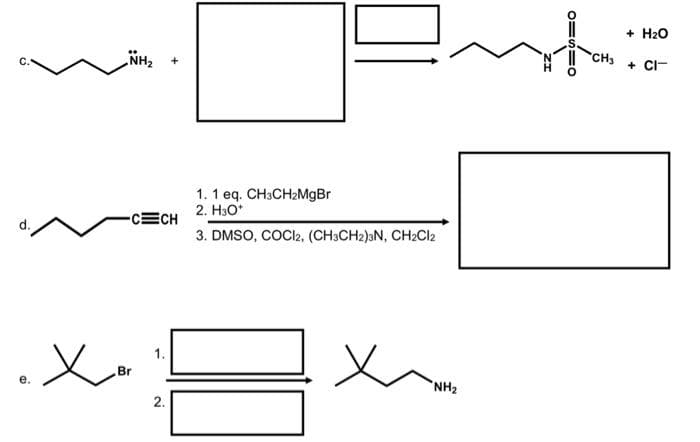
Q: This is a "retrosynthesis" problem. Identify the TWO MOLECULES necessary to prepare th amine shown…
A: The question is related to retrosynthesis.Retrosynthesis is a synthetic process used to synthesize…
Q: For each organic compound in the table below, enter the locant of the highlighted side chain. CH3 |…
A:
Q: Write the common (not systematic) name of each organic molecule. Hint: your answer should have more…
A: The question is based on the concept of organic chemistry.We need to write common name of given…
Q: Calculate the amount of solute needed to prepare the following solutions. Express your answer using…
A:
Q: draw a structural formula for the ketal formed when twomolecules of ethanol combined with 2-butanone…
A: When a ketone reacts with two molecules of an alcohol, a ketal will be formed. The reaction needs an…
Q: What volume of oxygen gas is produced when 0.682 mol of hydrogen peroxide (H202) reacts completely…
A: For the given reaction,Temperature, T = = (0 + 273.15) K…
Q: Draw the zwitterion of Leucine. CH3 CH₂ CH CH₂ Draw the zwitterion of Leucine.
A: Amino acids contain carboxylic acid and an amine functional group on the same carbon atom. Amino…
Q: Gaseous butane (CH₂(CH₂)₂CH3) reacts with gaseous oxygen gas (0₂) to produce gaseous carbon dioxide…
A: The given mass of butane = 31.4 gThe given mass of oxygen = 151 gThe theoretical yield of carbon…
Q: What is the IUPAC name for the following compound? CH3CHCH2 C-H Br
A:
Q: A 7.00 L tank at 18.3 °C is filled with 17.0 g of sulfur tetrafluoride gas and 15.2 g of chlorine…
A: According to the question,The mass of the chlorine pentafluoride (ClF5) gas is given by = 15.2 gThe…
Q: Use Le Chatelier’s principle to determine whether the equilibrium for the exothermic reaction below…
A: The given chemical reaction is N2(g) + 3H2(g) → 2NH3(l).We have to determine the equilibrium…
Q: The standard reaction free energy AGO = 1100. kJ for this reaction: 4 PC13(g) →P4(g) + 6 Cl₂(g) Use…
A: The given reaction is, 4 PCl3 (g) ----> P4 (g) + 6 Cl2 (g)…
Q: Draw the Lewis Structures (with formal charges) of each of the species in the indicated pair and use…
A: Order of bond length:Single bond > double bond > triple bondSo, single bonds are longer than…
Q: What is the relationship between the following compounds? CH3 H H C=C Br and H CH 3 CFC Br
A: The question is based on the concept of isomersim.We need to identify the relation between given…
Q: Calculate the pH for each case in the titration of 50.0 mL of 0.180 M HClO(aq) with 0.180 M KOH(aq).…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: Read the directions and create the graph described below. You will then click the submit button and…
A: First fill up the given values in the computer program excel and then draw the graph using a scatter…
Q: Part A The specific heat capacity of liquid water is 4.18 J/g-K. How many joules of heat are needed…
A: Specific heat tells us how much heat is needed to increase the temperature of 1 g of that substance…
Q: where did the x 10-6 come from on the 12.8 concentration value when it wasn't in the original…
A: Question :Calculate the extinction coefficient of each peak if the measurements were made for a…
Q: What volume in milliliters of a 0.111 M NaOH solution is required to reach the equivalence point in…
A: The balanced chemical equation is H2SO4(aq) + 2NaOH(aq) Na2SO4(aq) + 2H2O(l)…
Q: How many ¹H NMR signals (not peaks) would you predict for the following compound? (Consider all…
A: Given is organic compound.In 1H NMR number of signals is equal to number of chemically…
Q: 16) 6) 9) CH₂ KMnO4 NaOH (CH))NH (CH₂)2Culi 7) 11) 13) 1) SOCI₂ CH₂COO CH₂OH NaOH Heat 3) 8) 12) ,…
A:
Q: A research student used a new software package to calibrate the electronic map using a material…
A: The objective of the question is to determine which atom, gallium (Ga) or bromine (Br), would have…
Q: Consider an atom having BCC structure with atomic weight 167.5 g/mol. The density of the structure…
A:
Q: Consider the electrochemical cell reaction of hydrogen gas and chlorine gas to produce 1 mol of HCI…
A:
Q: 3) Draw a representation of the transition state for the following endergonic mechanistic step.…
A: The question is based on the concept of organic reaction.We need to draw transition state for the…
Q: 17. What is the major product of the reaction shown? Br (A) (C) CH3 NaOCHy CH₂OH CH₂ CH₂ (B) (D) CH3…
A: Alkyl halide reacts with a base in presence of polar solvent to form alkene through E1…
Q: Explanation Check molecule 0= 0= OH OH FO OH type of molecule (check all that apply) fatty acid…
A: Classify the following molecules:
Q: Write all the sets of quantum numbers possible for electrons in the n=3 energy level .
A: Quantum numbers are used to represent the position of an electron in an atom. Every electron has a…
Q: how would you solve this? it is a non graded practice
A: The question is asking about the interaction between Ethanal and water when they are mixed together.…
Q: Use the following Lewis diagram for methyl acetate to answer the questions: H :0: TH Remember that…
A: Given is a Lewis structure of Methyl acetate molecule.We have to find:a) geometry at atom.b) Bond…
Q: What type of Intermolecular forces exist between the side chains of isoleucine and valine?
A: The types of intermolecular forces possible between the amino acid side chains are electrostatic…
Q: FO 18: Identify resonance contributors. Circle any/all of the resonance contributors for this…
A: The objective of the question was to predict the major contributing resonating structure.The…
Q: This compound was synthesized from acetylene and an aldehyde/ketone. Draw the structure of the…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Draw all likely products of the following reaction. CH₂ -OH B H₂SO, heat Draw the molecules on the…
A: Alcohol gives dehydration reactions when it reacts with a strong acid like H2SO4 or H3PO4 to form an…
Q: OH EtOH 100° C, 30 min. OH Scheme 2: A Diels-Alder reaction between 9-anthracenemethanol and…
A: Given is Diels-Alder reaction. Diels-Alder reaction is [4+2] cycloaddition reaction.This is single…
Q: Calibration of a glass electrode gave a reading of 139.3 mV with a 0.05 m potassium hydrogen…
A:
Q: 9. As a review, provide the reagents for the following transformations. ▬▬ Br → ― 10. Provide the…
A:
Q: Draw the Lewis structure (with formal charges) of the indicated chemical species. If there is…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Q: Question 1: What volume (in mL) of a 0.122 M Ca(OH)2 solution should be used to neutralize 24.3 mL…
A: Answer:Number of moles of any specie in a solution is always equal to the product of molar…
Q: please do a detailed mechansim IZ CH3 H3O+ H₂O 3 HO V H + H3C-NH2
A: The given reaction scheme is shown below.We have to draw the mechanism of the above reaction.
Q: What is the extinction coefficient for the following absorbances? 1. 3.000 2. 2.022 3.1.2025 4.…
A: The objective of the question is to calculate the extinction coefficient for the given absorbances…
Q: Draw (R,S)-1,2-diaminocyclohexane. Draw its enantiomer. Name its enantiomer. Are these compounds the…
A: Isomers are two or more forms of a compound having same chemical formula but different structural…
Q: If 37.2 mL of 0.500 M K2Cr2O7 is mixed with 15.77g of PbSO4, which will be the limiting reactant?…
A: The molar mass of K2Cr2O7 = 294.185 g/molThe molar mass of PbSO4 = 303.26 g/molThe balanced…
Q: -Fe is cubic. Is it FCC or BCC? 20 30 110 sommy kambarys 40 50 60 200 Diffraction angle 20 70 211…
A: The type crystal system can be decided by seeing the peaks in the powder xrd spectrum. For BCC type…
Q: 9. While hydrogen peroxide is a common compound, it readily undergoes decomposition if the chemical…
A: According to Dalton's Law of Partial Pressure states that the total pressure exerted by a mixture of…
Q: A student measures the Pb2+ concentration in a saturated aqueous solution of lead hydroxide to be…
A: Solubility product constant is defined as the product of concentration of ions present in the…
Q: Consider an aqueous NaOH solution that contains 20 wt% NaOH and 80 wt% H₂O (MWNaOH = 40, MWH20 =…
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: Draw complete reaction mechanism for this reaction and lable to product.
A: Carbonyl compounds (aldehyde and ketone) react with an alcohol to form hemiacetal in the presence of…
Q: Draw the BEST lewis structure (with formal charges) of the indicated chemical species. (Draw all…
A: Lewis structures are used to show the electron pairing between the atoms to form a bond. It does not…
Q: A fentanyl addict has spilled 1000 L of gasoline into the street (MW=117 g/mol, density=0.7 g/mL).…
A: Given :- Volume of gasoline (solvent) = 1000 L = 1000000 mL Molar mass of gasoline (solvent) = M1 =…
Step by step
Solved in 3 steps with 2 images

- Calculate ∆Hrxn for the following reaction:N2H4(l) + CH4O(l) → CH2O(g) + N2(g) + 3H2(g)Use the following reactions and given ∆H values.2 NH3(g) → N2H4(l) + H2(g) ΔH = +22.5 kJ/molN2(g) + 3 H 2(g) → 2 NH3(g) ΔH = +57.5 kJ/molCH2O(g) + H2(g) → CH4O(l) ΔH = +81.2 kJ/molOrgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???SOURCE: GENERAL, ORGANIC AND BIOLOGICAL CHEMISTRY BY Smith 4th Edition
- SOURCE: GENERAL,ORGANIC AND BIOLOGICAL CHEMISTRY by Smith 4th EditionCompound ΔHf (kj/mol) C2H5OH(i) -277.6 H2O(i) -285.8 H2O(g) -241.8 CO2(g) -393.5 CO(g) -110.5 H2O2(i) -187.6 C3H8(g) -103.8 What is the ΔH for the following reaction? C2H5OH(i) + 3O2 (g) --> 2CO2(g) + 3H2O(i)Please follow the steps in the ohter picture
- Need Asap. Provide clear explanation and show work for each step. Please in clear writing or typed in a way that can be easily understood.From the enthalpies of reaction2 C1s2 + O21g2¡ 2 CO1g2 ΔH = -221.0 kJ2 C1s2 + O21g2 + 4 H21g2¡ 2 CH3OH1g2 ΔH = -402.4 kJcalculate ΔH for the reactionCO1g2 + 2 H21g2¡CH3OH1g2Use Drago’s E and C parameters to calculate ΔH for the reactions of NH3 and BF3 and of NH3 and B(CH3)3. Compare your results with the reported experimental values of −71.1 and −64 kJ/mol for NH3—B(CH3)3 and −105 kJ/mol for NH3—BF3. use NH3 as the base, and put the four reactions in order of the magnitudes of their ΔH values.
- !( answer with explanation plz step by step2A---> 2B + C (Kp1) 2D---> 2E + C (Kp2) Now find the corresponding Kp3= f(Kp1, Kp2) = ?for this reaction: D + B----> A + E elaborate throughly with every step and annotationplease help me solve these both questions by explaining the pieces that forms at each peak and the final structure for my Organic Chemistry 1 class. Thanks in advance! the solution is provided i just need to understand how to reach there please.



