htProblemID=D143793425&offset%3Dnext CHE154-H Gen Chem I Bronikowski S20 ints Part A a solution that is 0.195 M in HC2 H3O2 and 0.100 M in KC2H3 O2 Express your answer to two decimal places. • View Available Hint(s) AZ¢ pH = %3D
htProblemID=D143793425&offset%3Dnext CHE154-H Gen Chem I Bronikowski S20 ints Part A a solution that is 0.195 M in HC2 H3O2 and 0.100 M in KC2H3 O2 Express your answer to two decimal places. • View Available Hint(s) AZ¢ pH = %3D
Chapter28: Atomic Spectroscopy
Section: Chapter Questions
Problem 28.8QAP
Related questions
Question
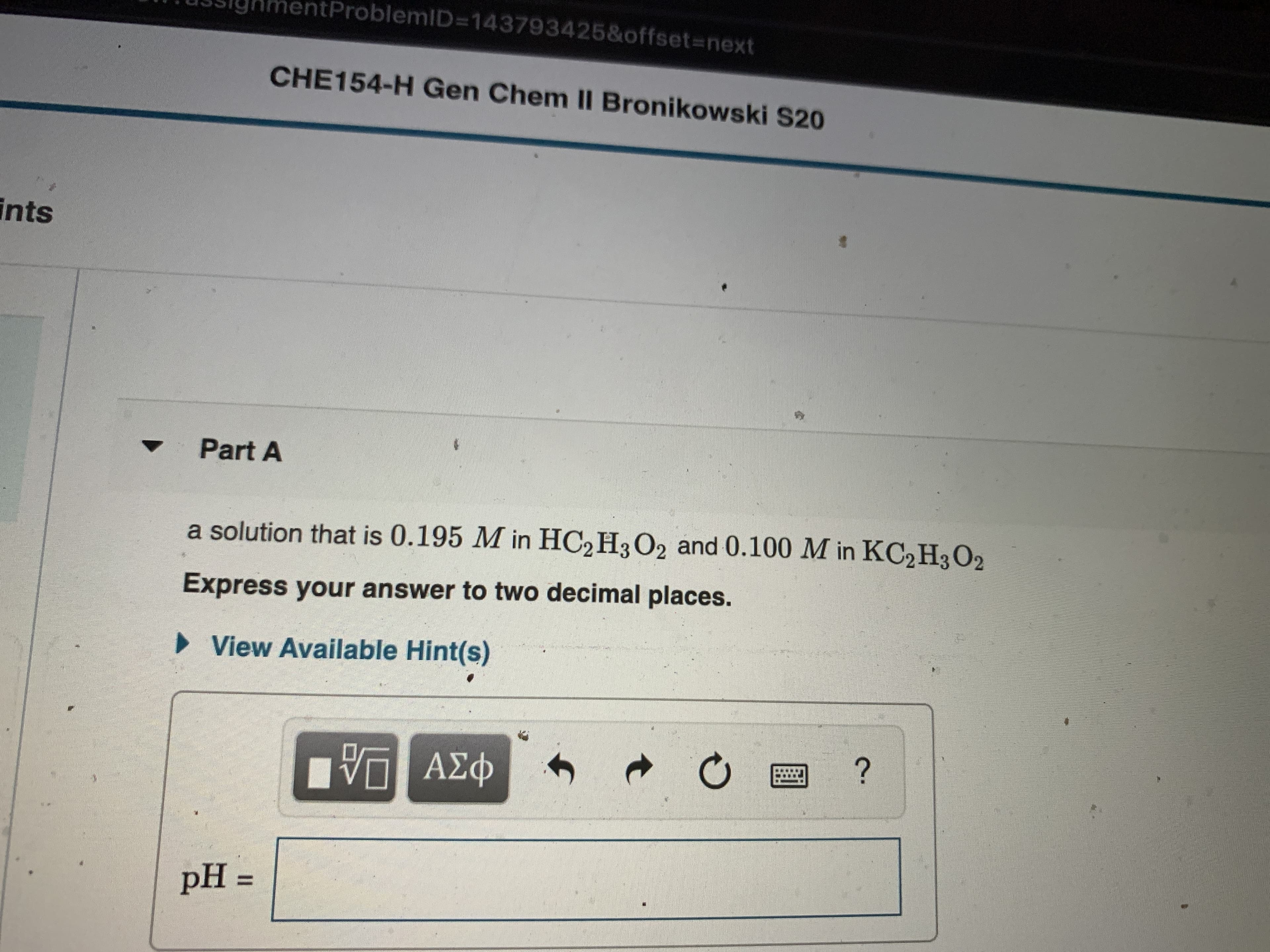
Transcribed Image Text:htProblemID=D143793425&offset%3Dnext
CHE154-H Gen Chem I Bronikowski S20
ints
Part A
a solution that is 0.195 M in HC2 H3O2 and 0.100 M in KC2H3 O2
Express your answer to two decimal places.
• View Available Hint(s)
AZ¢
pH =
%3D
Expert Solution
Step 1

Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning