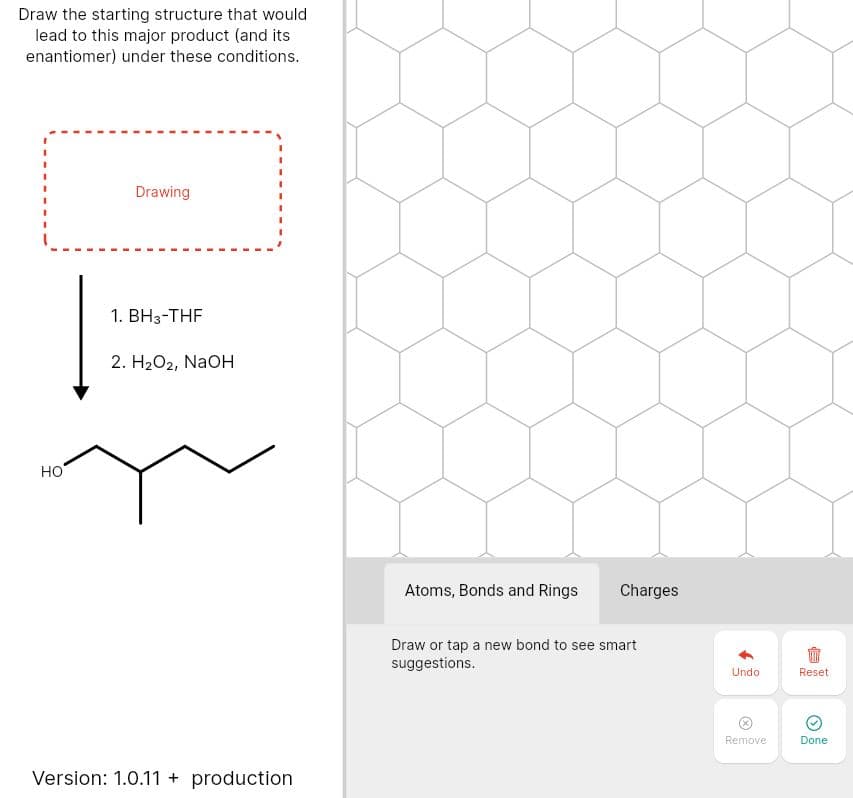
Draw the starting structure that would lead to this major product (and its enantiomer) under these conditions. Drawing 1. ВН-THF 2. H2O2, NaoH но
Q: Draw all stereoisomers formed in each reaction. Cl2 Br2 Br2 b. с. CHg а.
A: Stereoisomers are geometrical isomers with same molecular formula and connectivity but different…
Q: Draw the products of each SN1 reaction and indicate the stereochemistry of any stereogenic centers.…
A: Nucleophilic substitution reactions are those reactions in which one nucleophile substitute another.…
Q: Which of the following carbocations would you expect to rearrange? CH3 CH3 CH3 CH3CHCHCH3 + A B C O…
A:
Q: Answer the following questions by circling the most correct answ owing .. The relationship between…
A:
Q: Considering both 5-methyl-1,3-cyclopentadiene (A) and 7-methyl-1,3,5-cycloheptatriene (B), which…
A: Considering both 5-methyl-1,3-cyclopentadiene (A) and 7-methyl-1,3,5-cycloheptatriene (B), which…
Q: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary. CH3CH2 CH3…
A: In SN1 reactions at first a carbocation is formed from the substrate molecule. Then nucleophile can…
Q: Draw all stereoisomers formed in each reaction.
A: At the both end of the double bond, the anti-addition of halogen is occurred.
Q: Draw the structure of an alkyl bromide with molecular formula CgH13Br that fits each description:…
A:
Q: Which carbocation is more stable?H2C=C+H OR HC=C+ b HC= C+ OR CH3C+H2
A: 1) Since a carbocation which is present on a less electronegative element will be having more…
Q: Draw the product of each SN2 reaction and indicate stereochemistry. CH,CH C-Br + "OCH,CH3 b. a. +…
A: Alkyl halide compound shows substitution reaction because of highly reactive carbon and halogen bond…
Q: KMNO4, NaOH cold H2/ Pt(C)
A: The objective of the question is to write the product with proper mechanism.
Q: (a) What alkene yields A and B when it is treated with Br2 in CCl4? (b) What alkene yields C and D…
A: A and B product is obtained from the trans form of Phenyl-2-propene.…
Q: Draw the products of each reaction. Include all stereoisomers formed.
A: The product of given reaction can be drawn as
Q: Draw the two products A (which is the majority) and B that are obtained in the reaction. indicating…
A:
Q: 1) Provide the R/S and D/L designation for the following enantiomers: он но- CH,OH ČH,OH
A: Here we have to predict D,L and R,S configurations of the following compounds.
Q: Draw the products of each SN2 reaction and indicate the stereochemistry where appropriate. CH3…
A: Three substrates whose SN2 products are to be reported.
Q: Rank the following carbocations in order of increasing stability. CH2 CH3 CH3
A: The carbocation is basically a trivalent carbon whose hybridization is sp2. The higher the…
Q: 2. Arrange these carbocations in order of increasing stability. CH2 CH3 -CH3 C. b. a.
A: Stability of carbocation can be compared using hyperconjugation effect. Carbocations with having…
Q: ip the chair and draw the other chair conformer. Circle the more stable conformer and brlefly…
A: Interpretation: We are provided with reactants and we have to give missing products.
Q: C. NBS DMSO, H20 d. H20 H2SO4
A: In c part , alkene first react with NBS forms cyclobrominium cation then H2O will attack from…
Q: Taking into account anti periplanar geometry, predict the major E2 product formed from each starting…
A: Introduction : We have to tell the major eliminated product formed .
