Q: What is the correct order of stability in descending order for the conformations shown HIC H CH₂ n…
A: The stability of gauche conformers depends upon the steric interaction among two nearby groups.…
Q: OH HC t-Bu
A:
Q: Draw the stable conformation of the compound below.
A: In the given compound, the position of methyl, ethyl, and isopropyl decide the stability of the…
Q: Calculate the destabilization present in each eclipsed conformation. CH3. CH3 HCH3 b. a. CH3 H. CH3
A: Steric strain is an increase in energy when atoms are forced too close to one another. In eclipsed…
Q: OH CH3
A: In a chair conformation where the bigger substituents are present in equatorial position that…
Q: Calculate the destabilization present in each eclipsed conformation. (See attachment)
A: Given, The energy of eclipsed conformation is given below,
Q: Draw both chair conformations of compound M on the templates (X and Y) provided. Indicate which…
A: The position of groups on the chair confirmation can be determined as– Here all the substitutents…
Q: Which is the most stable chair conformer of the following compound? ON
A: For the chair conformations, the most stable conformer is the one in which the bulkier group…
Q: chair conformations of identiry more stable conformation.
A: Solution -
Q: Calculate the free-energy difference between the stable and unstable chair conformation of each the…
A: For substituted cyclohexane, the most stable chair conformation is the one when most of the…
Q: Given the planar trisubstituted cyclohexane below, fill in all boxes (with H, Cl or Br) to complete…
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: Refer to Compound X1 as shown below: но он HO 3 HO, HO
A: Chair conformation is diagrammatic representation of molecules in which atoms 2 , 3,5,6 lies in same…
Q: Which of the following conformations is the most stable? -CH3 S H₂C. CH3 CH3 CH3 -CH3
A:
Q: CH3 a) CI HO, HO" b) F Br HO c) Br 1101
A:
Q: H H H H H H H H H H. H. I or H II H H H. H
A: Torsional strain is the repulsion that arises between atoms or group of atoms when a molecule is…
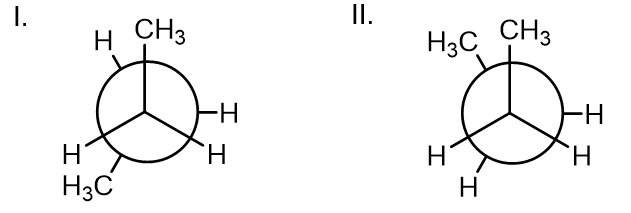
Q: ch of the following best explains the reason for the relative stabilities of the conformers vn? II.…
A: Strain cause by the repulsion of electrons in bonds . I is eclipsed conformation but anti-clinal…
Q: Which conformation in the picture attached represents the most stable ECLIPSED conformation of…
A: The arrangement of dihedral angle between the front bond and the back bond is 00 is known as…
Q: OH Draw both chair conformations of this compound and indicate which conformation is more stable.
A: Conformation of substituted cyclohexane:
Q: Select the lowest-energy conformation of the compound shown at the top. i Oi =: E: iii O iv V iv
A: Equatorial conformation is of lower energy than the “axial” conformation therefore equatorial…
Q: Which of the following is the most stable conformation of the following compound? но. CI CI но. Но.…
A:
Q: Which of the following is the most stable conformation of the following compound? HO CI HO. Но. HO.…
A: Given molecule : Most stable confirmation need to be write.
Q: Q6. Draw both chair conformations for the following compound (Carbons atoms are numbered in the…
A: In chair form we have 6 axial and 6 equatorial bonds. In general more bulky group tends to occupy…
Q: Which of the following chair conformation is the most stable? CI CI -CI ČI II II IV Gelect one:
A: The chair conformation is the most stable among all the forms of cyclohexane. There are two kinds of…
Q: Describe this conformation of butane. CH3 ный Ø ΗΨΗ H CH3 gauche eclipsed least-stable eclipsed anti
A: Conformation for butane: Generally there are 4 conformation for butane: fully eclipsed gauche…
Q: 4. Draw the most stable chair conformation for each of the following compounds: ОН (а) (b) (c) (d)…
A: In the chair conformation, the substituent may present at the axial or equatorial position. If the…
Q: Draw the two possible chair conformations of the (+)- B- Galactose molecule shown below, using the…
A: The given structure of (+)-β-Galactose is shown below.
