In 1911, Ernest Rutherford and his assistants Geiger and Marsden conducted an experiment in which they scattered alpha particles (nuclei of helium atoms) from thin sheets of gold. An alpha particle, having charge +2e and mass 6.64 x 10 27 kg, is a product of certain radioactive decays. The results of the experiment led Rutherford to the idea that most of an atom's mass is in a very small nucleus, with electrons in orbit around it. Assume an alpha particle, initially very far from a stationary gold nucleus, is fired with a velocity of 2.98 x 107 m/s directly toward the nucleus (charge +79e). What is the smallest distance between the alpha particle and the nucleus before the alpha particle reverses direction? Assume the gold nucleus remains stationary. fm
In 1911, Ernest Rutherford and his assistants Geiger and Marsden conducted an experiment in which they scattered alpha particles (nuclei of helium atoms) from thin sheets of gold. An alpha particle, having charge +2e and mass 6.64 x 10 27 kg, is a product of certain radioactive decays. The results of the experiment led Rutherford to the idea that most of an atom's mass is in a very small nucleus, with electrons in orbit around it. Assume an alpha particle, initially very far from a stationary gold nucleus, is fired with a velocity of 2.98 x 107 m/s directly toward the nucleus (charge +79e). What is the smallest distance between the alpha particle and the nucleus before the alpha particle reverses direction? Assume the gold nucleus remains stationary. fm
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter9: Relativity
Section: Chapter Questions
Problem 58P
Related questions
Question

Transcribed Image Text:In 1911, Ernest Rutherford and his assistants Geiger and Marsden conducted an experiment in which they scattered alpha particles (nuclei of helium atoms) from thin sheets of gold. An alpha particle, having
charge +2e and mass 6.64 x 10-27 kg, is a product of certain radioactive decays. The results of the experiment led Rutherford to the idea that most of an atom's mass is in a very small nucleus, with electrons in
orbit around it. Assume an alpha particle, initially very far from a stationary gold nucleus, is fired with a velocity of 2.98 × 107 m/s directly toward the nucleus (charge +79e). What is the smallest distance
between the alpha particle and the nucleus before the alpha particle reverses direction? Assume the gold nucleus remains stationary.
fm
Expert Solution
Step 1
Write down the given values using suitable variables.
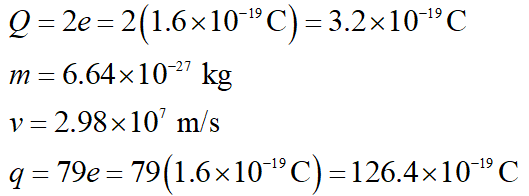
Here, Q signifies alpha-particle charge, e signifies one electron charge, m signifies alpha-particle mass, q signifies gold-particle charge.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Classical Dynamics of Particles and Systems
Physics
ISBN:
9780534408961
Author:
Stephen T. Thornton, Jerry B. Marion
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Classical Dynamics of Particles and Systems
Physics
ISBN:
9780534408961
Author:
Stephen T. Thornton, Jerry B. Marion
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Modern Physics
Physics
ISBN:
9781111794378
Author:
Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

University Physics Volume 3
Physics
ISBN:
9781938168185
Author:
William Moebs, Jeff Sanny
Publisher:
OpenStax