For the gas phase decomposition of t-butyl acetate, CH3COOC(CH3)3–(CH3)½C=CH, + CH;COOH the rate constant in s' has been determined at several temperatures. When In k is plotted against the reciprocal of the Kelvin temperature, the resulting linear plot has a slope of -2.04×104 K and a y-intercept of 30.7. The value of the rate constant for the gas phase decomposition of t-butyl acetate at 511 K is (Enter your answer to one significant figure.)
For the gas phase decomposition of t-butyl acetate, CH3COOC(CH3)3–(CH3)½C=CH, + CH;COOH the rate constant in s' has been determined at several temperatures. When In k is plotted against the reciprocal of the Kelvin temperature, the resulting linear plot has a slope of -2.04×104 K and a y-intercept of 30.7. The value of the rate constant for the gas phase decomposition of t-butyl acetate at 511 K is (Enter your answer to one significant figure.)
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.49PAE: The rate of photodecomposition of the herbicide piclo- ram in aqueous systems was determined by...
Related questions
Question
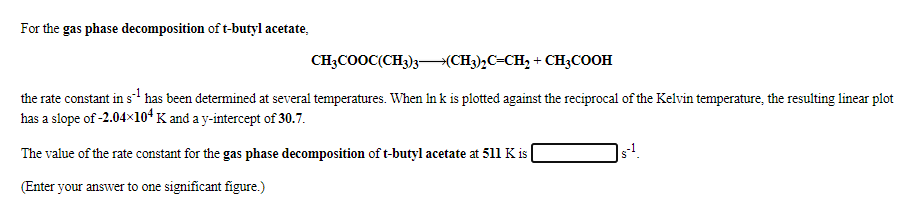
Transcribed Image Text:For the gas phase decomposition of t-butyl acetate,
CH3COOC(CH3)3–(CH3)½C=CH, + CH;COOH
the rate constant in s' has been determined at several temperatures. When In k is plotted against the reciprocal of the Kelvin temperature, the resulting linear plot
has a slope of -2.04×104 K and a y-intercept of 30.7.
The value of the rate constant for the gas phase decomposition of t-butyl acetate at 511 K is
(Enter your answer to one significant figure.)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning