
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Importance of melting point in Dehydration Reaction Of
2-Methylcyclohexanol And Phosphoric Acid.
Does it help us predict anything?
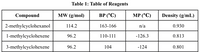
Transcribed Image Text:Table 1: Table of Reagents
Compound
MW (g/mol)
ВР (°С)
MP (°C)
Density (g/mL)
2-methylcyclohexanol
114.2
163-166
n/a
0.930
1-methylcyclohexene
96.2
110-111
-126.3
0.813
3-methylcyclohexene
96.2
104
-124
0.801
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- I know there is: 1. Carboxylic group 2. Ester. 3. Ortho-disubstituted phenyl. But my answer is still partial.arrow_forwardGive detailed Solution with explanation needed..don't give Handwritten answerarrow_forwardConsider propanoic (CH3CH2CH2CO2H) acid and pentanoic (CH3CH2CH2CH2CO2H) acid and the corresponding alcohols with the same number of carbons, propanol (CH3CH2CH2COH) and pentanol (CH3CH2CH2CH2COH). Select the class of compound that is more soluble. {one answer] Select the best explanation for your answer to the previous question. [a second answer] Group of answer choices a. The solubility of alcohols in water decreases with increasing chain length. b. Propanoic acid and pentanoic acid are more soluble in water than the corresponding alcohol. c. Carboxylic acids are generally more soluble in water than alcohols of the same chain length because of increased hydrogen bonding with the -COOH functional group. d. Propanol and pentanol are more soluble in water than the corresponding carboxylic acid e. The solubility of carboxylic acids in water decreases with increasing chain length. f. Alcohols are generally more soluble in water than carboxylic acids of the same…arrow_forward
- what is the 1UPAC Neme tos the fullwing Malecole? OH 1. 8-Ethyl- S-hydronyhexenal 2. 3- E thylhenanal - 3-01 g 4- E thyl -2-hydroxyhevanal 4- Ethyl-2-hydroxpentamel 5. Nave of thesearrow_forwardWhat are 5 reactions that can create benzoic acid? Draw them outarrow_forwardUsing the balanced equation in the first picture, write balanced equations for all the aldehydes/ketones from the list in the second picture.arrow_forward
- Draw the structural formulas of the "parent" carboxylic acid and the "parent" alcohol reacted to make each of the follow- ing esters: Cl a. CH-CH-C-0-CH,CH, b. CH,CH,-O-C- CH, O 15 CH,C-C-O-CH, C. CH, d. CH,CH,CH,СН - О—С—CH,arrow_forward1. The reactant (2-methyl-2-butanol) has a limited solubility in water at room temperature. 10.0 mL of 2-methyl-2-butanol will dissolve in ~ 80 mL of water. In this experiment, 10.0 mL of 2-methyl-2-butanol readily dissolved in 20 mL of 12 mol/L aqueous HCl. a. Why does this compound exhibit some solubility in water? Consider intermolecular forces and include a diagram to illustrate the relevant intermolecular forces. Why is it not completely miscible in water (in all proportions)?arrow_forwardCan you please answer this and explain why the correct answer is correct?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY