Q: d. Which average molecular weight determination did the fraction 3 most significantly affect and…
A: We have to give the answer of the given questions. D. We have to tell which molecular weight…
Q: How will you demonstrate the purity of your final product (crude or recrystallized)?
A: To demonstrate the purity of the final product
Q: Use the items to construct a flow chart for the procedure used to create a saturated borax solution…
A: The blanks in the given procedure is filled as follows,
Q: What is inert gas regeneration method?
A: Inert gases form the 18th group of periodic table. Inert gases like Helium, Neon are used in…
Q: In vacuum filtration, the filter paper should be wet before the final product solution is poured…
A: In vacuum filtration, the filter paper should be wet before the final product solution is poured…
Q: Although we will not use a Claisen adapter in our apparatus, what quality of vacuum distillation…
A: Vacuum distillation is a kind of distillation process based on the differences in the boiling points…
Q: What is the purpose of a brine wash (saturated sodium chloride solution) in a liquid/liquid…
A: We have to predict the purpose of brine washing.
Q: 2 is corrosive, so purging with N2. which is inert, helps to reduce reactivity within the…
A: Infrared spectroscopy is being used to identify the organic compounds by recognizing the functional…
Q: Below is a chromatogram observed from chemical separation of chlorinated pesticides using gas…
A: Gas chromatography is an analytical technique used for the separation of chemical components of a…
Q: Study figures 31-6 a & b in chapter 31 (page 863) of your prescribed textbook, and draw separation…
A: 1
Q: What is each cycle of a simple distillation called? Theoretical plate Distillate…
A: Simple distillation is process of separation.
Q: What is the purpose of using ether in preparing the dispersion of egg yolk sample? (the answer…
A:
Q: What is the maximum mass of cyclohexene (density 0.81 g/mL, MW = 82.14 g/mol) you can recover from…
A:
Q: Remember how you learned that a pure liquid distills within a narrow boiling point range, but that a…
A:
Q: 3.Why is it dangerous to perform distillation on a closed (airtight) system?
A: The process distillation involves the conversion of a liquid into vapour that is subsequently…
Q: In line with the correct answr to the previous question, indicate which statements are true about…
A: Fractional distillation is one of the most widely used separation techniques in chemistry. This…
Q: Why is sample dropped into the preheated furnace before the oxygen concentration reaches its peak in…
A:
Q: 1. What is the purpose of application of steam distillation method?
A: Applying basics concept of steam distillation purification method.
Q: Naphthalene is separated in the mixture (methylene blue, sodium chloride and and is converted back…
A:
Q: Please help me please. I promise to give a feedback.. I only need the answer of definitions please.…
A: Combustion, is a chemical reaction between a substance and oxygen, usually in the form of a flame,…
Q: High molecular weight step growth polymers can be obtained if the reaction is carried out at…
A: Step-growth polymerization
Q: Melting point: 122-123 experimental product melting point: 120 mass of starting material: 1.04…
A: A numerical problem based on yield, which is to be accomplished.
Q: Define, explain and elaborate
A: The heat of reaction, the amount of heat that must be added or removed during a chemical reaction in…
Q: Three sample, X (neutral), Y (basic), and Z (acidic), were mixed and dissolved together in…
A: Dissolve the mixture in dichloromethane and separate the organic and aqueous layer using liquid…
Q: Explain the working of fixed-bed carbon solvent recovery system.
A: An atom is the basic and fundamental unit of matter. An atom consists of electrons and the nucleus…
Q: Consider the calcination of CaCO3 Before calcination, the initial weight of CaCO3 is 1.005g. After…
A:
Q: Melting point: 98-105 Percent yield: 60% did you successfully make pure product? Explain.
A: A question based on IR spectroscopy, which is to be accomplished.
Q: Q2. What does “crude” product mean? Q3. What is the advantage of using Buchner filtration in…
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am…
Q: Draw and label a moisture sorption isotherm for any product of your choice at different temperature…
A:
Q: When should we stop collecting distillate in ethanol-alcohol simple distillation process? Give me…
A: We are find out when to stop collecting distillate in ethanol distillation process.
Q: At what stage was the internal standard added to the prepartions, in the protocol of this…
A: Step : 1 The required answer for the above question is follows as :
Q: Paraxylene, styrene, toluene, and benzene are to be separated with the array of distillation columns…
A: Accordingto the given Diagram, where F, D, B, D1, B1, D2 and B2 are the molar flow rates in…
Q: Crystal seeding
A: In this question we have to explain if there is no recrystallization due to the unsaturation of the…
Q: Please what's the observation for the combustion of flame test Combustion: Flame test 1.…
A: The observation for the combustion of the flame test has to be given,
Q: Upon completing a synthesis, you attempt to purify your material via recrystallization. You dissolve…
A: First we would explain recrystallization effect , then we can determine the issue in this given…
Q: A small intermittent fraction is collected and discarded. This is a good practice because…
A: all steam distillation , the distillation collected will be both mixture of liquid but since they…
Q: Contrast the physical appearance of the crude material before the recrystallization and that of the…
A: A question based on re-crystallization, which is to be accomplished.
Q: What parameters should be considered when choosing between distillation methods? Are there any…
A: Distillation is refining method of refining by alternating evaporation and condensation to remove…
Q: Synthesis of Orange II dye.
A: Orange II Synthesis a. Cool 10 mL of 10% NaOH in a 400 mL beaker. Add 1.8 g of 2-naphthol to this…
Q: Please choose an appropriate technique(s) to separate following compounds (it can be one, more than…
A: We can see the difference in boiling point of both that is the Phenol and Toluene.
Q: While performing a crystallization, you obtain a light tan solution after dissolving your crude…
A: It is observed that a light tan solution is obtained and because of this, it is better to filter it…
Q: you answer and explain this? a. Explain in your own words how the concept of vapor pressures would…
A: Answer Steam Distillation. Steam distillation is a separation method that involves distilling water…
Q: Briefly, discuss reflux and why it is commonly used in organic chemistry.
A: Here a term 'reflux' is given.Generally it is used in organic chemistry. We have to discuss about…
Q: In atomic spectroscopic techniques, flames and furnaces are commonly used to atomize the samples.…
A: In atomic spectroscopic techniques, flames and furnaces are commonly used to atomize the samples.…
Q: What might happen to your melting points if you do not cool the MelTemp between runs? The melting…
A: The temperature at which the solid becomes liquid is called melting point. Melting point is a…
Q: Purpose of quantitative transfer in experiments.
A: The dissolution of a solute in a solvent based on its solubility rule provides a solution. If the…
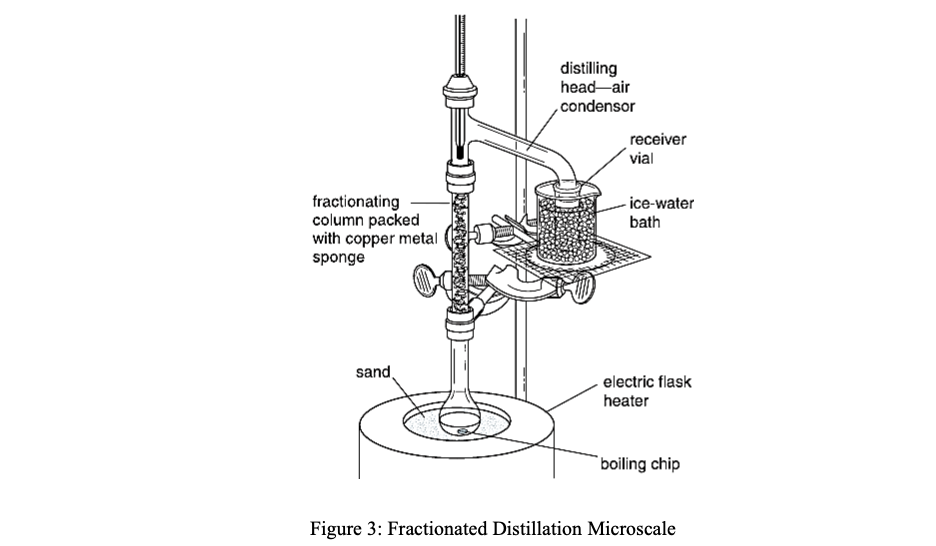
Fractioned Distillation Microscale
Looking at this set up, are there any potential problems with it? Please explain.
Step by step
Solved in 2 steps with 1 images

- Referring to the Al-Si Eutectic diagram discussed on the Phase Diagrams-Lever Rule handout, calculate the mass fraction of the phase, silicon, given a sample with 51.7 wt% aluminum at 576.99 ˚C.DATA Note: Use the videos as references except when data is provided here. Table 1. Selection of Recrystallizing Solvent. Solvents Dissolution at room temperature? (+ or −) Dissolution at elevated temperature? (+ or −) Distilled water - - Acetone + n/a 95% ethanol - + Toluene + n/a Solid sample: Naphthalene Table 2. Mass Measurements. Mass, g Sample solid 0.5021 Empty watch glass 35.7602 Watch glass with recrystallized sample 36.2485 Table 3. Melting Point Determination. Temperature, oC Appearance of Sample in Capillary Tube … Solid 78.4 Solid 79.3 Solid with signs of liquid 79.9 Solid, liquid 80.6 Liquid, solid 81.3 Liquid with signs of solid … Liquid Theoretical Melting Point, oC: 80.2°C (176.4°F) QUESTIONS Which solvent should be used as the recrystallizing solvent? Check if your chosen solvent satisfies all characteristics of a good…After a recrystallization, a pure substance will ideally appear as a network of Choose...smalllargeround Choose...clumpspowderscrystals . If this is not the case, it may be worthwhile to reheat the flask and allow the contents to cool more Choose...completelyquicklyslowly .
- solve as neatly as possible and show completesolution. Round your final answer to 4 decimal places and box / highlight all final answers. and all values must include proper units with proper conversion if needed in your solution. kindly folllow the format Given,Required,Solution subject physical chemistry 1. Calculate the mass of water vapor present in a room of volume 400 m3 thatcontains air at 290C when the relative humidity is 53.0%. Compute for the vaporpressure of water?Taylor was harvesting her beautiful, pure crystals via vacuum filtration from a methanol/water mixedsolvent system. She poured the solution containing her crystals into the Büchner funnel. Then sherinsed her flask with some warm methanol and poured that over the crystals resting in the Büchner…oops! Tell what happened and whydont provide handwritiing soution ...
- Matt was heating his methanol solvent in an E. flask on a hot plate to use for recrystallization. The TAcame by and noticed he didn’t have a stir stick or boiling chip in the flask and told him to use one,please. Matt dropped a stick in the flask and suddenly the liquid exploded out of the flask. Oops! Tellme why that happened. What should he have done instead?I s it possible to carry out steam distillation at a temperature higher than 100C at 1 atm? ExplainPolyethylene terephthalate is an example of synthetic material in Materials and Engineering. 1. Describe the relation of its building blocks in connection to its “bulk” (materials) properties. (No need for essay, just a brief scientific explaination!) If you may, please send link as well of the reference that you use for the answer).

