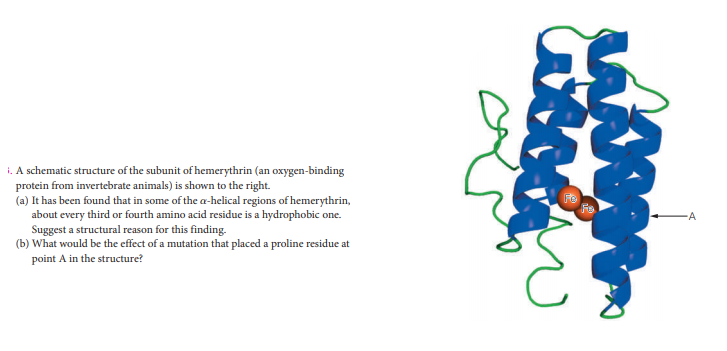
i. A schematic structure of the subunit of hemerythrin (an oxygen-binding protein from invertebrate animals) is shown to the right. (a) It has been found that in some of the a-helical regions of hemerythrin, about every third or fourth amino acid residue is a hydrophobic one. Suggest a structural reason for this finding. (b) What would be the effect of a mutation that placed a proline residue at point A in the structure?
Q: How would chymotrypsin's catalytic triad be affected by extremely low and extremely high pH values…
A: Enzymes are catalysts that increase the rate of the chemical reaction without undergoing any change…
Q: 11 12 Ala – Met -Cys -Lys – Trp –Glu –Ala – Gln –Pro –Asp – 13 14 15 16 17 18 19 20 21 22 23 24 25…
A: Structure of proteins are classified as different levels. They are, primary structure, Secondary…
Q: 18. Chaperones are frequently associated with polypeptides as they are being synthesized from the…
A: Chaperons are proteins that are primarily concerned with protein folding. It prevents the…
Q: Consider the peptide with the sequence SANTACLAUSISASTALKER. Assume this entire pepide were a single…
A: Proteins are the heteropolymers of amino acids. The proteins are made up of twenty standard amino…
Q: (b) r , The following polypeptide sequence has three alpha helices and two B turns. Based on what we…
A: An amino group and an acid group-containing organic molecules are called Amino acids.…
Q: The α and β subunits of hemoglobin bear a remarkable structural similarity to myoglobin. However, in…
A: Both Hemoglobin and Myoglobin share structural similarity by having α-helical secondary structure.…
Q: Ornithine is structurally similar to lysine except ornithine’s side chain is one methylene group…
A: Ornithine plays important role in urea cycle. Ornithine is structurally similar to lysine except…
Q: Based on the structural properties of your protein above , how resistant (or sensitive) would your…
A: Salmonella enterica serovar typhimurium produces 2-keto-3-deoxygluconate 6-phosphate in the…
Q: The sequence of a 29 aa long peptide can be determined from the following data. a) Treatment of the…
A: Amino acids are the molecules that join to form a protein structure. The bond formed between two…
Q: As an example, consider albumin, a protein made of a single polypeptide weighing 66,000 daltons (66…
A: Gel electrophoresis if the technique of separation of molecules on the basis of their size and…
Q: Most proteins contain more than 100 amino acid residues. If you decided to synthesize a “100-mer,”…
A: Protein is a macronutrient that is essential to building muscle mass.
Q: . Disulfide bonds have been shown to stabilize proteins (i.e., make them less likely to unfold).…
A: In disulfide linkage, the two cysteine amino acids are joined by a disulfide bond.
Q: Disulfide bonds have been shown to stabilize proteins (i.e., make them less likely to unfold).…
A: Disulfide bonds are considered the covalent bond, which is formed between the two cysteine residues.…
Q: Leu-Trp-Phe-Met-Ala-Ile-Val- Draw the structure of the peptide at pH7.4. and Indicate…
A: In the endoplasmic reticulum process of protein folding occurs and it is a very important…
Q: Remembering that the amino acid side chains projecting from each polypeptide backbone in a β sheet…
A: Amongst secondary structures of polypeptide chains made of amino acids, β-sheets are a more spacious…
Q: Which amino acid replacement would result in the biggest structural change in a protein if a Cys…
A: Amino Acids Introduction Proteins are polymers made up of nitrogenous chemicals known as amino…
Q: The effect of base-pair substitution mutations on protein function varies widely from no detectable…
A: It is because of the protein functionality isn't affected by the mutations occurred in the intron…
Q: Two peptide sequences are shown below. Peptide A: N-term – GDL – C-term Peptide B: N-term – GTL –…
A: A peptide is a short chain of amino acids that contains one or more amino acids. Peptide bonds…
Q: 5' UGG CAA UCC UAC GAU 3' < Is it possible for a single base pair substitution to cause a truncation…
A: Yes, it is possible for a single base pair substitution to cause a truncation in the peptide as…
Q: a. Suppose that the R group of a histidine residue in a protein in its native tertiary structure is…
A: In the protein, the nonpolar amino acids are usually present in the inner hydrophobic region. Also…
Q: Treatment of a polypeptide by 2-mercaptoethanol yields two polypeptides that have the following…
A: Amino acids are the building blocks of proteins. The amino acid polymer which joins together with…
Q: Let’s consider histidine as a free amino acid in aqueous solution. a) Draw the most likely…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Consider the following peptide sequence: Lys-Leu-Ile-Cys-Lys What is the net charge of this…
A: Physiological pH Physiological pH, where pH means power hydrogen, is the acid base balance in the…
Q: Imagine a globular protein with a histidine that is involved in a salt bridge to an aspartic acid,…
A: Globular proteins are a group of protein sub-types. They form colloids and are somewhat soluble in…
Q: If a Cys residue in a protein was replaced with either Ser or Phe amino acid, which substitution…
A: Amino acids Proteins are the polymers of nitrogenous compounds called amino acids. Each amino acid…
Q: f an Arg residue in a protein was replaced with either Lys or Glu amino acid, which substitution…
A: Proteins are large biomolecules that compose structural and motor elements of a cell, and also as…
Q: The amino acid that is often used as a tag (in a run of 6-8 of this amino acid in a row) for protein…
A: Living cells have a complex system for protein synthesis. Ribosomes synthesize amino acid chains.…
Q: Molecularly speaking, what is the difference with the sugar residues between DNA and RNA a. The…
A: DNA stands for the deoxyribonucleic acid and RNA stands for the ribonucleic acid . In most of the…
Q: Explain why a 50S ribosomal subunit and a 30S ribosomal subunit combine to form a 70S subunit,…
A: Ribonucleoproteins form the basic unit of ribosomes and they are a complex of RNA and proteins. They…
Q: If the molecule can inhibit toxic effects at 0.5mM against gastric carcinoma cells, does it mean…
A: IC 50 is half maximal inhibitory concentration which measures potency of substance to inhibiting…
Q: Apamine is a small protein toxin present in the venom of the honeybee. Ithas the…
A: Apamin is an 18 amino acid globular peptide neurotoxin found in apitoxin.
Q: Treatment of a polypeptide by 2-mercaptoethanol yields two polypeptides that have the following…
A: An amino acid is a natural atom that is comprised of a fundamental amino gathering (−NH2), an acidic…
Q: Consider two proteins, Protein A and Protein B: A is a monomeric protein, whereas B is a subunit of…
A: Proteins are usually made from basic amino acid structures. Thus amino acids are the building blocks…
Q: Propose an experimental strategy to characterize the sequence and linkages of different glycan…
A: Glycans are chain of carbohydrates/sugar molecules (like galactose, mannose, fucose, N-acetyl…
Q: Oftentimes, the major challenge in the determination of protein structure via X-ray crystallography…
A: X-ray crystallography of a protein is used to obtain the three-dimensional structure of the…
Q: Do you expect a Pro → Gly mutation in a surface-loop region of a globular protein to be stabilizing…
A: Mutation : It is an alteration in the nucleotide sequence of the genome of an organism, virus, or…
Q: Suppose that there is a protein consisting of two polypeptide chains with the given sequences in the…
A: Proteins are the polymers of amino acids, joined together by peptide bonds. The amino acid with the…
Q: 3. a. Draw the structure (at physiological pH) of the peptide PEACH (note that this is one letter…
A: Hi! Thanks for your question. As you have posted multiple questions and have not mentioned which one…
Q: Draw a complete structural representation for the tetrapeptide Phe-Met-Tyr-Asn (NOTE:use the…
A: Tetrapeptide is the combination of four amino acids gives linear tetrapeptide, in which three…
Q: Lys and Arg Glu and Lys Pro and Asp Among these amino acid combinations listed above, only the…
A: Proteins are composed of a linear chain of amino acids attached together via peptide bonds. All…
Q: Consider the dipeptideVal–Pro: What amino acid is the N terminal aminoacid? What amino acid is the…
A: The dipeptide mentioned in the question is composed of valine and proline. it is called dipeptide…
