Identify the nucleophile and electrophile in the following addition reaction of cyclohexanone I and cyclopropylamine II shown below. A) nucleophile = I; electrophile = || H2N II B) electrophile = I; nucleophile = || H
Identify the nucleophile and electrophile in the following addition reaction of cyclohexanone I and cyclopropylamine II shown below. A) nucleophile = I; electrophile = || H2N II B) electrophile = I; nucleophile = || H
Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.3: Stability Of Cycloalkanes: Ring Strain
Problem 8P: Each H↔H eclipsing interaction in ethane costs about 4.0 kJ/mol. How many such interactions are...
Related questions
Question
100%
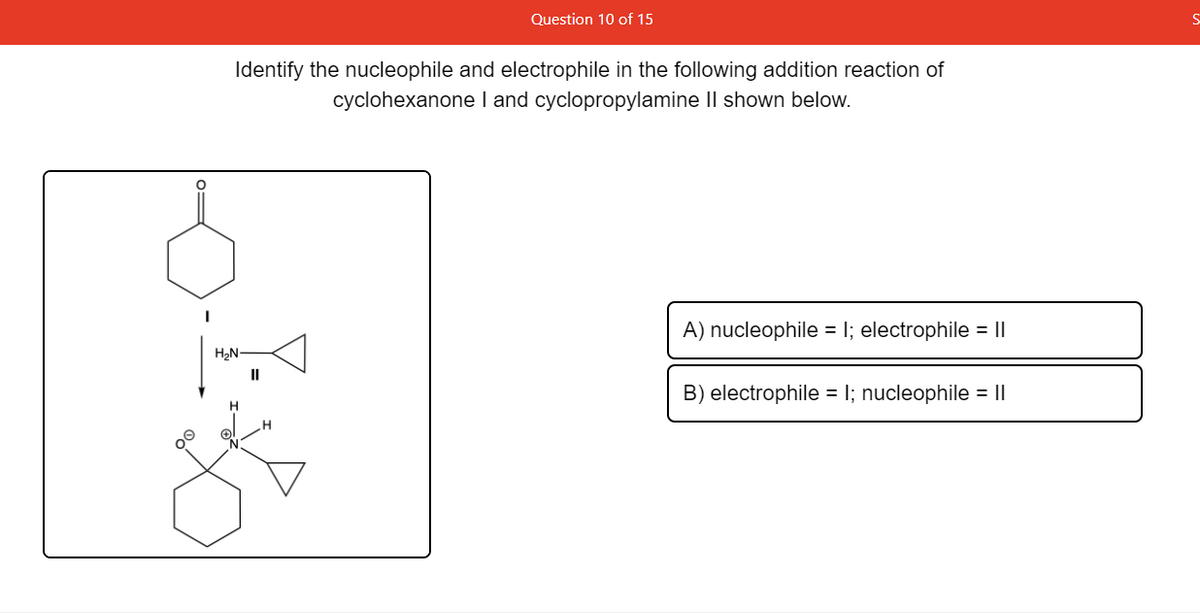
Transcribed Image Text:Question 10 of 15
Identify the nucleophile and electrophile in the following addition reaction of
cyclohexanone I and cyclopropylamine Il shown below.
H2N
A) nucleophile =
electrophile = ||
II
B) electrophile = I; nucleophile = ||
00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

