Q: At 20°C, protein-Z binds DNA with a free energy of -7 kJ/mole. Protein Z has an unfolding free…
A:
Q: If we made a Lineweaver-Burk plot of an irreversible inhibitor, which type of reversible inhibition…
A: In a Lineweaver-Burk plot for an irreversible inhibition, the most likely resemble inhibitor has to…
Q: The cleavage pattern for o-lodosobenzoate is C-terminal to tryptophan; the cleavage pattern for…
A:
Q: Sephadex G-75 has an exclusion limit of 80,000 molecular weight for globular proteins. If you tried…
A: The separation of alcohol dehydrogenase from β-amylase using Sephadex G-75 as the column material…
Q: How does the near attack conformation (NAC) contribute to enzyme function?
A: An enzyme is a biological catalyst, which catalyses many of the metabolic actions in a human body.
Q: What is the difference between the whole Aspartate Transcarbamoylase (ATCase) enzyme and an ATCase…
A: Allosteric enzymes are enzymes that change their conformational ensemble upon binding of an…
Q: In the "Catalysis by Preferential Transition State Binding" mechanism, the substrate perfectly fits…
A:
Q: . Delineate the detailed regulatory mechanism of activation of protein kinase A (PKA)…
A: Answer - According to the question - Protein kinase A (PKA) is activated by elevations in cAMP and…
Q: An enzyme whose KM is 10-4 M in the presence of a substrate concentration of 10-2 M It is capable of…
A:
Q: Sulfanilamide, a sulfur drug, acts as an antibiotic. Explain its mechanism of action in the context…
A: Ans. Sulfanilamide: An enzyme Inhibitor: Sulfa drugs work by binding and inhibiting a specific…
Q: Select ALL of the following statements that correctly describe how an enzyme lowers the activation…
A: Concept - Catalyst is a substance that increases the rate of a reaction without itself being…
Q: An enzyme catalyzes the hydrolysis of an ester with a certain activity, but this activity is lost in…
A: Any enzyme can be categorised a certain protein which loses its activity at a certain temperature,…
Q: Summarize three ways in which an enzyme might lower the energy of a activation of a raction 2. What…
A: PART A - The three ways in which an enzyme might lower the energy of a activation of a reaction are…
Q: Outline the four general stages in an enzyme-catalyzedreaction.
A: Catalysts are those compounds which alters the rate of the reaction, may or may not interact during…
Q: A low value of the Michalis constant (KM) in an enzyme-catalyzed reaction indicates--- The enzyme…
A: Km is the Michaelis-Menten constant which shows the concentration of the substrate when the reaction…
Q: How is the Km value influenced in the presence of the inhibitor?
A: The Km value is the concentration of substrate that provides the rate halfway to maximum velocity…
Q: If you had a protein X, which is a soluble enzyme found inside the peroxisome, and you wished to…
A: Proteins chain of amino acids which join together to form protein. Proteins are found in the…
Q: One of the beolow statements is FALSE Amytase Pepain Arginese 4. 10 12 O a. Irreversible enzyme…
A: The given graph in the question is a plot of activity of an enzyme vs. pH of the medium. In this…
Q: pinds to the active site of an enzyme clows the reaction down at low substrat
A: A competitive inhibitor binds to the active site of an enzyme.
Q: From the given activity, 1.) What is the effect of temperature to the enzyme? 2.) What are some…
A: Hi, we are supposed to answer one question. To get the remaining questions solved please mention the…
Q: Concentration, uM Vo (umol/min) w/o Vo (umol/min) with substance B substance B 0.4 0.08 0.069 0.7…
A:
Q: The enzyme responsible for the rapid formation of H2CO3 in the RBCs is: Group of answer choices…
A:
Q: A particular enzyme's activity is low at high pH but increases at lower pH. These data suggest that…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: How is the maximal rate of the enzyme changed in the presence of an inhibitor?
A: Maximum rate of reaction is characteristic of a particular enzyme at a particular concentration and…
Q: OH HNL CN R' + HCN One of the proposed mechanisms for this enzyme is shown in the scheme below.…
A:
Q: Explain why an irreversible inhibitor of an enzyme might be more toxic to humans than a reversible…
A: Enzyme are the biological catalyst. The inhibitor is the molecule that binds to an enzyme and…
Q: Describe the differences between chain termination, chain transfer and inhibition. Use suitable…
A: chain termination : Polymerization of many radical reactions involve chain reactions or chain…
Q: If the equations for the extreme lines (a and e) using units shown in graph above are: y= 2.6x + 3.0…
A: We have find out Vmax for enzyme.
Q: Calculate the index of hydrogen deficiency of this compound Q.) Urea, CH4N2O
A: The rings and multiple bonds reduces the hydrogen count by two and it is known as index of hydrogen…
Q: 23. Enzyme activity is best determined by performing a(n) a. enzyme international unit b. enzyme…
A: Enzyme assays are laboratory methods for measuring enzymatic activity. The quantity or concentration…
Q: what are the roles of glutamate and histidine in the catalytic mechanism?
A: Please find the below attachment.
Q: When weighing the protein at the end of the activity, what major assumption is made about the…
A: Normalization alludes to the act of changing the arrangement of cheddar milk to augment monetary get…
Q: When the relaxivity of a Gd-based magnetic resonance agent is high, this means that slight changes…
A: The given statement is False. Slight changes in Gd concentration will result into big changes in…
Q: A plot of activity versus pH for an enzyme that functions in the cytosol of a cell at pH of about 7…
A: The activity of enzymes varies at different pH levels.
Q: Review the structure of the standard amino acids and listthose that are capable of acting as acids…
A:
Q: Following data were obtained for an enzyme in the absence and presence of an inhibitor. What are the…
A: Given data:
Q: Chemistry Given TETRAPEPTIDE: Prolinyl-Argininyl-Cysteinyl-Tryptophan Predict the direction of the…
A: The given peptide according to the given sequence is: Proline makes the N terminal of the peptide.…
Q: Indicate the mechanism of enzyme inhibition represented in each plot, and justify your answer with…
A: Interpretation : Curves are given and we have to indicate the type of mechanism involved:
Q: For an enzyme that obeys Michaelis-Menten kinetics, which one of the following best indicates a…
A: Given: Vo = (1/3)Vmax
Q: How does changing the concentration of enzyme at a fixed concentration of substrate affect the rate…
A: The rate of reaction depends on the number of enzymes present and the substrates concentration.
Q: In order to optimize acetylcholinesterase inhibition, a set of benzylamine derivatives were studied,…
A:
Q: Does Km value change with an increase of enzyme concentration? Explain
A: In the simplest case of a monomeric enzyme having a single active site, the Km is independent of the…
Q: Explain why site-directed mutagenesis of aspartate to asparagine in the active site of rypsin…
A: Answer - Aspartate - Aspartate is a vitamin-like substance called an amino acid. As a dietary…
Q: What is the important values needed to calculat Km and Kcat afor an enzyme data?
A: We have to tell the important values are needed to calculate the Km and Kcat for an enzyme catalyzed…
Q: The shaded area in the following diagram shows CH₂ CH₂ ? H N-HO -CH₂ C-OH-N Gly 193 H RN O Covalent…
A:
Q: The Km value of an enzyme is affected in the presence of a competitive inhibitor because it requires…
A:
Q: The enzyme d-amino acid oxidase has avery high turnover number because the d-amino acids are…
A:
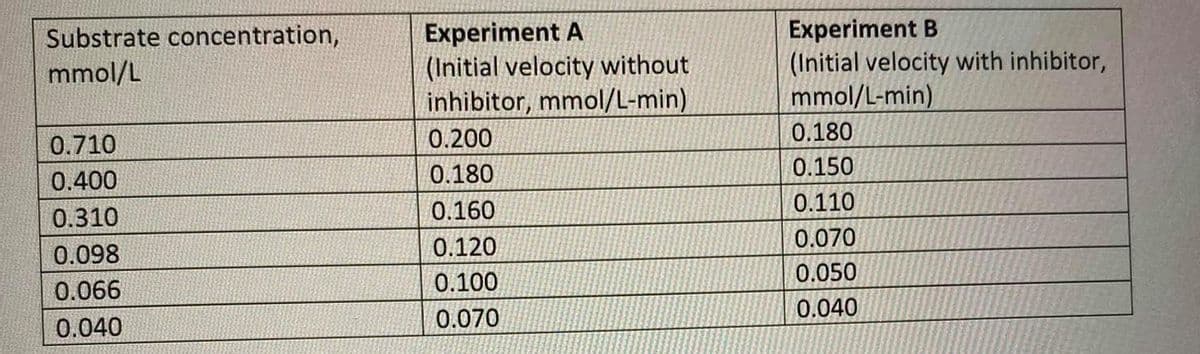
Given the following data in enzyme-catalyzed reaction, what is the Vm, Km and type of inhibition of Experiment B?
Step by step
Solved in 3 steps with 3 images

- 2I– (aq) + H2O2 (aq) + 2H3O + (aq) → I2 (aq) + 4H2O (l) (slow) C6H8O6 (aq) + 2H2O (l) + I2 (aq) → C6H6O6 (aq) + 2H3O + (aq) + 2I– (aq) (very fast) I2 (aq) + I– (aq) ⇌ I3- (aq) I3- (aq) + starch → blue I3- · starch complex (aq) (fast) A 0.100 L solution is prepared with initial concentrations of 4.0 × 10−3 M iodine I2 , 8.0×10−3 M iodide I– , and 5.0×10−3 M ascorbic acid C6H8O6 . After the second reaction goes to completion, what will the molar concentrations of iodide and ascorbic acid in the solution be?0% 25% 50% 75% 100% Depth of H2O2 Solution (d) 2.1 cm 2.1 cm 2.1 cm 2.1 cm 2.1 cm Trial 1 Time 180 sec 84.61 sec 43.52 sec 36.90sec 25.90 sec Trial 2 Time 180 sec 92.25 sec 38.16 sec 34.36 sec 23.57sec Trial 3 Time 32.53 sec 18.82 sec Average Time (t) 180 sec 88.43 sec 40.84 Sec 34.5 sec 22.76 sec Rate of the Reaction(R = d/t) 0.012 cm/sec 0.02 cm/sec 0.051 cm/sec 0.061 cm/sec 0.0923cm/sec , graph rate of reaction on the y-axis and percent concentration of enzyme on the x-axis. If the points are linear, draw a “best-fit” straight line through or near all of the data points. Based on the information in the data table and your graph, explain the relationship between percent concentration of catalase and rate of reaction. Did your actual results match your hypotheses? If not, why?Q) water hardness of each trial and average ppm with calculation, please. Hard Water Trial 1 Hard Water Trial 2 Hard Water Trial 3 Initial Syringe Reading 1.0ml 1.0ml 1.0ml Final Syringe Reading 0.88ml 0.84ml 0.85ml Volume of EDTA Consumed 0.12ml 0.16ml 0.15ml Water Hardness ppm CaCO3 Average ppm
- analyte concentration(C)(mg/ml) injection volume (ul) elution time (time) peak DAD signal(mAU) caffeine 1 1 4.67 302.85 aspartame 5 1 7.53 15.83 benzoic acid 1 1 8.14 89.98 saccharin 1 1 1.91 84.86 mixture(add everything above with 1:1:1:1 ratio) 1 4.47 69.58 How to get the concentration of the mixture in this case?2I– (aq) + H2O2 (aq) + 2H3O + (aq) → I2 (aq) + 4H2O (l) (slow) C6H8O6 (aq) + 2H2O (l) + I2 (aq) → C6H6O6 (aq) + 2H3O + (aq) + 2I– (aq) (very fast) I2 (aq) + I– (aq) ⇌ I – 3 (aq) I3- (aq) + starch → blue I3- · starch complex (aq) (fast) A different 0.100 L reaction mixture is prepared with initial concentrations of 1.0×10−2 M iodide I– , 2.0 × 10−3 M ascorbic acid C6H8O6 , and 0.135 M hydrogen peroxide H2O2 . If the first two reactions above procede at the same rate (they’re limited by the first one!), which of these reactants will be completely consumed first?2I– (aq) + H2O2 (aq) + 2H3O + (aq) → I2 (aq) + 4H2O (l) (slow) C6H8O6 (aq) + 2H2O (l) + I2 (aq) → C6H6O6 (aq) + 2H3O + (aq) + 2I– (aq) (very fast) I2 (aq) + I– (aq) ⇌ I – 3 (aq) I3- (aq) + starch → blue I3- · starch complex (aq) (fast) (a) A 0.100 L solution is prepared with initial concentrations of 4.0 × 10−3 M iodine I2 , 8.0×10−3 M iodide I– , and 5.0×10−3 M ascorbic acid C6H8O6 . After the second reaction goes to completion, what will the molar concentrations of iodide and ascorbic acid in the solution be?
- Which of the following describes a gradient elution process? stationary phase has varying concentration mobile phase has varying concentration stationary phase has static concentration mobile phase has static concentrationYou finish doing an experiemnt with Benzoin. These are the results you get:Boiling start point: 137 ceciusBoiling end point: 130 ceciusVile while empty (with cap on): 15.348Vile with crude prod (with cap on: 15.748 Your starting weight: 1.00115.748 - 15.348 = 0.400 / 1.001 = 0.3996 or 39.96% YieldAnswer the following questions with this information:Initial Mass of impure sample:Mass of recrystallized benzoin:Percent reccovery of Benzoin:Melting pot range of purified benzoin:Literature melting point for benzoin:Please write a paragraph of the objective stated using the two images attached below Please please answer as fast as possible thank you
- if you perform QC for sodium on a chemistry analyzer and the value is greater than 2SD above the mean. This is the second day that this level of control has been >2SD for that analyte. You mixed the vial and repeated. QC was still "out". It is time to troubleshoot. What do you do?If some of the analytes were found to be out of range after doing the Quality Control and the machine repeatedly gives invalid result. What are the possible problem and best solution as a Medical Technologist?18. Jet is an undergraduate chemistry student, he’s out in the laboratory trying to determine the volatile organic compounds as well as overall protein content of the leaf and stem of a malunggay (Moringa oleifera). He subjected the leaf and stem in a separate digestion reaction (treatment of sulfuric acid), afterwards he subjected the products to high temperature induction to get a dry ash like substance. Which type/s of chemical analysis did Jet employ to reach his objective?I. Qualitative AnalysisII. Quantitative Analysis III. Instrumental Chemical AnalysisIV. Wet Chemical AnalysisA. II & IV onlyB. I & IV onlyC. I, II & IV onlyD. I, II, III, IV

