In a generic chemical reaction involving reactants A and B and products C and D, aA + bB¬cC+dD, the standard enthalpy AHm of the reaction is given by ΔΗΔΗ; (C) + dΔH (D) -ΔH (A)- bΔΗ (Β) Notice that the stoichiometric coefficients, a, b, c, d, are an important part of this equation. This formula is often generalized as follows, where the first sum on the right- hand side of the equation is a sum over the products and the second sum is over the reactants: where m and na represent the appropriate stoichiometric coeficients for each substance. Part A What is AHim for the following chemical reaction? H2O(1) + CCL4 (1)→COCI2 (g) + 2HC1(g) You can use the following table of standard heats of formation (AH; ) to calculate the enthalpy of the given reaction. Standard Heat of Standard Heat of Element/ Compound Element/ Compound Formation (kJ/mol) Formation (kJ/mol) H(g) 218 N(g) 473 H2(g) O2(8) CCL (1) -139.5 O(g) 249 H2O(1) -285.8 HCI(g) -92.30kJ C(g) 71 COC2(g) -218.8kJ C(s) HNO:(aq) -206.6
In a generic chemical reaction involving reactants A and B and products C and D, aA + bB¬cC+dD, the standard enthalpy AHm of the reaction is given by ΔΗΔΗ; (C) + dΔH (D) -ΔH (A)- bΔΗ (Β) Notice that the stoichiometric coefficients, a, b, c, d, are an important part of this equation. This formula is often generalized as follows, where the first sum on the right- hand side of the equation is a sum over the products and the second sum is over the reactants: where m and na represent the appropriate stoichiometric coeficients for each substance. Part A What is AHim for the following chemical reaction? H2O(1) + CCL4 (1)→COCI2 (g) + 2HC1(g) You can use the following table of standard heats of formation (AH; ) to calculate the enthalpy of the given reaction. Standard Heat of Standard Heat of Element/ Compound Element/ Compound Formation (kJ/mol) Formation (kJ/mol) H(g) 218 N(g) 473 H2(g) O2(8) CCL (1) -139.5 O(g) 249 H2O(1) -285.8 HCI(g) -92.30kJ C(g) 71 COC2(g) -218.8kJ C(s) HNO:(aq) -206.6
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter5: Thermochemistry
Section: Chapter Questions
Problem 80E: Ethanol, C2H5OH, is used as a fuel for motor vehicles, particularly in Brazil. (a) Write the...
Related questions
Question
100%
Transcribed Image Text:I Review | Constants | Periodic Table
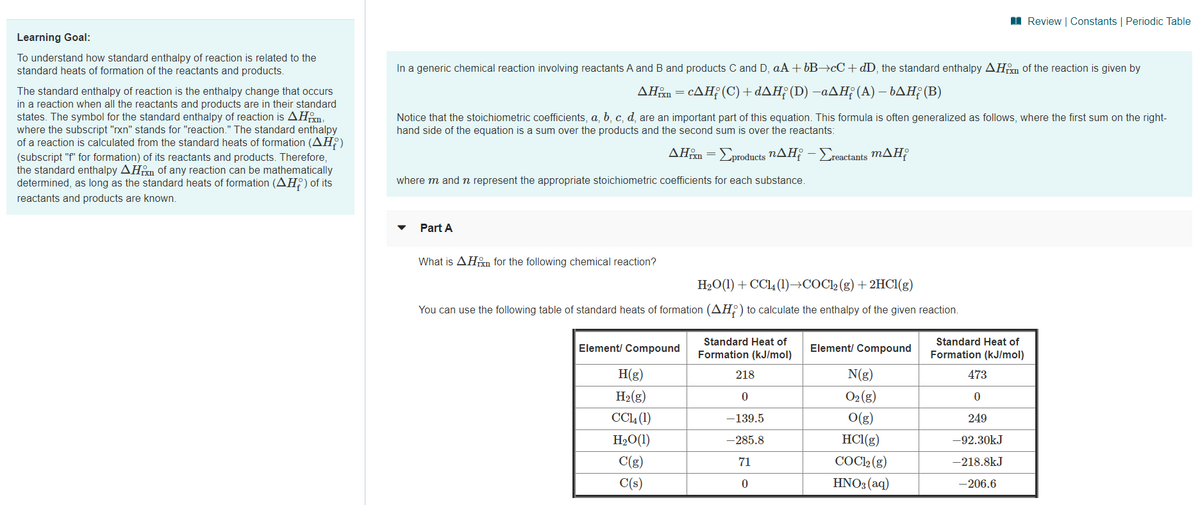
Learning Goal:
To understand how standard enthalpy of reaction is related to the
standard heats of formation of the reactants and products.
In a generic chemical reaction involving reactants A and B and products C and D, aA + 6B→¢C+dD, the standard enthalpy AHm of the reaction is given by
The standard enthalpy of reaction is the enthalpy change that occurs
in a reaction when all the reactants and products are in their standard
states. The symbol for the standard enthalpy of reaction is AHm,
where the subscript "rxn" stands for "reaction." The standard enthalpy
of a reaction is calculated from the standard heats of formation (AH?)
ΔΗΡcΔΗ (C) + dΔΗ; (D) -αΔΗ (Α) - bΔΗ; (Β)
Notice that the stoichiometric coefficients, a, b, c, d, are an important part of this equation. This formula is often generalized as follows, where the first sum on the right-
hand side of the equation is a sum over the products and the second sum is over the reactants:
ΔΗΒΣροducts nΔΗΡ-Σactanis mΔΗ
(subscript "f" for formation) of its reactants and products. Therefore,
the standard enthalpy AHm of any reaction can be mathematically
determined, as long as the standard heats of formation (AH) of its
where m and n represent the appropriate stoichiometric coefficients for each substance.
reactants and products are known.
Part A
What is AHm for the following chemical reaction?
H2O(1) + CCl4 (1)→COCl2 (g) +2HC1(g)
You can use the following table of standard heats of formation (AH;) to calculate the enthalpy of the given reaction.
Standard Heat of
Standard Heat of
Element/ Compound
Element/ Compound
Formation (kJ/mol)
Formation (kJ/mol)
H(g)
218
N(g)
473
H2(g)
O2 (g)
CCL (1)
-139.5
O(g)
249
H2O(1)
-285.8
HC1(g)
-92.30kJ
C(g)
71
COCl2 (g)
-218.8kJ
C(s)
HNO3 (aq)
-206.6
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning