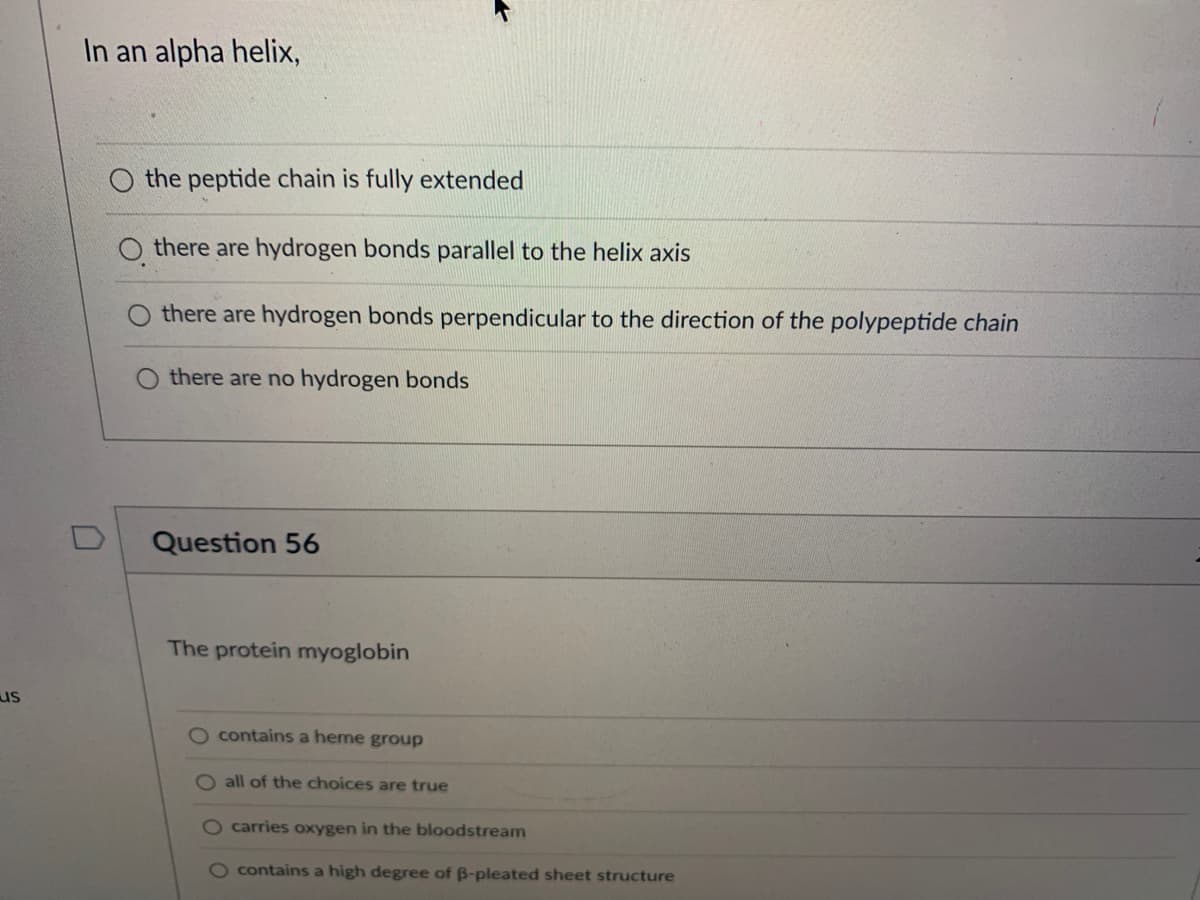
In an alpha helix, U the peptide chain is fully extended there are hydrogen bonds parallel to the helix axis there are hydrogen bonds perpendicular to the direction of the polypeptide chain O there are no hydrogen bonds Question 56 The protein myoglobin O contains a heme group O all of the choices are true O carries oxygen in the bloodstream O contains a high degree of B-pleated sheet structure
In an alpha helix, U the peptide chain is fully extended there are hydrogen bonds parallel to the helix axis there are hydrogen bonds perpendicular to the direction of the polypeptide chain O there are no hydrogen bonds Question 56 The protein myoglobin O contains a heme group O all of the choices are true O carries oxygen in the bloodstream O contains a high degree of B-pleated sheet structure
Biology: The Dynamic Science (MindTap Course List)
4th Edition
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Chapter3: Biological Molecules: The Carbon Compounds Of Life
Section: Chapter Questions
Problem 9TYK: In living systems: a. proteins rarely combine with other macromolecules. b. enzymes are always...
Related questions
Question
Transcribed Image Text:us
In an alpha helix,
0
the peptide chain is fully extended
Othere are hydrogen bonds parallel to the helix axis
there are hydrogen bonds perpendicular to the direction of the polypeptide chain
O there are no hydrogen bonds
Question 56
The protein myoglobin
O contains a heme group
O all of the choices are true
carries oxygen in the bloodstream
O contains a high degree of ß-pleated sheet structure
Transcribed Image Text:LIS
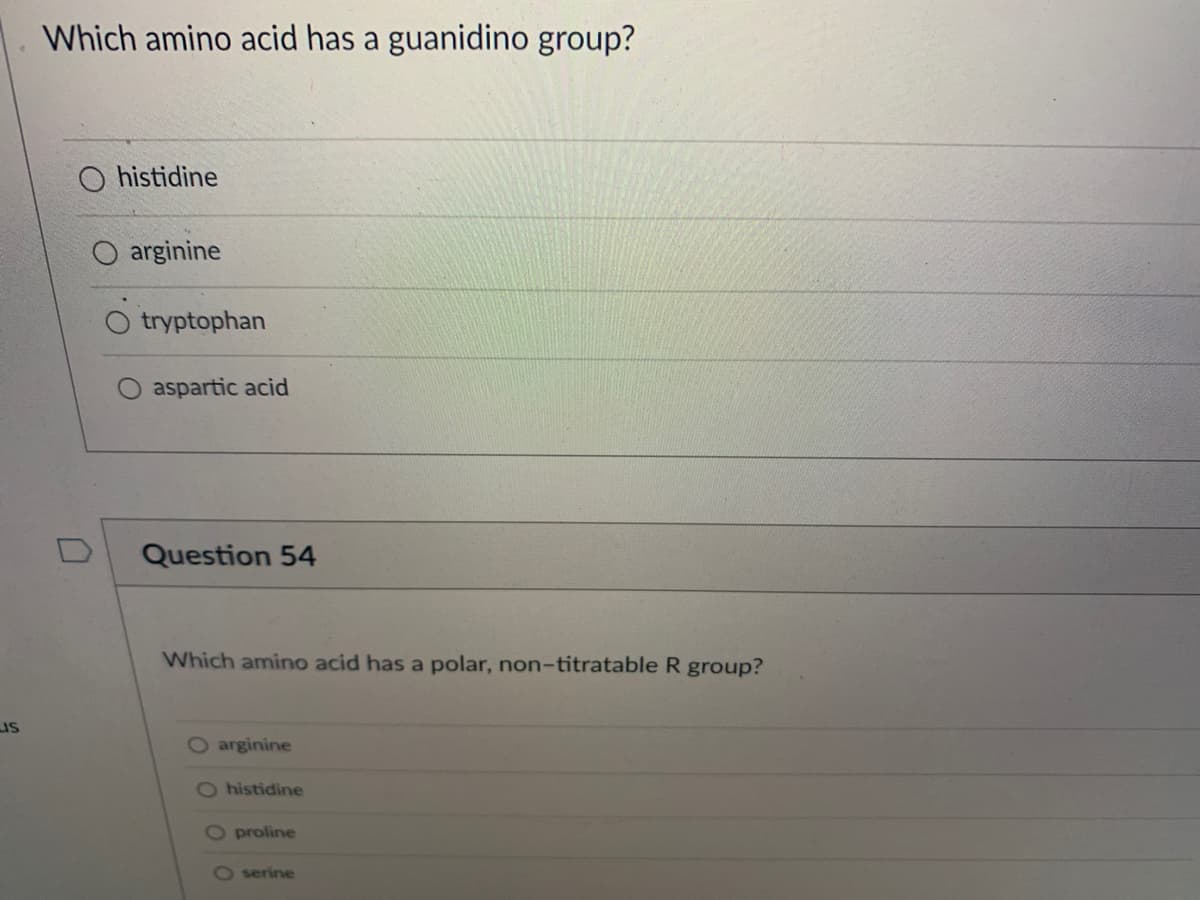
Which amino acid has a guanidino group?
U
histidine
arginine
tryptophan
aspartic acid
Question 54
Which amino acid has a polar, non-titratable R group?
O arginine
O histidine
O proline
O serine
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning