Q: Required is 350 mL of 3/5 (v/v) alcohol in water solution. Perform the calculations needed to prepar...
A:
Q: Consider the same reaction carried out two more times, both at the same condition(including concentr...
A:
Q: Suppose a 0.48M aqueous solution of phosphoric acid (H,PO,) is prepared. Calculate the equilibrium m...
A: Given, Concentration of aqueous Phosphoric acid solution = 0.48 M Required, ...
Q: Here is a graph of the molarity of formic acid (HCO,H) in a reaction vessel during a certain chemica...
A: A question based on kinetics of reactions in solution that is to be accomplished.
Q: When sodium hydroxide, NaOH, is dissolved in water, what processes occur? Select all that apply Hydr...
A: The attraction forces between the different molecules are known as intermolecular forces. Different ...
Q: Proposed a mechanism on how to synthesize 2, 3 dimethylbutane
A: Given that Synthesis of 2, 3 dimethylbutane from propane
Q: Select the method that would be successful in producing the given product in good yield starting fro...
A:
Q: Describe the spontaneity and entropy of the processes. Sublimation of naphthalene balls as represen...
A:
Q: Give two examples of common radioisotopes used in medicine. Be sure to write the full name of the is...
A: In medical world radioisotopes used in various ways.Radioisotopes can occurred as naturally or artif...
Q: Construct a simulated 'H NMR spectrum for 1,1-dibromoethane. Drag and drop the appropriate splitting...
A: The 1H NMR analysis of 1,1-dibromoethane is given below
Q: Vinegar contains acetic acid (HC2H3O2), which is responsible for its acidity. In one analysis of a c...
A:
Q: Elucidate the bonding of the following complexes according to the valence-bond theory approach: (a) ...
A:
Q: A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 4....
A: Here we have to determine the final temperature of bath containing 4.5Kg of water at 35.3°C if it ab...
Q: Give the ratio of tetrahedral sites to oxygen in tetrahedral coordination. • Alkali feldspar • Pyrox...
A:
Q: partially hydrogenated vegetable oil
A: In this question we have to explain the equation for baeyers , bromine and sulfuric acid, and flamma...
Q: SIMULATION Equilibria and Volume I N204(g) 2 2 NO2(g) Equilibrium Concentration K = 0.880 NO2 0.339 ...
A:
Q: What is the pH of 50.00 mL buffer solution which is 2.00M of HC2H3O2 and NaC2H3O2 each? (Ka=1.8x 10-...
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The buffe...
Q: For the reaction below: 2 NO(g) + 2 H2(g) → N2(g) + 2 H2O(g) What is the reaction constant (Q) if...
A:
Q: NaHCO, can be used to neutralize excess acid in situations such as, for instance, acid spills in the...
A: Given, NaHCO3 can be used to neutralize excess acid Volume of nitric acid = 75.5 mL Concentration o...
Q: A 0.04M solution was found to be 1.6% ionized. what is the concentration of the conjugate base?What ...
A:
Q: 7) Which of the following structures represent D-glucose (Circle all that apply)? Но но HO OH HO D-g...
A: Given-
Q: Give the nuclear symbol (isotope symbol) for the isotope of bromine, Br, that contains 46 neutrons p...
A: an atom consist of three subatomic particle proton, neutron, electron here we are required to find t...
Q: Analysis of sphalerite using X-ray Fluorescence technique is shown in Table 1. Sphalerite may be nea...
A: A question based on molecules that is to be accomplished.
Q: 1.Write the balanced, net ionic equation for the reaction occurring when (a) Cl is added to a soluti...
A: The ionic reactions are given below
Q: AU
A:
Q: UESTION 6 Which of the following may be possible structures for malic acid, C4H6O5? There may be mor...
A: The mentioned compound contains five oxygen atoms in the form of carboxylic and hydroxyl group.
Q: Name the following compound. Inlcude cis/trans, R and S configuration if needed.
A: Name of given compound is:
Q: what is the maximum % recovery of TNT?
A: At low temperature- 100g ethanol : 0.70 g TNT At low temperature: 100 g ethanol : 18.58 g TNT Now,...
Q: Which of the following pairs of compounds are labeled correctly. . enantiomers enantiomers CI enanti...
A:
Q: A solution is prepared by dissolving 370 g of sucrose (C12H22011) in 642 g of water. What is the vap...
A:
Q: 10. For the Isotope 6731Ga A. What is the atomic number? B. What is the mass number? C. What is the ...
A: The answer to the following question :
Q: According to Riippi et al. (1998), briefly discuss the effects of increased compression force to the...
A: As per the guideline, since you have posted a question with multiple sub-parts, we have solved the f...
