Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter6: Alkanes & Alkenes
Section: Chapter Questions
Problem 2CTQ
Related questions
Question
What is the mechanism to get from the reactant to the products

Transcribed Image Text:Functional Group Transformations
Typical Reagents Compound
Class
Starting
Compound
and Reaction
Class
Conditions
Formed
HX (1 equiv), cold
(7)
Conjugated diene
1,2-Adduct
HX (1 equiv), warm
(8)
Conjugated diene
1,4-Adduct
Expert Solution
Introduction
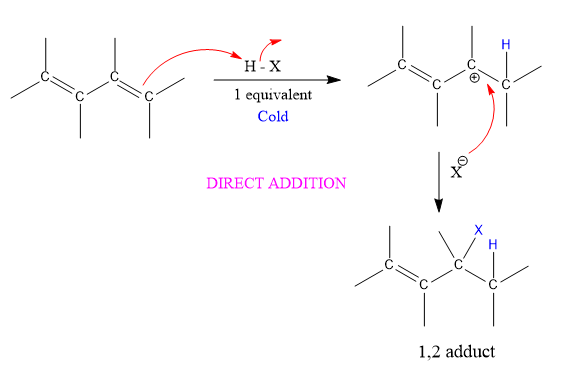
Addition of HX to conjugated alkenes occurs via two modes:
- Direct addition (1,2 addition): HX adds directly across the ends of a C=C bond.
- Conjugate addition (1,4 addition): HX adds across the ends of conjugated system.
The distribution of the products depends on the reaction conditions (temperature).
1,2 adduct
At low temperature, the reaction is under Kinetic control (rate, irreversible conditions) and the major product is that from fastest reaction, that of the bromide with the secondary cation.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
