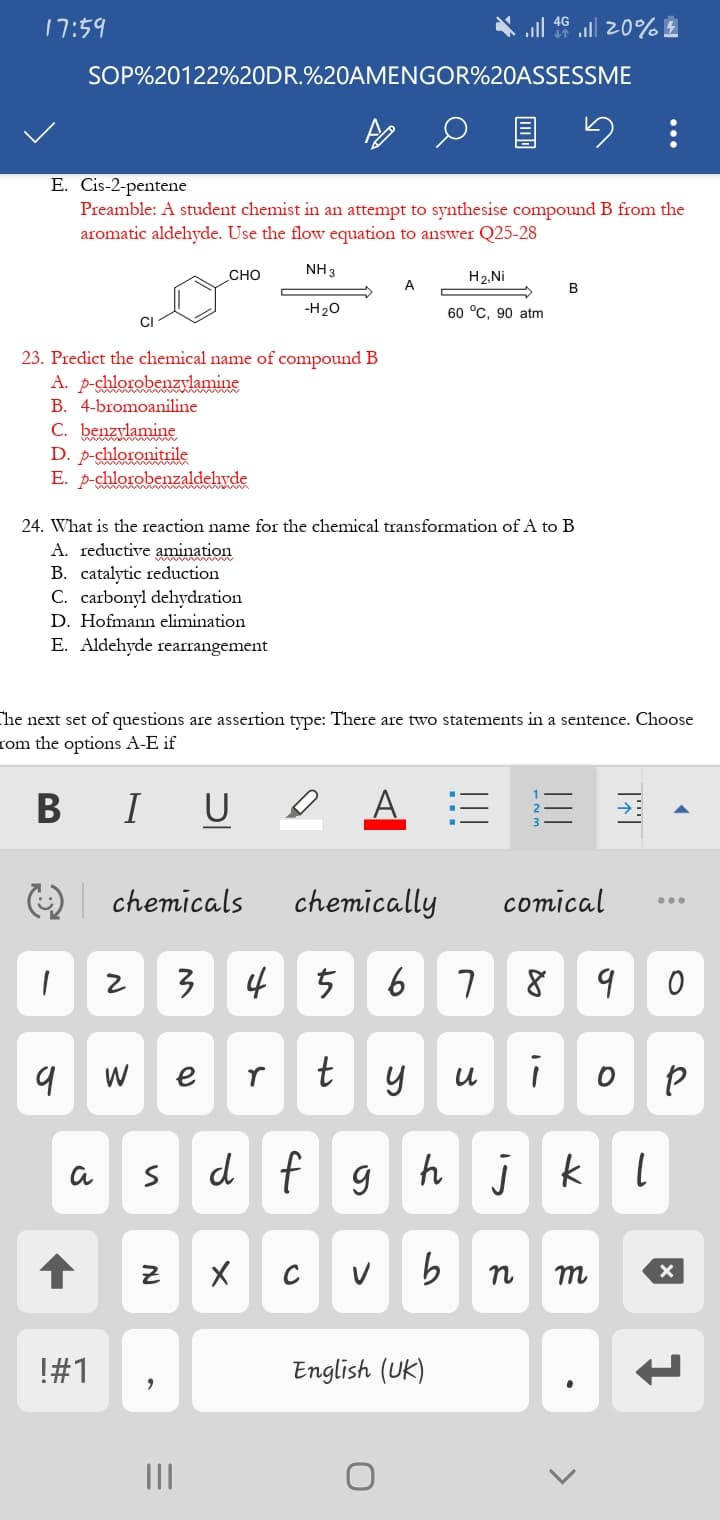
E. Cis-2-pentene Preamble: A student chemist in an attempt to synthesise compound B from the aromatic aldehyde. Use the flow equation to answer Q25-28 сно NH 3 H2, Ni A -H20 60 °C, 90 atm 23. Predict the chemical name of compound B A. p-chlorobenzslamine B. 4-bromoaniline C. benzylamine D. p-chloronitrile E. p-chlorobenzaldehvde mam
Q: 7H5O2 = 160.21 C7H8O2 = 122 Density: C7H8 = 0.87 g/mL KMnO4 = 2.7 g/mL KC7H5O2 = 1.5 g/mL…
A: The solution is as follows:
Q: Extraction (Part 2) Experiment: Separation of Neutral and basic Substances NH2 Handle aromatic…
A: The above experiment is separation of basic and neutral compounds.
Q: (a) How will you carry out the following conversions?(i) Acetylene to Acetic acid (ii) Toluene to…
A: In organic chemistry conversions play an important role to convert one functional group into another…
Q: Explain the following trend: Base strength when reacting with BME3 (in gas phase): 4-methylpyridne >…
A:
Q: Based on the procedure given below, answer the following questions about the aldol condensation…
A: Given that
Q: event is not an oxidation reaction? A. Product formation as a result of the reaction of benzoine…
A: Answer The event is not an oxidation reaction All events are…
Q: 4.85 deche 7.5 7.0 2.04 2.05 2.93 5.0 4.5 4.0 3.5 3.0 2.5 2.0 (c)
A:
Q: Pease indicate the compound that best fits in the blank of the separation scheme below. OH HO. H2N'…
A:
Q: Preamble: A student chemist in an attempt to synthesise compound B from the aromatic aldehyde. Use…
A: Since we have partial positive charge on the carbon making double bond with O in aldehyde and there…
Q: "One-pot" DCC (1.5 equiv) CH2CI2 (2mL) rt, 2h CF3CH,OH (4mL) Phl(OAc)2 (1.0 equiv) 5 minutes OH B Но…
A:
Q: Explain each of the following true statements briefly: 1. Hexane (C6H14, a hydrocarbon) is not a…
A: Nucleophile is a rich electron species that contains a lone pair of electrons and can donate to a…
Q: At what position, and on what ring, is nitration of phenyl benzoate MOST likely to occur? Give the…
A: Answer:- This question is answered by using the simple concept of directive effect of substituents…
Q: OH OH H2N Benzoie Acid Biphenyl Benzocaine Phenol benzocaine phenol biphenyl benzoic acid | dissolve…
A: The organic compounds present in the mixture are benzocaine, phenol, biphenyl and benzoic acid.
Q: . In the synthesis of 1-bromobutane, what is the inorganic by-product left in the reaction flask…
A: Explained as follows
Q: Water disinfection by halogens may not produce the following by-product: a. Chloramine (NH2CI) b.…
A:
Q: Pease indicate the compound that best fits in the blank of the separation scheme below. 0000 OH OH…
A: The organic compounds present in the mixture are benzocaine, phenol, biphenyl and benzoic acid.
Q: Is any other acid good for catalyze nitration to aromatic ? The textbook use sulfuric acid as…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: 3. Consider the following carboxylic acid (propanoic acid) and answer the question that follows:…
A: Since you have posted a question with multiple subparts, we will solve first three subparts for you.…
Q: Succinic anhydride + NH3 ->
A: Succinic anhydride reacts with NH3 to form 4-amono-4-oxobutanoic acid.
Q: N' 'N' CH3 CH3 II
A: I is more favorable enamine
Q: A chemist wanted to synthesize the anesthetic 2-ethoxy-2-methylpropane. He used ethoxide ion and…
A:
Q: Preamble: A student chemist in an attempt to synthesise compound B from the aromatic aldehyde. Use…
A: Since in the first step of reaction, we have aldehyde which has C making double bond with O having…
Q: explain some of the hazards (name at least three) involved in the Friedel-Crafts Acylation:…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid…
A:
Q: A. Compute the amount of reagents which will be used for the preparation of the following reagents.…
A: Hello. Since more than one question has been asked and the first question contains more than three…
Q: CH₂CH₂CH₂CH₂Br CH,CH,O Na* CH,CH,ON=" ? CH₂CH₂C(CH₂)BrCH, ? yatab aayilovoetanol/S5°C hang ilvesboli…
A: Hydrocarbons having halogen group such as bromine is known as alkyl bromide. These compounds are…
Q: The synthesis of acid orange 7 is shown below. Please answer the following questions. 8 7. 1 2 OH…
A: OH is an electron rich group.
Q: Question 4
A: Nucleophiles are those species which act as electron rich. These species offer electrons during any…
Q: Preamble: A student chemist in an attempt to synthesise compound B from the aromatic aldehyde. Use…
A: Since in step 1 of the reaction, we have an aldehyde and ammonia reacting. And ammonia has N with…
Q: is this a one to one reaction? what does the chemical equation look like? Preparation of Acetanilide…
A: one to one reaction is that when from one reactant, one product is formed.
Q: a) Br b) OH c) d) Br. Br e) NC
A: Since you have posted a question with multiple subparts, we will solve first three subparts for you.…
Q: Which of the following compounds would be the most soluble in H0 during extraction purification? A.…
A: ✓It has been found that {\rm{p}}{{\rm{K}}_{\rm{a}}}pKa value of 2- chlorobutanoic acid is lower…
Q: ROVIOW Topics References] 1. NH3 CI NaOH NH2 Naci H20 to 2. OH NaHCO, 12 H20 CO2 Nal Proton transfer…
A: Applying mechanism of reaction it is determined that which type of reaction is taking place.
Q: Starting from benzene, please examine the synthetic routes and choose the option that will lead to…
A: Friedel craft alkylation involves electrophilic substitution of alkyl groups on aromatic rings when…
Q: compounds E and F from the group of compounds below and asked to identify them according to their…
A: The question is based on the concept of qualitative analysis. We have to distinguish between the…
Q: I. Explain in your own words any mistake in the following given. The same construct of sentences…
A:
Q: You are planning to carry out a reaction between propyne, CH3C≡CH and sodium amide, NaNH2. You also…
A: The reactants given are CH3C≡CH and NaNH2.
Q: a. Mix 1 mL of glacial acetic acid, 1 mL of ethyl alcohol and 3 drops of concentrated sulfuric acid…
A:
Q: 2. The hydrolysis of a nitrile under acidic or basic conditions to form a carboxylic acid parent…
A: 2. a. We have to tell why the reaction in the scheme (1) is easily done under mild conditions and…
Q: Vanillin, NaI/NaOCL, ethanol/H2O, bleach are reacted in an electrophilic aromatic substitution…
A: NaOCl (Sodium hypochlorite) oxidises the NaI to gives I2 molecule, a source of iodonium ion. In…
Q: General procedure A mixture of 4-hydroxycoumarin (1 mmol), aromatic aldehyde (1 mmol), methyl…
A:
Q: Synthesis of 1-bromobutane from 1-butanol (11 mL) – N-butanol (7.4 mL) – Sulfuric acid Give the…
A:
Q: 8. As mentioned in exercise 5, carboxamides are compounds used for cancer treatments, and studies of…
A: Here we have to write the intermediate products formed during the given sequence of reactions.
Q: Preamble: A student chemist in an attempt to synthesise compound B from the aromatic aldehyde. Use…
A: In the given equation, we have an aldehyde which has partial +ve charge on carbon attached to the…
Q: tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid…
A:
Q: Mechanism of the formation of the electrophilic nitrosonium ion
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: 1.- Why are carbon nanotubes and graphene currently considered as reinforcing additives for…
A: Since you have asked multiple questions, we will solve first question for you. If you want any…
Q: b) Fill in the gaps by drawing the structures of the missing reagents, intermediates and/or products…
A: The electrophilic substitution reaction is the significant reaction of aromatic benzene ring which…
Question 23 pls
Step by step
Solved in 3 steps with 2 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4AI(C2H5)3 →3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d = 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3): d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257 L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?(a) How will you carry out the following conversions?(i) Acetylene to Acetic acid (ii) Toluene to m-nitrobenzoic acid(iii) Ethanol to Acetone(b) Give reasons :(i) Chloroacetic acid is stronger than acetic acid.(ii) pH of reaction should be carefully controlled while preparing ammonia derivatives of carbonyl compounds.
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…
- tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate Typically yield 6.34g How would I calculate theoretical and percent yield for the reaction ? Please and thank you!tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate 1 (CH3)COH + 1 H3O^+ Cl^- -> 1 (CH3)3CCl + 2 H2O Typically yield 6.34g Why is the percent yield be lower than expected? 53.9% .. Is it due to evaporation? Is it not the major product? If this reaction was with 2-butanol/HCl instead of tert butanol, how would I obtain the products/mechanism, and determine major products? Thank you!!tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate 1 (CH3)COH + 1 H3O^+ Cl^- -> 1 (CH3)3CCl + 2 H2O Typically yield 6.34g How would I calculate theoretical and percent yield for the reaction ? Please and thank you!
- tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate 1 (CH3)COH + 1 H3O^+ Cl^- -> 1 (CH3)3CCl + 2 H2O Typically yield 6.34g Why is the percent yield be lower than expected? 53.9% .. Is it due to evaporation? What is the major product of this reaction? isobutene or t-butylchloride? If this reaction was with 2-butanol/HCl instead of tert butanol, how would I obtain the products/mechanism (SN2?), and determine major products? If sn2, why? Thank you!! This was…Benzene, C6H6 undergoes substitution reaction with concentrated nitricacid, HNO3 to produce compound L. The reaction of compound L withbromine, Br2 in the presence of iron tribromide, FeBr3 produced compoundM. Benzene also undergoes Fridel-crafts alkylation reaction withchloroethane, CH3CH2Cl using catalyst N to produce compound P (i) Draw the structural formula of L, M and P (ii) State catalyst N. (iii) Show the formation of electrophile that will be reacted with benzene for theformation of compound P.Doxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is synthesized in the following series of reactions starting with ethyl 2-oxocyclopentanecarboxylate. Except for the Nef reaction in Step 8, we have seen examples of all other types of reactions involved in this synthesis. Q. Write equations to show that Step 6 can be brought about using either methanol or diazomethane (CH2N2) as a source of the -CH3 in the methyl ester.



