Chapter32: Gas Chromatography
Section: Chapter Questions
Problem 32.22QAP
Related questions
Question
According to the reaction and NMR what is the product?
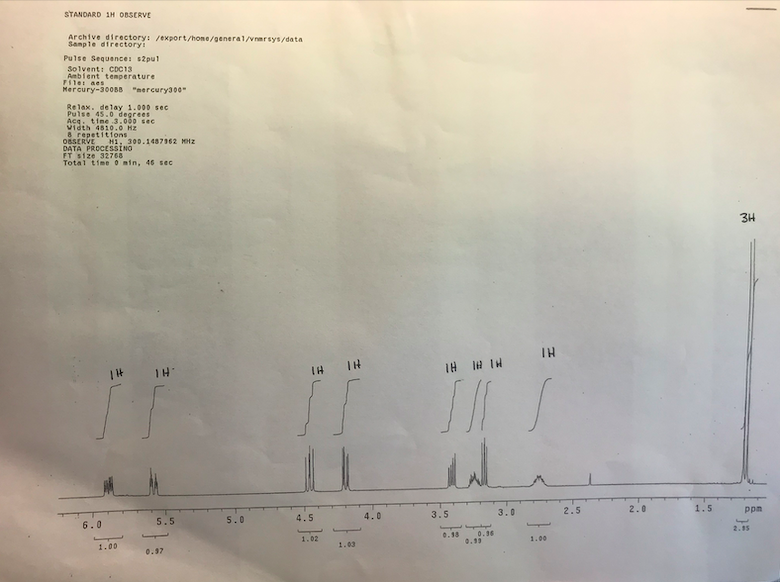
Transcribed Image Text:STANDARD 1H OBSERVE
Archive directory: /export/hone/general/vnarsys/data
Sample directoryi
Pulse Sequence: s2pul
Solvent: CDC13
Ambient temperature
Filer aes
Mercury-300BB "mercury300
Relax. delay 1.000 sec
Pulse 45.0 degrees
Acq. time 3.000 seC
Vidth 4510.0 Hz
8 repetiti1ons
OBSERVE
DATA PROCESSINO
FT size 32768
Total time 0 min, 46 sec
H1, 300.1487162 MHZ
3H
TH
IH
IH # IN
3.5
3.0
2.5
2.0
1.5
ppm
5.5
5.0
4.5
4.0
6.0
2.15
0.36
0.19
0.18
1.02
1.00
1.00
1.03
0.97
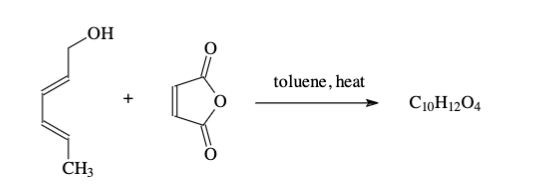
Transcribed Image Text:LOH
toluene, heat
C10H1204
CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