Q: Using the above pattern, identify the circled electrons using the four quantum numbers. 回回画 [1 March...
A: There are four set of quantum numbers which describes or gives all the information about an electron...
Q: Enter electrons as e¯. The following skeletal oxidation-reduction reaction occurs under basic condit...
A: The basic steps to balance redox reaction in basic medium are- 1. Find oxidation no of each atom and...
Q: Predict how many 'H NMR NMR signals (individual resonances, not counting splitting) are expected for...
A: Here, we have to find the number of 1HNMR signals that are expected for the given compound.
Q: Eheat
A:
Q: A chemist prepares a solution of mercury(I) chloride (Hg,Cl,) by measuring out 0.347 mg of mercury(I...
A: Molarity is a concentration term for a solution. The molarity of a given solution is defined as the ...
Q: acid, base, conjugate
A:
Q: Q Curved arrows are used to illustrate the flow of electrons. Using the provided starting and produc...
A:
Q: Consider three C,H,Cl, isomers and three sets of 'H NMR data that one would expect on application of...
A:
Q: For this reason there has been a great deal of international discussion about whether to regulate th...
A: (A) When decane, C10H22 is burnt with air , then Carbon dioxide gas and water vapour are produced. T...
Q: Calculate the specific heat capacity of a new alloy if a 15.4 g sample absorbs 393 J when it is Heat...
A: The heat capacity of the alloy can be calculated as
Q: Consider the same reaction taking place in three vessels. The vessels all contain the same amount of...
A: Rate of reaction
Q: How many milliliters of 0.491 MHNO3 contain 3.53 g HNO3? mL
A:
Q: For each of the following pairs or reactions, which is correctly labeled as having the larger equili...
A: Here we have to predict which acid- base reaction's equilibrium constant is larger in the followin...
Q: A reverse osmosis desalination plant treats 20 MGD (million gallons per day) brackish water. The wat...
A:
Q: cientists is correctly identified? There may be more than one correct answer. Adolf von Baeyer...
A:
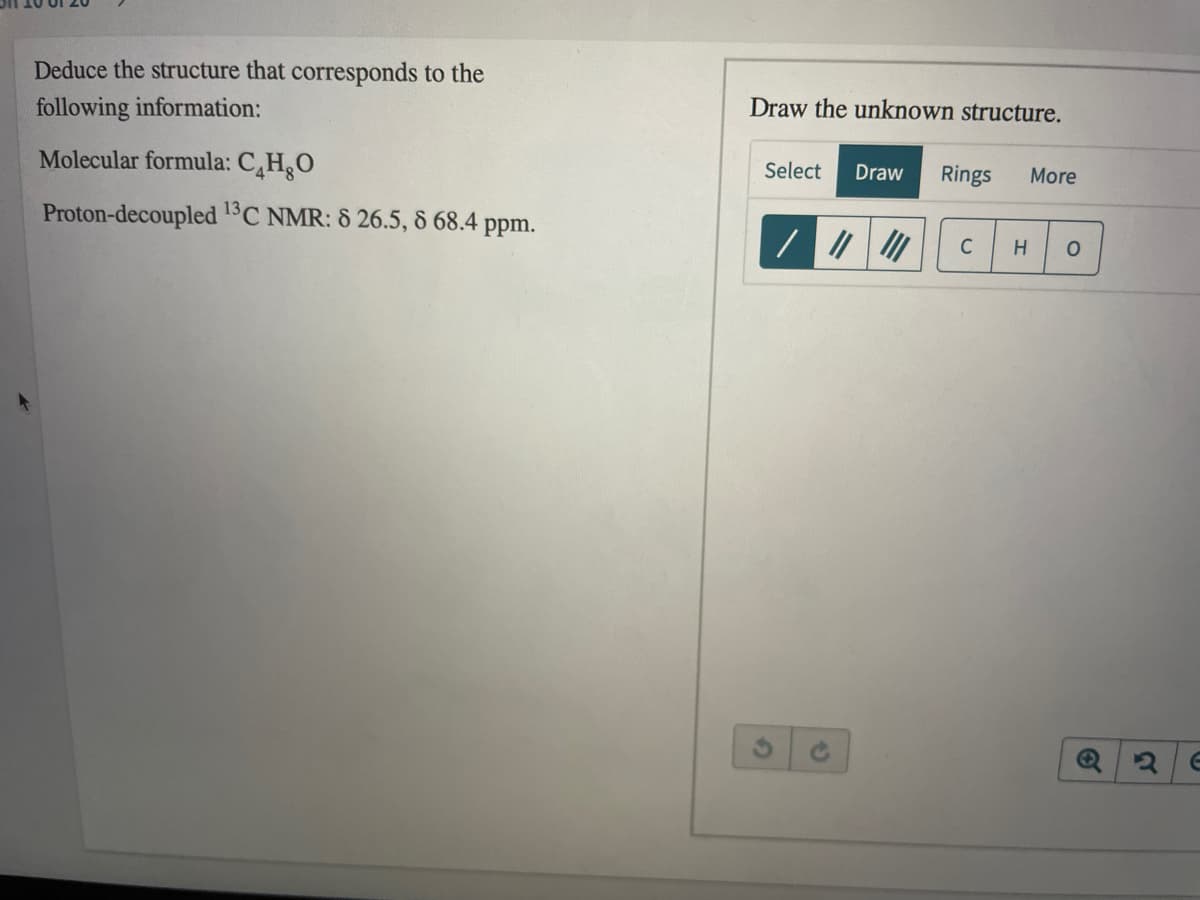
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum: C,H,F,I:...
A: The analysis of the results obtained from proton NMR, 13C-NMR, and mass spectra such as HRMS, LCMS p...
Q: Entropy Change Suppose we have 1.50kg of water at a temperature of 0 degree Celsius. We put the wate...
A: Mass of water = 1.50kg or 1500 g Temperature = 0°C or 273.15 K
Q: Draw the reactants that would react to form this cyclohexene derivative in a Diels-Alder reaction. I...
A: Diels Alder reaction is the cycloaddition reaction between diene and dienophile. This is also known ...
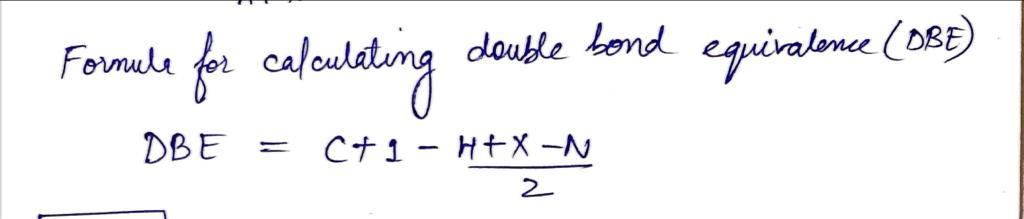
Step by step
Solved in 2 steps with 2 images

- A 13C NMR spectrum of commercially available 2,4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8, and 68.1 . Explain.Show the 13C NMR for the following compound and Explain each peak for formed in detail.what is is the structure of a compound of molecular formula c 10 h 14 o 2 that shows a strong ir absorption at 3150-2850 cm − 1 and give the following 1 h nmr absorptions: 1.4 (triplet, 6 h), 4.0 (quarter, 4h), and 6.8 (singlet, 4h) ppm?
- 1) Propose the structures for the following 1H and 13C NMR spectrum. b) Molecular Formula: C8H8O3 1H-NMR, CDCl3 Solvent, Molecular Formula: C8H8O3 13C-NMR, CDCl3 Solvent, Molecular Formula: C8H8O3 in the picturesA compound of molecular formula C4H8O2 shows no IR peaks at 3600–3200 or 1700 cm-1. It exhibits one singlet in its 1H NMR spectrum at 3.69 ppm, and one line in its 13C NMR spectrum at 67 ppm. What is the structure of this unknown?You have a sample of a compound of molecular formula C11H15NO2, which has a benzene ring substituted by two groups, (CH3)2N− and −CO2CH2CH3, and exhibits the given 13C NMR. What disubstituted benzene isomer corresponds to these 13C data?
- You have a sample of a compound of molecular formula C11H15NO2, which has a benzene ring substituted by two groups, (CH3)2N – and – CO2CH2CH3, and exhibits the given 13C NMR. What disubstituted benzene isomer corresponds to these 13C data?Propose a structure for an unknown compound whole molecular formula is C(5)H(10)O(2) and is consistent with the 1H NMR data listed. doublet, at 1.23 ppm (6H) singlet, at 2.10 ppm (3H) septet, at 4.98 ppm (1H)Predict the structure of the molecule with molecular formula C8 H10 O using given information about its 1H NMR, 13C NMR, and IR.
- Select the unknown with the following information from 1H-NMR, IR and MS.Draw the structure of the ether that has a chemical formula C8H10O2, has a molecular ion peak at 138, and is represented by the IR, 'H NMR, and 13 C NMR belowTreatment of butan-2-one (CH3COCH2CH3) with strong base followed byCH3I forms a compound Q, which gives a molecular ion in its massspectrum at 86. The IR (> 1500 cm−1 only) and 1H NMR spectra of Q aregiven below. What is the structure of Q?