Q: Draw the products of each reaction and indicate the stereochemistry at any stereogenic centers.…
A: The product with stereochemistry at stereo genic centers are given below a) b) c)
Q: Rank the following carbocations from most stable to least stable: H H CH3 H CH, H3C H3C CH3 O A>C> B…
A:
Q: Draw the starting structure that would lead to this major product (and its enantiomer) under these…
A: We have to draw the starting structure for the following given product (and its enantiomer) as…
Q: 1. Now draw all possible stereomers for this compound, and label the relationships, and D or L on…
A: Stereoisomers have same molecular but differs in the spatial orientation.
Q: A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that…
A: Given data : A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. Each alkene…
Q: 4. Rank the following leaving groups from best to worst. OH2 -OH -CH3 -NH2 LG LG LG LG LG A B C D E
A: Ability to leave of a leaving group depends upon: Electronegativity of the leaving group: A high…
Q: Draw the starting structure that would lead to this major product (and its enantiomer) under these…
A:
Q: H2 OH H2SO, Pd/C K 7. Major product only Only one stereoisomer Stereochemstry required NaBHa, MEOH…
A:
Q: Draw the starting structure that would lead to this major product (and its enantiomer) under these…
A:
Q: Question 4 What stereochemical and/or molecular requirement needs to be satisfied for an E2…
A:
Q: What is the major product for the following reaction sequence (MCPBA is a peracid RCO3H)? 1. МСРВА…
A: Alkene mainly gives electrophilic addition reaction. Being electron rich, an electrophile easily…
Q: Identify the secondary carbocation from the following: o (CH3),CHCH2* O CH3CH2* o (CH3),CH* O CH3*
A:
Q: Draw all stereoisomers formed in each reaction. а. Br2 b. Cl2 H2O
A: Answer of the question given below,
Q: Please Prouide the missing Feagents, NH2 Please Prouide the missing reagents. OH Br
A:
Q: Q8.4 Structure CH3 CH3 ВЕ .C -E D I II What substituent is positioned at B on Structure II? O -H O…
A: Equatorial conformation is more stable than axial confirmation. The stable chair conformation of the…
Q: 9) Draw the conformer structure, Newman projection, before elimination and the elimination product…
A: Due to strong base and 3° C it gives E2 elemination reaction
Q: What stereoisomers are obtained from the following reaction?
A: Since in the presence of peroxide, the addition of HBr follows a free radical mechanism This is…
Q: (1) BH3 (2) H,O,, H,O, NaOH Product is (circle one) i. A racemic mixture ii. A single enantiomer,…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary.
A:
Q: NaNg DMSO Br LDA CH;CH2-Br, 80 °C
A: The reactions given are,
Q: What stereoisomers are obtained from the following reaction?
A: When alkene react with HBr that to in presence of peroxide then the substitution takes place…
Q: 2,2,5-Trimethyl-hex-3-yne is a(n) a.) terminal alkyne b.) internal unsymmetrical alkyne c.)…
A:
Q: 1. a) Draw the most stable chair conformation for each of the compounds shown below: Br X…
A: 1. We know, a conformation of a compound is the spatial arrangements of atoms which can be easily…
Q: 2. (a) (i) HO HO Convert each of the compounds above into its more stable chair form. Explain your…
A: Here we have to write the most stable chair conformation of the following cyclohexane compounds. In…
Q: Which structure is a meso compound? В. CH3 A. C. D. CH3 CH3 CH 3 H--C1 H--C1 H--C1 H--C1 Cl--CH3…
A:
Q: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary.
A: 1) Since in SN1 reaction, formation of carbocation takes place. And since the carbocation can be…
Q: 1. Which one of the following compounds has four stereocenters? A D 3. Which carbocation will…
A: 1) No one has 4 stereocentres. 3) A, B, C and E will rearrange.
Q: 1. Draw the major and minor products. Circle the major product. CH3 CH3-C=C-CH3 H* H20 H. a. C. b.…
A: Que-1 , Given reaction is hydration reaction in which water is added on alkene to give alcohol as…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- An inexperienced student adds a solution of catalytic H2SO4 and water to the alkene shown below, expecting to produce 1-cyclobutylethan-1-ol. What major product is she surprised to isolate instead?What is the product (A, B, C, or D) of the reaction shown in Image30? a. C b.D c. B d. AShow each possible product for the reaction below and predict the major product. Be sure to include all geometric isomers. B) Draw the conformer that leads to the major product for the reaction above as a Newman projection. Show the mechanism for its formation. C) How would you change the reaction conditions to get the Hoffman product? include all parts pls
- Draw mechanism arrows and then predict the major products and stereochemistry. Also include the labels of thermodynamic and kinetic -propyne with 2 equivalences of br2 - no stick structure please.Identify the HOMO and LUMO in the first step of the reaction shown here:Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?
- Draw an approximate energy diagram withannotations for the rotamers of 1,2-dibromoethanefrom 0-180°. Give the Newman projections andnames of the most important rotamers.Why does the final product has the opposite configuration compared to the reactant? Shouldn’t it form OMs first, then OMs gets substituted by Cl- via Sn2 (the 1st inversion of configuration) then the Cl- gets substituted by OCH3- (the 2nd inversion of configuration? To my understanding 2 inversions = same configurationAssign the stereochemical configuration (E or Z) for the alkene below. Show your work, indicating clearly which groups are assigned high priority (e.g., through assigning the groups numbers, circling only the high priority groups, or labeling groups as high or low).