Q: 86. Which is the other chair conformer of the following compound? A. В. C. D.
A: Cyclohexane is a cyclic compound containing six carbon atoms. Conformers are the structures which…
Q: Which of the following is the more stable chair conformation of the substituted cyclohexane shown in…
A: Given structure,
Q: Draw the most stable conformations of the following: HO Br Cy Br
A:
Q: Calculate the energies of the 4 conformers below:
A: The 4 different conformations of the molecule are given of which the relative energies have to be…
Q: 5. Draw the most stable chair conformation of the given trisubstituted cyclohexane? Br
A: Answer of this question:- The most stable chair confirmation is:-
Q: How many chlorines are axial in the most stable chair conformation of the molecule below? (enter 0,…
A: The equatorial position will be mainly for the bulky group while the axial position will be for the…
Q: Draw the two chair conformations of the following compound A, and indicate which is more stable.…
A: Six membered ring chair form is drawn for the stability of the compound. Stability of equatorial…
Q: Given the planar trisubstituted cyclohexane, fill in the missing substituents (with H or Cl) to…
A: Conformational isomers: The conformational isomers are formed by the rotation of a carbon-carbon…
Q: 6.4 whether the substituents are axial or equatorial. Indicate which chair conformation is For each…
A: cyclohexane can adopt two conformer chair and boat form
Q: What are the relative energy levels of the three staggered conformations of 2,3-dimethylbutane when…
A: Given: 3 Different conformation and some of the statements regarding it. To find: The correct…
Q: 2. Give the IUPAC name of this compound: CH,OCH:CH: CHCO 3. Draw the most stable chair conformation…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Rank these conformations from Teast to most Stable lless c more stably H CH3 Hj C HgC CH3 H. HgC
A:
Q: Circle the most stable conformer 3) Draw the two chair conformations for the compound shown below Cl…
A:
Q: Which of these is the most stable conformation of butane, looking down the C2-C3 bond? H;CCH3 CH3…
A: Given compound is butane. Given compound is in Newman conformation. Methyl (CH3 ) group must be
Q: 7. Shown on the right is the sawhorse projection of a conformer of compound HOLDEN. What are the…
A: In this question, we will see the type of strain present in this staggered conformation which…
Q: How many gauche butane interactions are present in this chair conformation? 신 01 ○3 ㅇㅇ ○ 2 04
A: Ans: 4 (four) The gauche interaction occurs in butane occurs when the two methyl groups have…
Q: CH3 CH- ČI ČI ČH3 ČI A
A: If the substituents are present at axial position, the energy is higher when compared to the…
Q: 6. The following compound exhibits two chair conformations. Draw them and indicate which one is more…
A: Here we are required to find the most stable chair conformation of given compound
Q: 20. Which of the following is a gauche conformation for butane? CH3 H, H CH3 CH3 CH3 H3C CH3 H, H YH…
A:
Q: For the compounds below, draw both chair conformations. of 4,5,6
A:
Q: Calculate the destabilization present in each eclipsed conformation.
A: Given,
Q: 5. Draw the most stable chair conformer of the compound shown below
A: Any chair conformation can have different isomers, depending on the position of substituents. A…
Q: 2) Draw the most stable chair conformer for the molecules below and explain why? b) Br CH3
A:
Q: Q6. Draw both chair conformations for the following compound (Carbons atoms are numbered in the…
A: Cyclohexane exists in chair conformation as shown below : There occurs ring flipping between these…
Which of the following best explains the reason for the relative stabilities of the conformers shown?
Step by step
Solved in 3 steps with 1 images