Q: (a) Tropomyosin, a 70-kDa muscle protein, is a two-stranded α -helical coiled coil. Estimate the…
A: Tropomyosin is a two-stranded alpha-helical , coiled coil protein found in actin-based…
Q: 17. How would you computationally predict the thermodynamic stability of a long, extended RNA helix?…
A: Thermodynamic stability is the state when the structure is in a conformation that has the lowest…
Q: Consider the phenolic hydroxyl group of a particular Tyr residue in a protein. Suppose the hydroxyl…
A: The unfolded region protein is hydrophilic and protein resent in the folded region are called…
Q: Hydrogen bonds are usually 2.7-3.3 angstrom long. According to this information, do you think there…
A: There is no direct hydrogen bonding between H840 and the phosphodiester bond that links dG13 and…
Q: Since mammalian DNA contains roughly 25% thymine residues, why do mammalian cells need a thymine-DNA…
A: Introduction: DNA is the type of nucleic acid that is present in the nucleus of the cell. It is the…
Q: Many plasma proteins found in an aqueous environment are globular in shape. Which amino acid side…
A: The outer surfAce of the globular protein is coated with hydrophilic protein is coated side chains,…
Q: 3A. For the following tripeptide, provide its full name, abbreviated name, describe what type of…
A: Proteins are the polypeptide molecule that is joined together by the peptide molecule.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Consider the phenolic hydroxyl group of a particular Tyr residue in a protein. Suppose the hydroxyl group in the unfolded protein in aqueous solution, where the group is exposed to H2O, has a pKa of 10.0. If that group is found in a hydrophobic environment in the interior of the protein when the protein is folded into its native tertiary structure, would you expect the pKa of the phenolic hydroxyl to be higher or lower in the folded protein interior than in H2O? Explain your reasoning.Patients suffering from sickle cell anemia have a mutation in the gene that codes for one of the hemoglobin chains, in which a single glutamate is replaced by a valine. Propose an explanation for why this substitution has such a striking effect on protein structure and function with explanation. Suggest two other amino acids that would be less likely to cause such serious impairment of hemoglobin function if each was substituted for glutamate. Briefly explain your answer.Both hsp60-like and hsp70 molecular chaperonesshare an affinity for exposed hydrophobic patches on pro-teins, using them as indicators of incomplete folding. Whydo you suppose hydrophobic patches serve as critical sig-nals for the folding status of a protein?
- Considering the chemical characteristics of the amino acids valine and glutamic acid (see Figure 5.14), propose a possible explanation for the dramatic effect on protein function that occurs when valine is substituted for glutamic acid.GT 3 A. Write the structure of the pentapeptide GLDSC. B. What is the complete name of this pentapeptide? Show a tertiary structure of ACGGC after a disulfide bond forms. A sample of an unknown peptide was divided into two aliquots. One aliquot was treated with trypsin; the other was treated with cyanogen bromide. Given the following sequences (N-terminal to C- terminal) of the resulting fragments, deduce the sequence of the original peptide. Trypsin treatment Asn-Thr-Trp-Met-lle-Lys Gly-Tyr-Met-Gln-Phe Val-Leu-Gly-Met-Ser-Arg Cyanogen bromide treatment Gln-Phe Val-Leu-Gly-Met lle-Lys-Gly-Tyr-Met Ser-Arg-Asn-Thr-Trp-Met1. In a protein, why does when Ala is replaced with Ile, it loses its activity but when Lys is replaced by Arg and Leu to Ile, it only has little effect on protein structure and function? Explain. 2. Why do proteins cannot be denatured reversibly when they are chemically altered to change the chemical composition of certain side chains? Explain.
- Consider a short peptide that forms an alpha-helix within a larger protein structure. Suppose that one glutamate residue at some specific position in the helix were mutated to a leucine residue. The mutation could either make the helix more stable, or less stable. i) Describe two situations in which a Glu-to-Leu mutation could make the helix more stable. ii) Describe two situations in which the Glu-to-Leu mutation could make the helix less stable. Explain briefly the basis for the stabilizing and destabilizing effect in all cases.Draw Glu-His-Trp-Ser-Gly-Leu-Arg-Pro-Gly peptide. What is the net charge of this peptide at pH 3, 8 and 11? What can you say about pI value of this peptide?1. In the separation of lysine from histidine using a sulfonated polystyrene column will it be easier to separate at pH 4 or pH 8 ? explain! 2. A peptide of the order Glx-Ala-Gly-Arg bound to a sulfonated polystyrene column at neutral pH. Is Glx Glu or Gln ? Explain !
- A hypothetical protein has a molar mass of 23,300 Da. Assume that the average molar mass of an amino acid is 120. • How many amino acids are present in this hypothetical protein? • What is the minimum number of codons present in the mRNA that codes for this protein? • What is the minimum number of nucleotides needed to code for this protein?Draw the structural formula for the pentapeptide segment being synthesized. (Val-Thr-Arg-Pro-Glu) Describe the attractive forces that the amino acid residue on the c-terminus might engage in.You are in the process of determining theamino acid sequence of a protein and must reconcile contradictory results. In one trial, you determine a sequence with glycine asthe N-terminal amino acid and asparagine as the C-terminal aminoacid. In another trial, your results indicate phenylalanine as theN-terminal amino acid and alanine as the C-terminal amino acid.How do you reconcile this apparent contradiction?

