Given the data in Figure 2, answer the following questions. You may refer to the color wheel and the color/wavelength chart on the cover sheet. n Values Concentration LH Relationship Between Concentration and Absorbtion 0.70- (c.u./L) Absorbance 5.000 0.289 0.65- 10.000 0.464 15.000 0.638 20.000 0.709 0.60- 0.55- Linear mc+b m =0.0286 0.50- b =0.167 r=0 985 0.45- 0.40 - 323 nm 0.35- 0.30- Unknown Concentration Concentration 0.25- (c.u./L) Absorbance 10 13 15 18 20 0.444 Concentration (c.u./L) Figure 2: Determination of an Unknown Solution Concentration What is the concentration of the unknown solution in c.u./L (color units per liter) а. a. 9.68 b. 15.52 c. 9.01 d. 12.87 Absorbance
Given the data in Figure 2, answer the following questions. You may refer to the color wheel and the color/wavelength chart on the cover sheet. n Values Concentration LH Relationship Between Concentration and Absorbtion 0.70- (c.u./L) Absorbance 5.000 0.289 0.65- 10.000 0.464 15.000 0.638 20.000 0.709 0.60- 0.55- Linear mc+b m =0.0286 0.50- b =0.167 r=0 985 0.45- 0.40 - 323 nm 0.35- 0.30- Unknown Concentration Concentration 0.25- (c.u./L) Absorbance 10 13 15 18 20 0.444 Concentration (c.u./L) Figure 2: Determination of an Unknown Solution Concentration What is the concentration of the unknown solution in c.u./L (color units per liter) а. a. 9.68 b. 15.52 c. 9.01 d. 12.87 Absorbance
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter4: Stoichiometry: Quantitative Information About Chemical Reactions
Section: Chapter Questions
Problem 77PS
Related questions
Question
Short Answer 1a.
Question shown in picture.
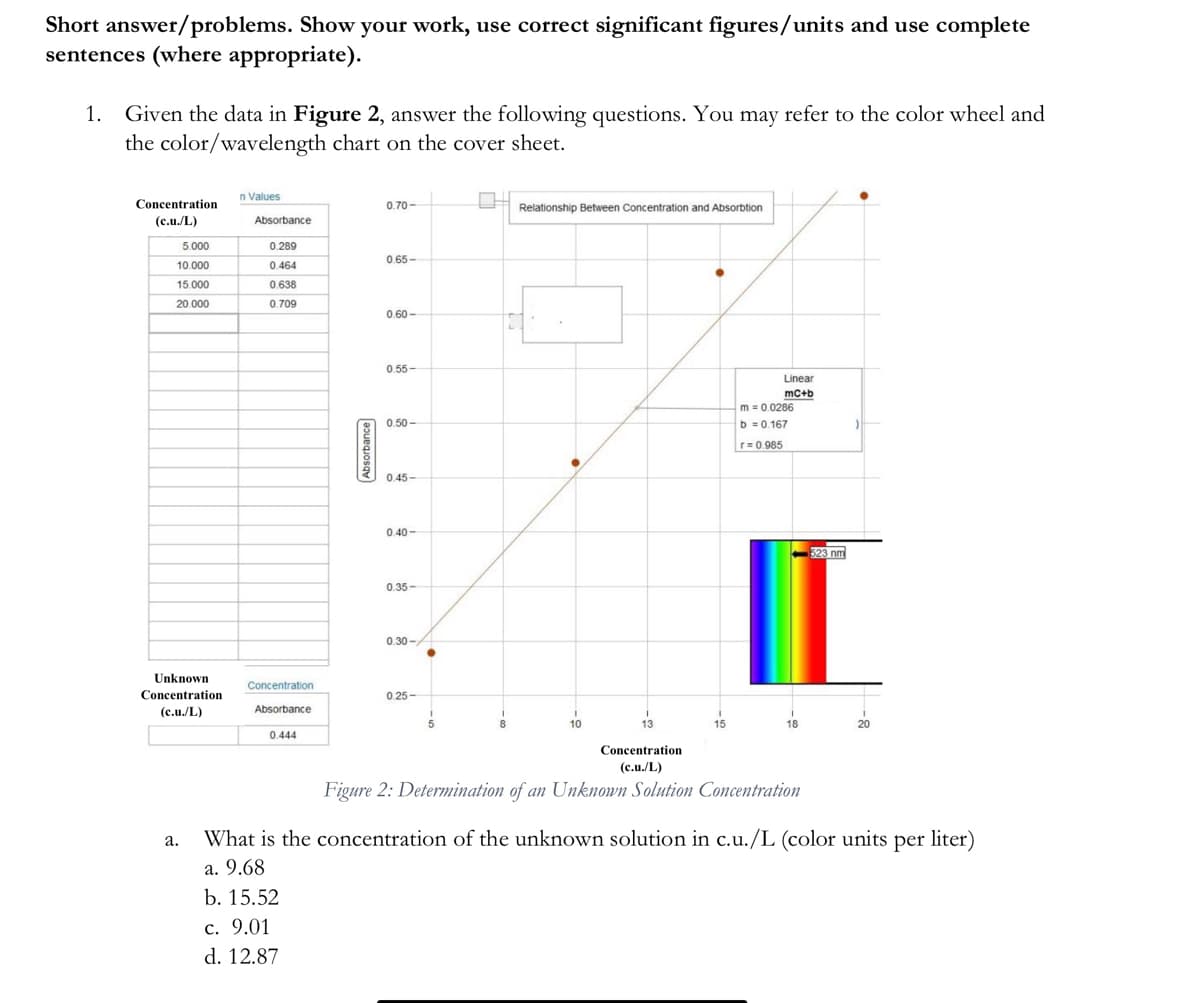
Transcribed Image Text:Short answer/problems. Show your work, use correct significant figures/units and use complete
sentences (where appropriate).
1.
Given the data in Figure 2, answer the following questions. You may refer to the color wheel and
the color/wavelength chart on the cover sheet.
n Values
Concentration
0.70-
Relationship Between Concentration and Absorbtion
(c.u./L)
Absorbance
5.000
0.289
0.65-
10.000
0.464
15.000
0.638
20.000
0.709
0.60 -
0.55-
Linear
mc+b
m = 0.0286
b = 0.167
0.50 -
r= 0.985
0.45-
0.40 -
523 nm
0.35-
0.30 -
Unknown
Concentration
Concentration
0.25-
(c.u./L)
Absorbance
10
13
15
18
20
0.444
Concentration
(c.u./L)
Figure 2: Determination of an Unknonn Solution Comcentration
What is the concentration of the unknown solution in c.u./L (color units per liter)
а.
a. 9.68
b. 15.52
с. 9.01
d. 12.87
Absorbance
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning