Living By Chemistry: First Edition Textbook
1st Edition
ISBN:9781559539418
Author:Angelica Stacy
Publisher:Angelica Stacy
ChapterU4: Toxins: Stoichiometry, Solution Chemistry, And Acids And Bases
SectionU4.25: Mole Tunnel: Stoichiometry
Problem 3E
Related questions
Question
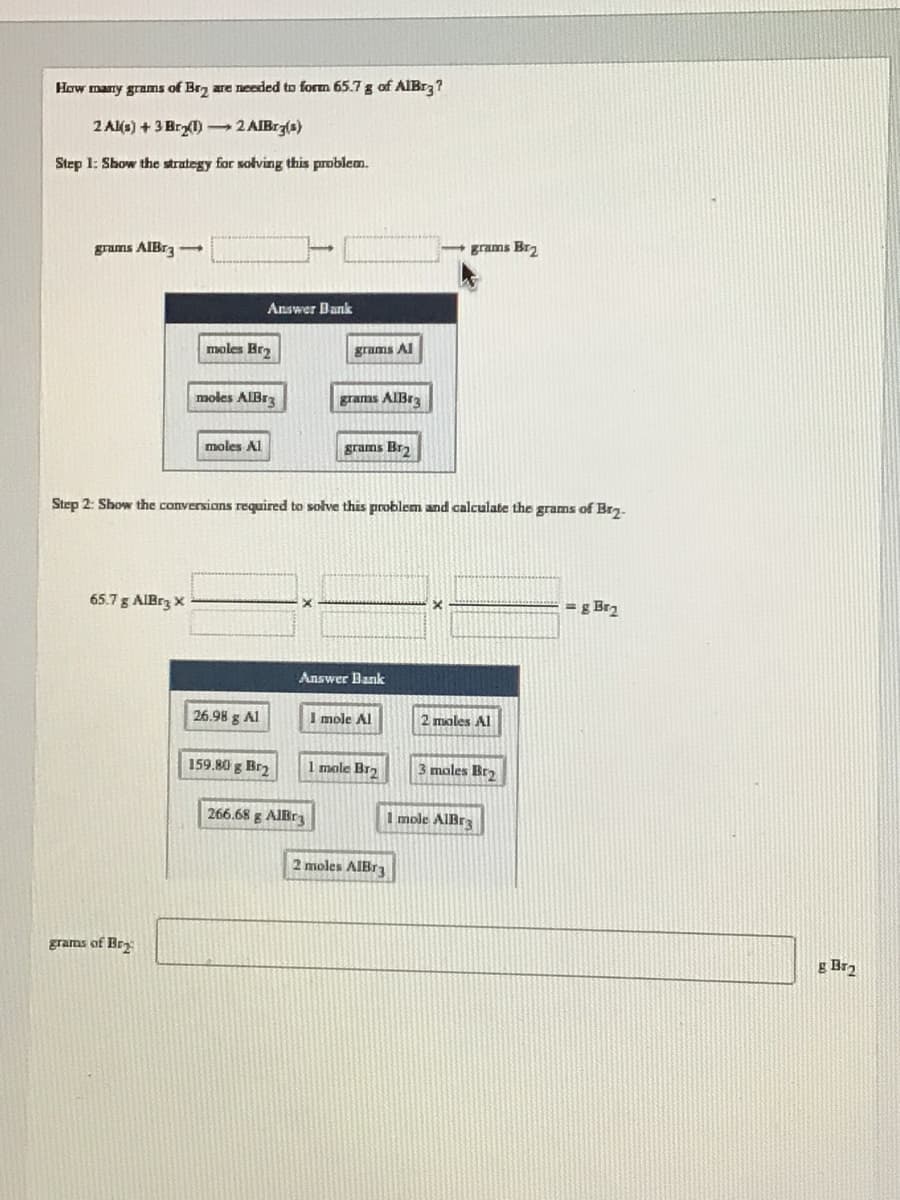
Transcribed Image Text:How marry grams of Br, are needed to form 65.7 g of AIBr3?
2 Al(s) + 3 Br) 2 AIBrz(s)
Step 1: Show the strategy for solving this problem.
grams AIBr3-
grams Br2
Answer Bank
moles Bra
grams Al
moles AIBr3
grams AIBra
moles Al
grams Br,
Step 2: Show the conversions required to solve this problem and calculate the grams of Brg.
65.7 g AIBrz X
g Bra
Answer Bank
26.98 g Al
I mole Al
2 moles Al
159.80 g Br2
1 mole Brz
3 moles Bra
266.68 g AIBr
I mole AlIBr3
2 moles AIBr3
grams of Br:
g Br2

Transcribed Image Text:How many grams of Cl, are needed to form 83.4 g of AICI3?
2 Al(s) + 3Clg) 2 AlCiz(s)
Step 1: Show the strategy for solving this problem.
grams AICI2
grams C,
Answer Bank
grams AICI,
grams Al
moles Cl
males Al
grams Cl,
moles AICI3
Step 2: Show the conversi
required to solve this problem and calculate the grams of Cl2
83.4 g AICI; x
= g Cl,
Answer Bank
26.98 g Al
I mole Al
2 moles Al
70.90 g Cl2
1 mole Cl
3 moles Cla
133.33 g AICI3
I mole AICI3
2 moles AICI
grams of Cl
g Cl2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning