Four amino âĉid samples were subjected to a reverse-phase chromatography set up and yielded the following Rf values. Which among the samples is the most polar? Sample Rf values 0,49 0.53 0.21 0.55 OD B
Four amino âĉid samples were subjected to a reverse-phase chromatography set up and yielded the following Rf values. Which among the samples is the most polar? Sample Rf values 0,49 0.53 0.21 0.55 OD B
Principles Of Radiographic Imaging: An Art And A Science
6th Edition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Chapter8: X-ray Production
Section: Chapter Questions
Problem 2RQ
Related questions
Question
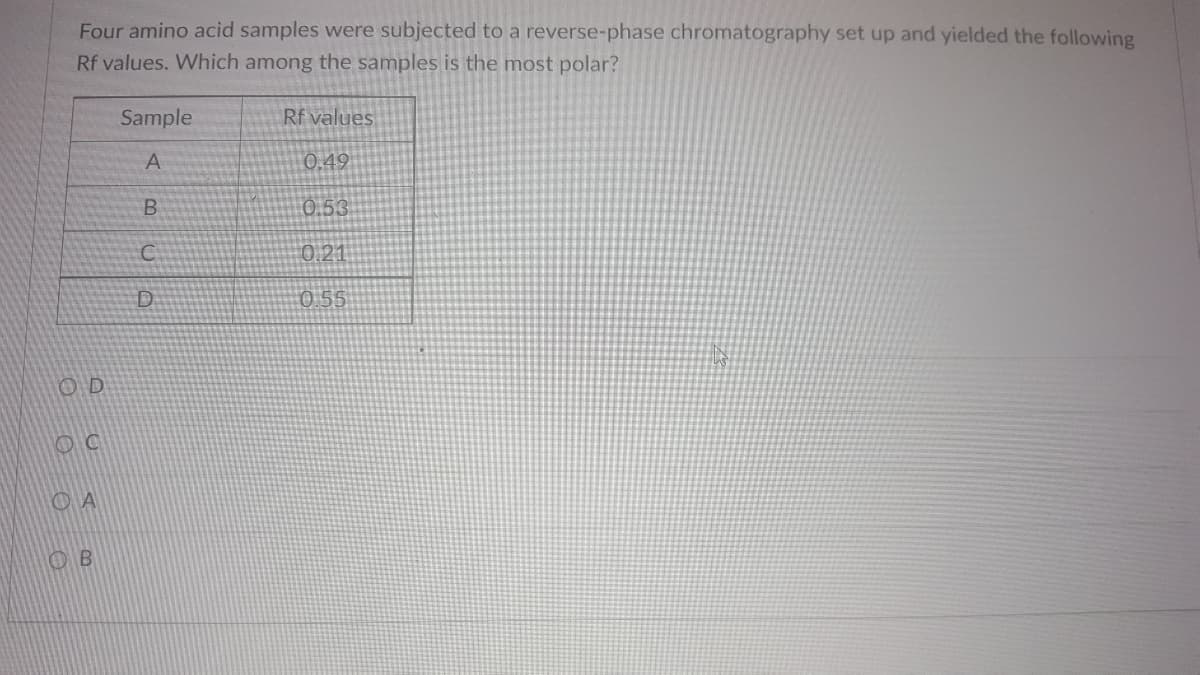
Transcribed Image Text:Four amino acid samples were subjected to a reverse-phase chromatography set up and yielded the following
Rf values. Which among the samples is the most polar?
Sample
Rf values
A
0,49
0.53
0.21
0.55
OD
OB
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning


Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning


Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning