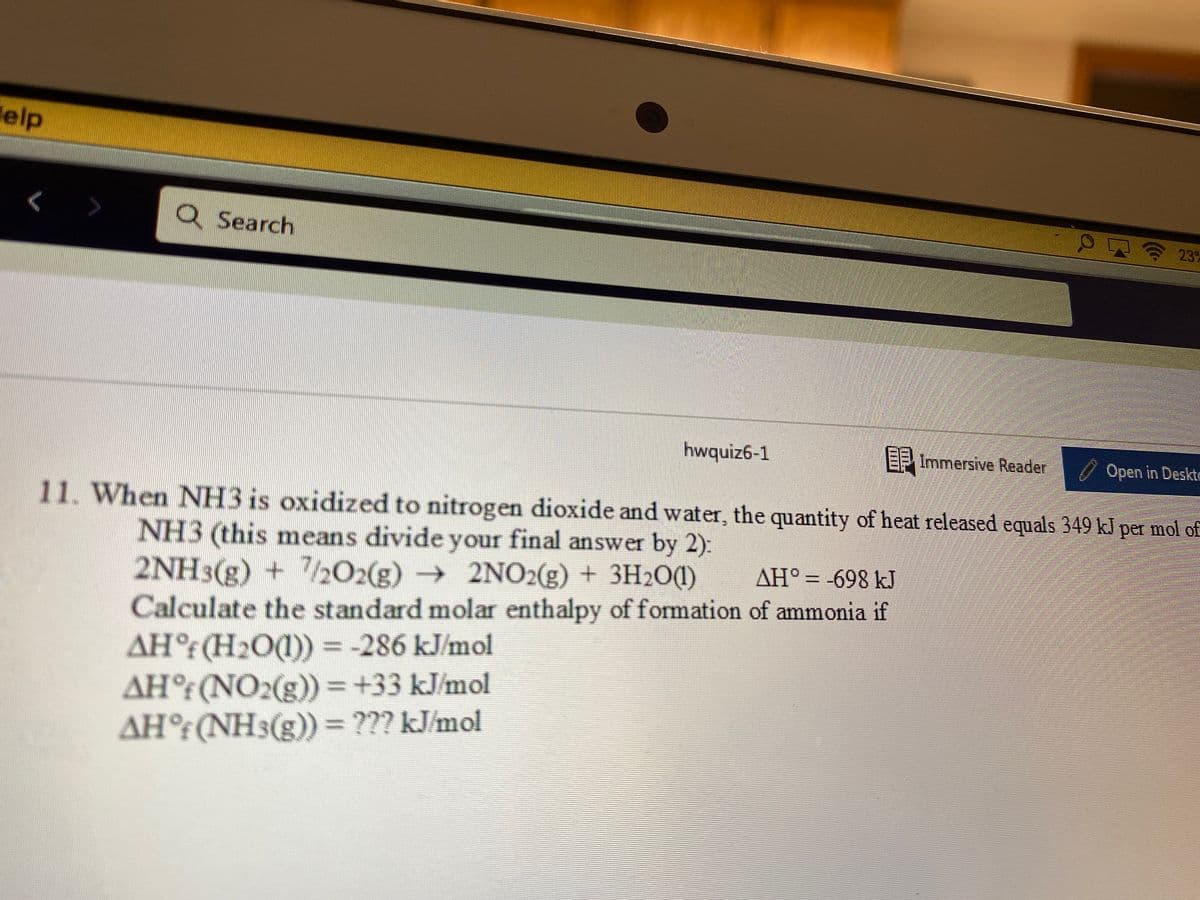
EF Immersive Reader Open in Des When NH3 is oxidized to nitrogen dioxide and water, the quantity of heat released equals 349 kJ per mol NH3 (this means divide your final answer by 2): 2NH3(g) + /2O2(g) → 2NO2(g) + 3H2O(1) Calculate the standard molar enthalpy of formation of ammonia if AH° (H2O(1)) = -286 kJ/mol AH° (NO2(g)) = +33 kJ/mol AH°(NH3(g)) = ??? kJ/mol AH° = -698 kJ
EF Immersive Reader Open in Des When NH3 is oxidized to nitrogen dioxide and water, the quantity of heat released equals 349 kJ per mol NH3 (this means divide your final answer by 2): 2NH3(g) + /2O2(g) → 2NO2(g) + 3H2O(1) Calculate the standard molar enthalpy of formation of ammonia if AH° (H2O(1)) = -286 kJ/mol AH° (NO2(g)) = +33 kJ/mol AH°(NH3(g)) = ??? kJ/mol AH° = -698 kJ
Chapter26: Biodiesel
Section: Chapter Questions
Problem 2Q
Related questions
Question
Transcribed Image Text:lelp
Q Search
239
hwquiz6-1
眼!
E Immersive Reader
Open in Deskto
11. When NH3 is oxidized to nitrogen dioxide and water, the quantity of heat released equals 349 k per mol of
NH3 (this means divide your final answer by 2):
→ 2NO2(g) + 3H2O(1)
AH° = -698 kJ
2NH3(g) + 2O2(g)
Calculate the standard molar enthalpy of formation of ammonia if
AH° (H2O1) = -286 kJ/mol
AH°(NO2(g)) = +33 kJ/mol
AH°:(NH3(g))= ??? kJ/mol
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning