
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
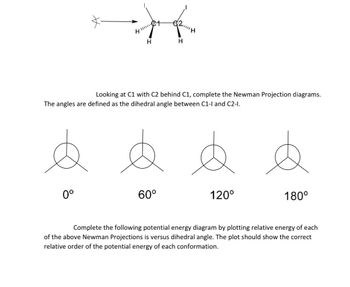
Transcribed Image Text:HI!!!!!
0°
H
€2H
Looking at C1 with C2 behind C1, complete the Newman Projection diagrams.
The angles are defined as the dihedral angle between C1-1 and C2-1.
60°
H
120°
180°
Complete the following potential energy diagram by plotting relative energy of each
of the above Newman Projections is versus dihedral angle. The plot should show the correct
relative order of the potential energy of each conformation.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using solid and dashed wedges, draw the structure of (1S, 2S, 4R, 5R)-1,5-dibromo-2,4- dichlorocyclohexane, then draw the most stable conformer.arrow_forwarduarrow_forward3.) 2-Methylbutane (isopentane) is used, in conjunction with liquid nitrogen, to freeze tissues for cryosectioning in histology. Below is the potential energy diagram for all the conformers of 2-methylbutane. Provide structures that match structure A, B, and C in the boxes provided. B F IM C A potential energy Conformer A 0° D angle of internal rotation Conformer B E F H 360° (=0°) Conformer Carrow_forward
- Please don't provide handwritten solution ...arrow_forwardOrganic Chemistry Мaxwell presented by Macmillan Learning Complete the most stable chair conformation of trans-1-tert-butyl-3-methylcyclohexane by filling in the missing atoms. Use the numbering provided on the ring. H Answer Bank 1 H. methyl tert-butyl H H H- 4 3 H H. CO 2.arrow_forward1) For the molecules below draw a Newman projection looking down the indicated bond (1→2). Your Newman projection needs to be in the same conformation that shown in the structural formula. a) b) Br, 1 2 H H view HO viewarrow_forward
- I need help with problem 4.arrow_forwardConvert the following structures into line structures (skeletal). Be sure to include correct stereochemistry. H₂C H₂C -CHICHI CH₂ Br H₂C H CH₂CH₂ H₂C H₂CH₂C CH₂ H CH₂CH₂ H Br H- CH₂ -H CH₂ CH₂CH₂ H CI -CI CH,CH.CH,arrow_forwardAssume that you have a variety of cyclohexanes substituted in the positions indicated. Identify the substituents as either axial or equatorial. For example, a 1,2-cis relationship means that one substituent must be axial and one equatorial, whereas a 1,2-trans relationship means that either both substituents are axial or both are equatorial. a) 1,4-cis-disubstituted b) 1,6-cis-disubstituted c) 1,2-cis-disubstituted Submit Answer Try Another Version 6 item attempts remaining optioins fill please both axial only both equatorial only both axial or both equatorial one axial-one equatorialarrow_forward
- 4 Practice 2 3-9 4.8 Conformational Analysis of Butane The graph below represents the relative energies of the conformations of butane: ww B Relative energy -1- —— 0° 60° 120° 180° 240° 300° 360° Angle of rotation Select the Newman projection that corresponds with position C on the graph (an angle of rotation of 120 degrees). HA 3 H I I I I # CH3 CH 3 CH 3 CH3 -I H HH CH3 CH3 CH3 80⁰0 CH3 H I I Н 4 000 000 % 6 MacBook Air 7 8 DIL FB 9 0arrow_forwardNonearrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY