In the carbon skeleton structure below, identify the carbon atom(s) with sp hybridization by clicking on each one. +amnt 2 SUBMIT A Smartwork5 - Mozi... DELL How To Print 1. Print via the internet. MS Word or Pages files must be uploaded to your Google Drive. on the toolbar or select "print" from the menu bar (File >> 2. Click on the print icon Print) 1e change document destination.
In the carbon skeleton structure below, identify the carbon atom(s) with sp hybridization by clicking on each one. +amnt 2 SUBMIT A Smartwork5 - Mozi... DELL How To Print 1. Print via the internet. MS Word or Pages files must be uploaded to your Google Drive. on the toolbar or select "print" from the menu bar (File >> 2. Click on the print icon Print) 1e change document destination.
Chapter9: Covalent Bonding: Orbitals
Section: Chapter Questions
Problem 11Q: Why are d orbitals sometimes used to form hybrid orbitals? Which period of elements does not used...
Related questions
Question
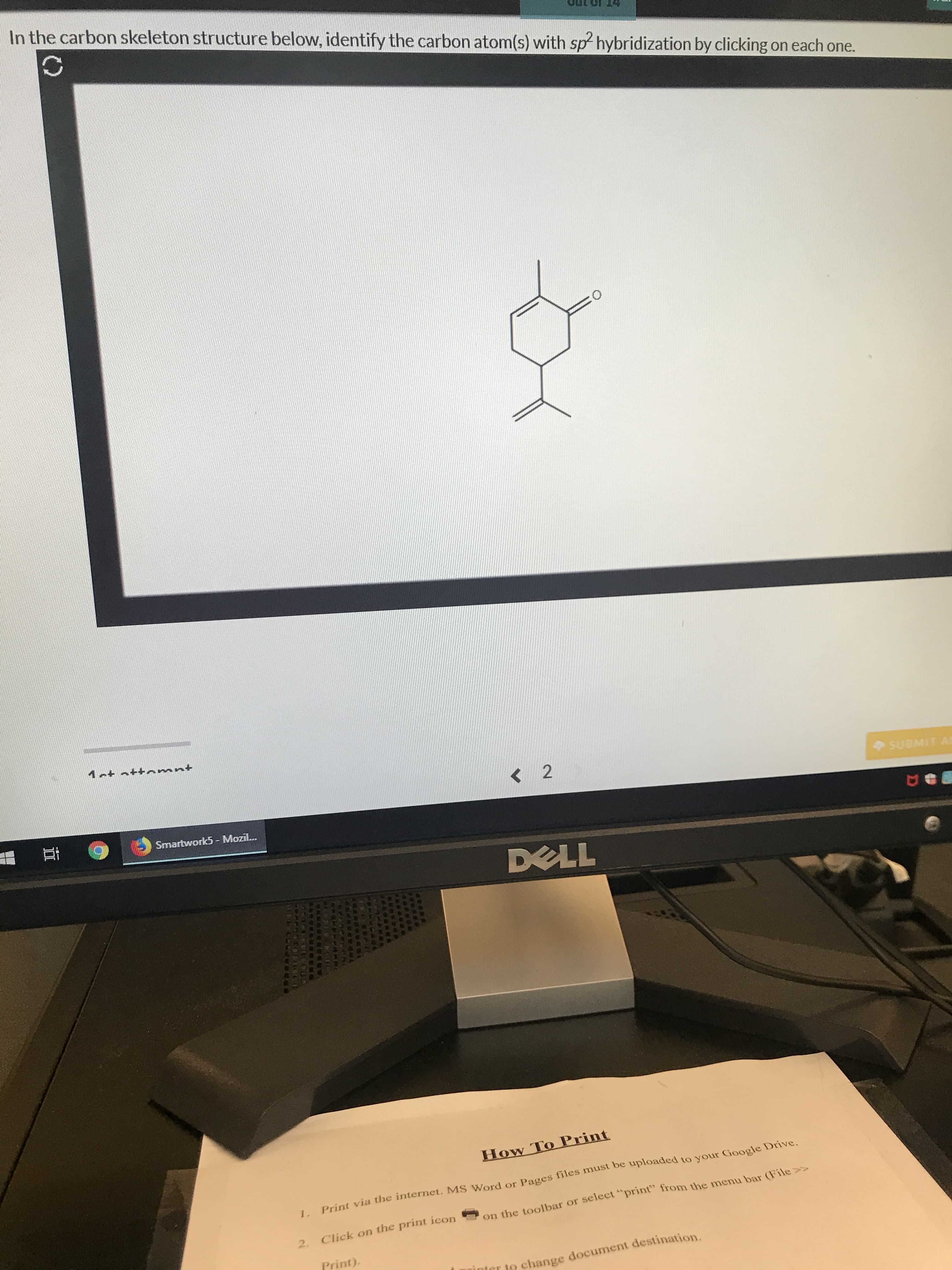
Transcribed Image Text:In the carbon skeleton structure below, identify the carbon atom(s) with sp hybridization by clicking on each one.
+amnt
2
SUBMIT A
Smartwork5 - Mozi...
DELL
How To Print
1. Print via the internet. MS Word or Pages files must be uploaded to your Google Drive.
on the toolbar or select "print" from the menu bar (File >>
2. Click on the print icon
Print)
1e change document destination.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning