List the possible values of L, ML, S, MS, J, and MJ for the following: a two coupled p electrons in...
Related questions
Question
Transcribed Image Text:uick Prep
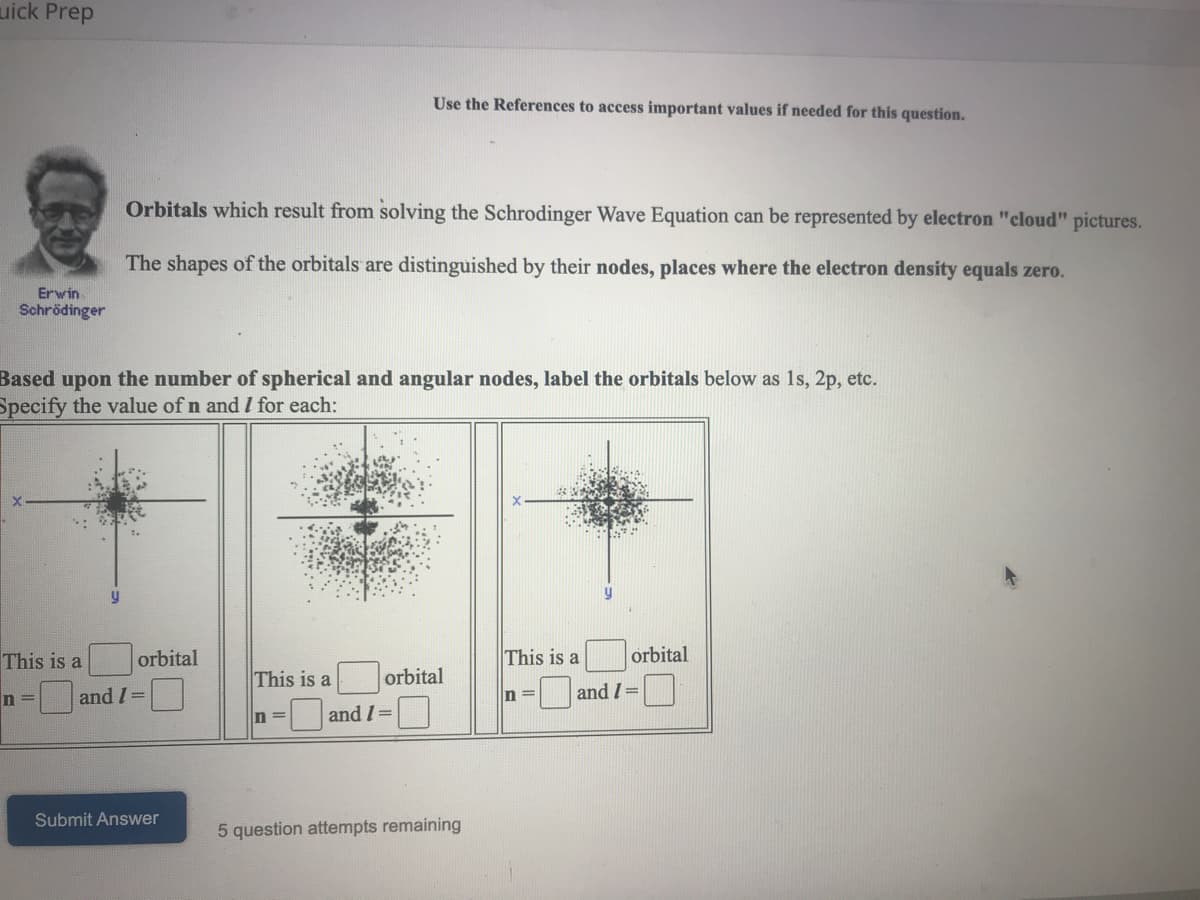
Use the References to access important values if needed for this question.
Orbitals which result from solving the Schrodinger Wave Equation can be represented by electron "cloud" pictures.
The shapes of the orbitals are distinguished by their nodes, places where the electron density equals zero.
Erwin
Schrödinger
Based upon the number of spherical and angular nodes, label the orbitals below as 1s, 2p, etc.
Specify the value of n and I for each:
y
This is a
orbital
This is a
orbital
This is a
orbital
and I =
and I =
In3D
n =
n =
and I =
Submit Answer
5 question attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning