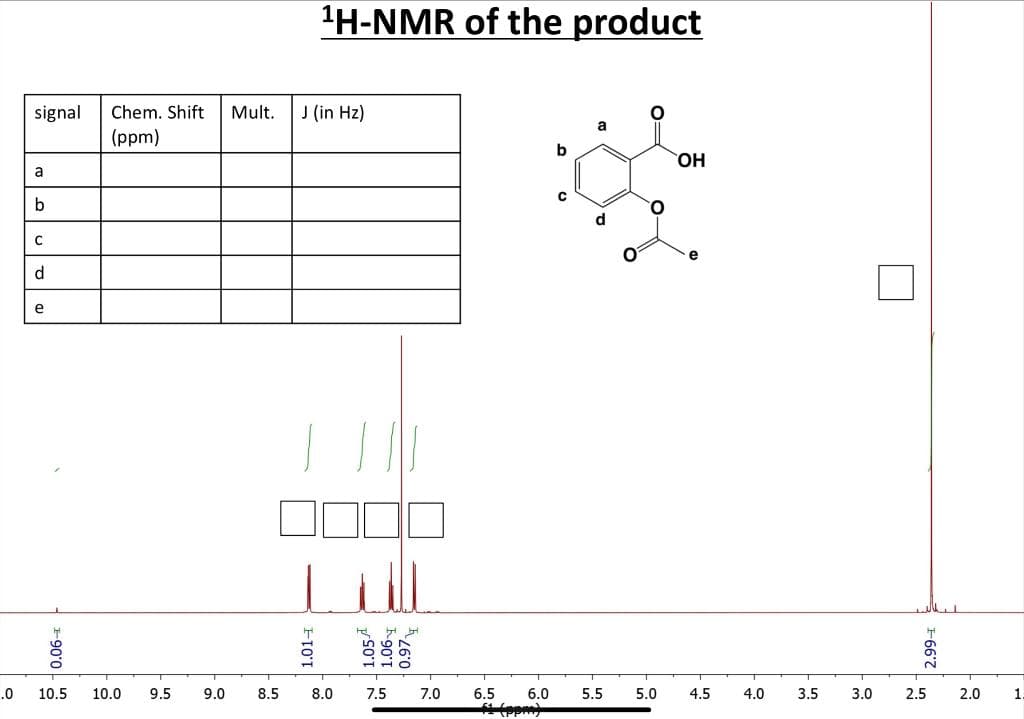
"H-NMR of the product signal Chem. Shift Mult. J (in Hz) (ppm) b HO, a d e d. e ll 10.5 10.0 9.5 9.0 8.5 8.0 7.5 7.0 6.5 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 E90°0 -8 1.014 1.05A K90°I n 0.97 0= in
Q: 2. MF Relative Intensity 100- 80 40 20 0-|rrrrrr 25 -5 50 75 m/z 100 125 150
A:
Q: Spectrum Scan (1/1] Data saved in "c:\projects\data\2020\02\27\5\20200227-112637-5-1D PROTON" START…
A: The question is based on the concept of molecular structure determination. we have been provided…
Q: Red 40 is also known as Allura Red. Peak absorbance, 504 nm (blue-green). NaO `ONa Erythrosine, also…
A: Hydrophobic substance are those which are not soluble in water. Wheras hydrophilic susbstances are…
Q: A. A student performed the following reaction, and took an IR spectrum of the product. Do you think…
A: The chemical reaction performed is the oxidation of cyclohexanol to cyclohexanone in the presence of…
Q: он OH NH2 a. Label important functional groups above 1500 cm* in the IR spectrum of synthesized…
A: a. position ρm-1 function at group 3321 m-1 single peak…
Q: C,H140 IR: 1715 cm MS: M* at m/z 114, a-cleavage fragment at m/z 71. no McLafferty rearrangement…
A: Ketones contains the carbonyl group and carbonyl groups shows IR spectra around 1715cm-1. If two…
Q: IR: no peak near 3300 cm C5H6 В UV: Amax -230 nm H, , Pt A H2, Pt C H2, Pt C5H6 IR: ~3300 cm1, sharp…
A: Unsaturation in compound=2no. of C+2-no. of Hydrogen2…
Q: Analyze the following spectra. Propose a structure that fits the spectra. Show your analysis. The…
A: To analyze the given mass spectrum of a molecule having a molecular formula of C9H12O.
Q: Treatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula…
A: Given: When (CH3)2CHCH(OH)CH2CH3 is treated with TsOH, the products M and N having molecular formula…
Q: Which of these molecules best corresponds to the IR spectrum below? а) H3C. b) e) HO OCH, c) d) OH…
A:
Q: 'H NMR Spectrum (200 MHz, CDCI, solution) expansion 2.0 1.0 ppm TMS 10 9 8 7 6 4 2 1 8 (ppm) 3.
A:
Q: Specific Chemical Shift multiplicity Integration 4b # of 13C-NMR Proton (s, d, t, etc...) HC (ppm)…
A: The correct answer about NMR spectrum is given below
Q: B They 15 we o d from e unclonalty. Delm pro H.C 10 5 10 6 3 Chemical Shift (ppm) Splitting pattern…
A: Chemical shift value of a proton depends on the electronic environment. Greater is the electron…
Q: using the NMR spectrum tables, predict the shift for the carbon indicated by the arrows.
A:
Q: Which IR spectrum below is consistent with a carboxylic acid? 100 100 80 60 40 1500 1000 2500 2000…
A: This question is related to IR spectroscopy which is used to identify the structure of a molecule in…
Q: 13C NMR Spectrum (100.0 MHz, D20 solution) Y X proton decoupled 200 160 120 80 40 0 8 (ppm) 3 'H NMR…
A: Degree of unsaturation (DU) = (2C + 2 + N –H – X)/2
Q: 'H NMR Spectrum (200 MHz, CDCI, solution) expansion 2.0 1.0 ppm TMS 4 1 8 (ppm) 10 7 2. 3.
A: DBE = (C+1)-H/2 = (4+1)-8/2 = 5-4 = 1 Hence, it is an aliphatic compound. Chemical shift value of…
Q: 1. PBR3 IR (cm-1): 3480 (broad), 2800-3000, 1708 (strong) HO MS (m/z): 86 (molecular ion) 2. но 3.…
A: There is error in your 5th step. You can check out 2 step to get your mistake.
Q: Base peak m/z (Dalton) Relative intensity of mlz=97 peak Product Molecular ion (Dalton) А B C D
A: for the compound D, the following table is concluded on the basis of the mass spectrum:
Q: IR Spectrum L00 'H NMR Data 3000 Signal Ha Chemical Shift Integration 4H Multiplicity multiplet…
A: Due to DBE of seven indicates benzene ring (4), two carbonyl compounds (2 CO) and five member-ed…
Q: Which of the following best fit this spectroscopic data? All 'H NMR data shown as x.xx ppm…
A: At aromatic proton range we got two peaks i.e. two doublets . Thus compound must be para…
Q: 13C NMR Spectrum (50.0 MHz, CDCI, solution) DEPT CH CH, cCH solvent proton decoupled 200 160 120 80…
A: 13C-NMR: 5-signals indicates the five types of carbons. 1H-NMR: Three signals indicates three types…
Q: identify the corresponding peaks in each spectra. "CNMR IR 140 120 100 80 60 40 20 180 160 ppm…
A: We have to predict the structure from the given C-13 , 1-H NMR And IR spectra.
Q: 13C NMR Spectrum C3H;NO2 (100.0 MHz, D20 solution) DEPT CH, CH,t CH proton decoupled 200 160 120 80…
A:
Q: Which of the following IR spectra best match the expected product of the following reaction? OH 1)…
A: When aliphatic ester compound is treated with excess of MeMgBr followed by acidification then…
Q: Wittig Reaction *PPH3 H. NANH2 H. + Br + NH, + NaBr PPH3 H. THE + Ph,PO Assign the characteristic…
A: Wittig reaction is a C-C bond formation reaction. In this reaction a phosphorous ylide attacks a…
Q: Q1. Why is it common to use CCl4 as a solvent in IR spectroscopy?
A: Infrared spectroscopy is the study of measurement of infrared radiations with matter. The…
Q: MS: m/z: 102.1 (100.0%) and m/z =103.1 (6.5%) IR (cm-1): 3000-3300 (strong, broad), 2950; 1150…
A:
Q: Give one advantage of the proposed HPLC identification test over the current UV-Visible Spectroscopy…
A: We have been asked to give advantage of HPLC over UV-visible Spectroscopy method of amiloride…
Q: Table 6. MS of 1,1-Diethoxy-cyclohexane m/z Peak value & fragmentation group 6. 1. 7. 2. 8. 3. 9. 4.…
A: The instrumental method which is used to determine the structure of chemical compounds using…
Q: This spectrum shows the presence of a(n) group. INFRARED SPECTRUM 0.8 0.6 0.4 3000 2000 1000…
A: A question based on IR spectroscopy interpretation, which is to be accomplished.
Q: 11 The two 1H-nmr signal between 6.0- 7.0 (d and dt) is due to * 'H NMR Spectrum (200 MHz. CDCI,…
A:
Q: На Ha He Hb Compund D Table 04: Predicted NMR peak assignments data for compound D Predicted…
A:
Q: M peak at m/z 228, M + 2 peak at m/z 230 (~1:1 intensity) IR: 3400 – 2500 cm1 (intense, broad), 1710…
A: A 1:1 intensity peaks for M+ and M+2 indicates towards the presence of Bromine atom. This is because…
Q: Mass spectrum of p-bromoacetanilide 100 MS-NU-0171 80 - 60 40 20- 40 60 80 100 120 140 160 180 200…
A: Mass spectrometry is not a true spectroscopic method as it does not involve any electromagnetic…
Q: g) HOT h) OH Br 1) 2) CI CI CI DMAP 1) Mg(0), dry ether 2) Å 3) NH4CI, H₂O , Et3N Na₂Cr₂O7 H₂SO4,…
A: I have given the answer in the explanation section. Hope you will understand that.
Q: (a) (c) % Transmittance 12.4 Which IR spectrum is consistent with an alkene? 100 100 80 80 60 60- 40…
A: The main characteristic IR peak of alkene is a medium stretch in the range 3000 - 3100 cm-1 due to…
Q: EI-MS of ortho nitrotoluene shows a large fragment ion at m/z 120 .The EI-MS of Tri deuteron ortho…
A: The structures of ortho-nitrotoluene and trideuteroorthonitrotoluene are:
Q: NMR data Summary NMR solvent: DMSO-d6 'H NMR: Spliting (Multiplicity) Chemical Shift Integration 1H…
A:
Q: Yellow Dye Calibration Curve y = 254.78x- 0.1414 R? = 0.9792 2.5 2 1.5 1 0.5 0.002 0.004 0.006 0.008…
A: Given: Transmittance = 14.4 % And the calibration curve equation => y = 254.78 x - 0.1414 Where y…
Q: Analyze the following spectra. Propose a structure that fits the spectra. Show your analysis. Th…
A:
Q: 100 MS-NU-0661 80 60 40 20 10 20 30 40 50 60 70 80 90 100 m/z Relative Intensity
A:
Q: The treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formulaC5H10O) as one of the products.…
A: 1H NMR and IR spectroscopy are the techniques, which helps to determine the structure of the organic…
Q: c. In the brewing process, hops play an important role in imparting bitterness in the finished beer…
A: Based on the structure of given alpha acids, we have to determine whether we can analyze them…
Q: Table 1-H-NMR Chemical Shift (ppm) Multiplicity Integrations Fragments 9.398 1H 6.479 1H 2H 2.371…
A: Nuclear magnetic resonance (NMR) spectroscopy is used to determine the structure of the molecule.…
Q: spectrum. d. "CNMR spectra cannot normally be integrated without the use of a specialized NMR…
A: The answers the following questions are:- d) 1,1-dimethylcylochexane at temperatures that prevent…
Q: OH NH2 X Y .8 .6 .4 .2 .0 з000 2000 1000 Waven umber (cm-1) 00
A: The given ir spectrum is for benzophenone(Z) because the peak at 1701 cm^-1 is only possible for the…
Q: Yield [g and %] Compound Name (2-2-(3,4- dmethontenadidepe- 23 Structure dihydro-1Hinden-1.one…
A:
Q: Which of the following best fit this spectroscopic data? All 'H NMR data shown as x.X ppm…
A: From the given spectrum it is clear that there is no peaks in the aromatic region. Hence the…
7
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- From the proton NMR spectrum in Figure 19-44, deduce the structure of this hydrocarbonProvide a structure that is consistent with the data below: - C6H12O2 - IR (cm-1): 2880-2980, 1738(strong) - 1HNMR (ppm): 5.0(1H, sept), 2.3(2H, q), 1.2(6H, d), 1.1 (3H, t)13 - CNMR (ppm)(proton coupled, DEPT): 174(s, none), 67(d, positive), 28(t, negative), 21(q, positive), 9(q, positive) (The attached image to this question is an incorrect answer, please identify the mistakes made in the given answer and please explain how to get the correct answer. )Rate = -(Delta[RCl])/Delta t andln[RCl]t=-k*t+ln[RCl]0 < [R-Cl]-time equation Test tube A = 0.30 of 0.10M NaOH(aq), 6.70ml DI H2O & 1 drop of bromophenol bue Test Tube B = 3.00M of 0.10M R-CL in acetone. Mix together in 3 water baths Bath A Trial 1= 20C, 53.6s Trial 2= 20C, 49s for color to changeBath B Trial 1= 32C, 18s Trail 2= 32C, 20s Bath C Trial 1= 10C, 204s Trial B= 10C, 200sQ.6 Rearrange the Arrhenius equation into a linear form relating k and TQ.6a To determine Ea, a graph of (xaxis) ____ (in units of ___) vs. (yaxis) ____ needs to be made. This will yioeld a _____ line with a slope = ____ Q.6b enter values used in the table Q.6c Determine the exp. values of Ea with correct sig figs and units
- Please help me analyze this 1H and 13C Acetylferrocene NMR data. I am not sure how to read the peaks and what information they give.The leaves of the Brazilian Tree Senna multijunga contain a number of pryidine alkaloids that inhibit acetylcholinterinase. Two recentyl isolated isomeric compounds have the strcture have the strcture shown below. (NOTE: M=293) Use the mass spectral data provided to determine the precise location of the hydroxyl group in each isomer. Isomer A: EI-MS, m/z(rel. int): 222(20), 150(10), 136(25), 123(100) Isomer B:EI-MS, m/z(re;. int): 236(20), 150(10), 136(25), 123(100)1. For each set of spectral data, propose an acceptable structure that is consistent with the data. a. MS: M+ m/z=71, IR: 3340, 2960, 2870 cm-1 , 13C NMR: 47.20 (CH2), 25.64 (CH2) ppm b. MS: M+ m/z= 102, IR: 2960, 2870 cm-1, 13C NMR: 72.73 (t), 23.20 (t), 10.80 (q) ppm c. MS: M+ m/z=74, IR: 3310, 2960, 2870 cm^-1, 13C NMR: 62.44 (down), 34.86 (down), 18.98 (down), 13.88 (up) ppm (DEPT-direction) d. MS: M+ m/z=92 (M:M+2=3:1), IR: 2960, 2870 cm^-1, 13C NMR: 67.29 (C), 34.46 (CH3) ppm e. MS:M+ m/z=70, IR: 3070, 2960, 2870, 1666 cm^-1, 13C NMR: 138.92 (t), 114.26 (d), 35.96 (t), 22.17 (t), 13.59 (q) ppm f. MS: M+ m/z= 72, IR: 2960, 2870 cm^-1, 13C NMR: 31.68 (t), 29.80 (d), 22.20 (q), 11.69 (q) ppm g. MS: M+ m/z= 84, IR: 2960, 2870, 1734 cm^-1, 13C NMR: 220.16 (C), 38.30 (CH2), 23.24 (CH2) ppm h. MS:M+ m/z= 138, (M:M+2=1:1), IR: ~3010(broad), 1715 cm^-1, 13C NMR: 173.61(s), 25.21(t) ppm i. MS:M+ m/z=88, IR: 2960, 2870, 1736cm^-1, 13C NMR: 170.95 (C), 60.34 (CH2), 20.98 (CH3), 14.23 (CH3)…
- ORGO II NMR please help fill out the chart and label. my unknown is C6H10O.EI-MS of ortho nitrotoluene shows a large fragment ion at m/z 120 .The EI-MS of Tri deuteron ortho nitrotoluene does not show any peak at m/z 120 but does have a peak at m/z 122 .Show the fragmentation pattern that accounts for the above observation.Please use 3 sig figs, 0.08206 for R, and 1C = 273.15K. Solve it within 30-40 mins I'll upvote your answer
- Answer Q40, 41, 42 showing detailly all explanationsPotansiyometride, aşağıdakilerden hangisi standart elektrottur? A. Kalsiyum elektrodu B. hidrojen elektrodu C. bakır elektrodu D. potasyum elektrodu Permanganat iyonunun mangan(II) iyonuna dönüştüğü titrasyonun dönüm noktası tayininde A. Kırmızı veya mavi turnosol kağıdı kullanılır B. pH probu kullanılır C. metil oranj belirteci eklenir D. permanganat iyonunun pembe kaybolur …………………………………….. gives the relative response of an ion-selective electrode to different species with the same charge.A. Transition coefficientB. ion coefficientC. counterion coefficientD. selectivity coefficientinterpret NMR spectra and report NMR data stating, in this order: nucleus (1H or 13C), solvent, machine frequency, peaks in order from high to low chemical shift including multiplicity, coupling constants and integration

